Abstract
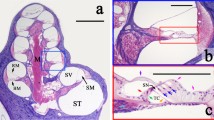
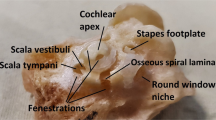
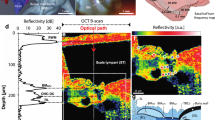
Since it has been difficult to directly observe the morphology of the living cochlea, our ability to infer the mechanical functioning of the living ear has been limited. Nearly all our knowledge about cochlear morphology comes from postmortem tissue that was fixed and processed using procedures that possibly distort the structures and fluid spaces of the organ of Corti. In this study, optical coherence tomography was employed to obtain volumetric images of the high-frequency hook region of the gerbil cochlea, as viewed through the round window, with far better resolution capability than had been possible before. The anatomical structures and fluid spaces of the organ of Corti were segmented and quantified in vivo and over a 90-min postmortem period. We find that the arcuate-zone and pectinate-zone widths change very little postmortem. The volume of the scala tympani between the round-window membrane and basilar membrane and the volume of the inner spiral sulcus decrease in the first 60-min postmortem. While textbook drawings of the mammalian organ of Corti and cortilymph prominently depict the tunnel of Corti, the outer tunnel is typically missing. This is likely because textbook drawings are typically made from images obtained by histological methods. Here, we show that the outer tunnel is nearly twice as big as the tunnel of Corti or the space of Nuel. This larger outer tunnel fluid space could have a substantial, little-appreciated effect on cochlear micromechanics. We speculate that the outer tunnel forms a resonant structure that may affect reticular-lamina motion.










Similar content being viewed by others
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper. Additional data related to this paper may be requested from the authors.
References
Cooper NP, Vavakou A, van der Heijden M (2018) Vibration hotspots reveal longitudinal funneling of sound-evoked motion in the mammalian cochlea. Nat Commun 9:3054. https://doi.org/10.1038/s41467-018-05483-z
Echteler SM, Fay RR, Popper AN (2012) Structure of the mammalian cochlea. In: Comparative Hearing: Mammals. Springer Science & Business Media pp 134–171
Edge RM, Evans BN, Pearce M et al (1998) Morphology of the unfixed cochlea. Hear Res 124:1–16
Fallah E, Strimbu CE, Olson ES (2019) Nonlinearity and amplification in cochlear responses to single and multi-tone stimuli. Hear Res 377:271–281. https://doi.org/10.1016/j.heares.2019.04.001
Finck A, Schneck CD, Hartman AF (1972) Development of cochlear function in the neonate Mongolian gerbil (Meriones unguiculatus). J Comp Physiol Psychol 78:375
Gao SS, Wang R, Raphael PD et al (2014) Vibration of the organ of Corti within the cochlear apex in mice. J Neurophysiol 112:1192–1204. https://doi.org/10.1152/jn.00306.2014
Guinan JJ (2020) The interplay of organ-of-Corti vibrational modes, not tectorial- membrane resonance, sets outer-hair-cell stereocilia phase to produce cochlear amplification. Hear Res 395:108040. https://doi.org/10.1016/j.heares.2020.108040
Gummer AW, Hemmert W, Zenner HP (1996) Resonant tectorial membrane motion in the inner ear: its crucial role in frequency tuning. Proc Natl Acad Sci 93:8727–8732. https://doi.org/10.1073/pnas.93.16.8727
Hardie NA, MacDonald G, Rubel EW (2004) A new method for imaging and 3D reconstruction of mammalian cochlea by fluorescent confocal microscopy. Brain Res 1000:200–210. https://doi.org/10.1016/j.brainres.2003.10.071
Henson MM, Jenkins DB, Henson OW (1983) Sustentacular cells of the organ of Corti — the tectal cells of the outer tunnel. Hear Res 10:153–166. https://doi.org/10.1016/0378-5955(83)90051-5
Homer M, Champneys A, Hunt G, Cooper N (2004) Mathematical modeling of the radial profile of basilar membrane vibrations in the inner ear. The Journal of the Acoustical Society of America 116:1025–1034. https://doi.org/10.1121/1.1771571
Huang D, Swanson EA, Lin CP et al (1991) Optical coherence tomography. Science 254:1178–1181
Hubbard AE, Mountain DC (2012) Analysis and synthesis of cochlea mechanical function using models. In: Auditory computation. Springer Science & Business Media pp 62–120
Hu X, Evans BN, Dallos P (1999) Direct visualization of organ of Corti kinematics in a hemicochlea. J Neurophysiol 82:2798–2807
Iyer JS, Batts SA, Chu KK et al (2016) Micro-optical coherence tomography of the mammalian cochlea. Sci Rep 6:33288. https://doi.org/10.1038/srep33288
Jackson RP (2014) Rotational axes and lever arms for the middle-ear ossicles of several land mammals. Dissertation, Stanford University
Kapuria S, Steele CR, Puria S (2017) Unraveling the mystery of hearing in gerbil and other rodents with an arch-beam model of the basilar membrane. Sci Rep 7:228. https://doi.org/10.1038/s41598-017-00114-x
Lee HY, Raphael PD, Park J et al (2015) Noninvasive in vivo imaging reveals differences between tectorial membrane and basilar membrane traveling waves in the mouse cochlea. Proc Natl Acad Sci 112:3128–3133. https://doi.org/10.1073/pnas.1500038112
Lee HY, Raphael PD, Xia A et al (2016) Two-dimensional cochlear micromechanics measured in vivo demonstrate radial tuning within the mouse organ of Corti. J Neurosci 36:8160–8173. https://doi.org/10.1523/JNEUROSCI.1157-16.2016
Lim DJ, Anniko M (1985) Developmental morphology of the mouse inner ear: a scanning electron microscopic observation. Acta Otolaryngol 99:5–69. https://doi.org/10.3109/00016488509121766
Lukashkin AN, Richardson GP, Russell IJ (2010) Multiple roles for the tectorial membrane in the active cochlea. Hear Res 266:26–35. https://doi.org/10.1016/j.heares.2009.10.005
Meenderink SWF, Shera CA, Valero MD et al (2019) Morphological immaturity of the neonatal organ of Corti and associated structures in humans. J Assoc Res Otolaryngol 20:461–474. https://doi.org/10.1007/s10162-019-00734-2
Merchant SN, Nadol JB (2010) Schuknecht’s pathology of the ear, Third Edition. People’s Medical Publishing House
Müller M (1996) The cochlear place-frequency map of the adult and developing Mongolian gerbil. Hear Res 94:148–156
Nankali A, Wang Y, Strimbu CE et al (2020) A role for tectorial membrane mechanics in activating the cochlear amplifier. Sci Rep 10:17620. https://doi.org/10.1038/s41598-020-73873-9
O’Malley JT, Merchant SN, Burgess BJ et al (2009) Effects of fixative and embedding medium on morphology and immunostaining of the cochlea. Audiol Neurotology 14:78–87. https://doi.org/10.1159/000158536
Overstreet EH, Ruggero MA (1998) Basilar membrane mechanics at the hook region of the Mongolian gerbil cochlea. Assoc Res Otolaryngol mid-Winter Meet Abst 21:181
Peterson LC, Bogert BP (1950) Erratum: a dynamical theory of the cochlea. J Acoust Soc 22:640–640. https://doi.org/10.1121/1.1906668
Plassmann W, Peetz W, Schmidt M (1987) The Cochlea in Gerbilline Rodents BBE 30:82–102. https://doi.org/10.1159/000118639
Richter C-P, Edge R, He DZZ, Dallos P (2000) Development of the gerbil inner ear observed in the hemicochlea. J Assoc Res Otolaryngol 1:195–210. https://doi.org/10.1007/s101620010019
Sasmal A, Grosh K (2019) Unified cochlear model for low- and high-frequency mammalian hearing. Proc Natl Acad Sci 116:13983–13988. https://doi.org/10.1073/pnas.1900695116
Schweitzer L, Lutz C, Hobbs M, Weaver SP (1996) Anatomical correlates of the passive properties underlying the developmental shift in the frequency map of the mammalian cochlea. Hear Res 97:84–94. https://doi.org/10.1016/S0378-5955(96)80010-4
Secomb TW (1978) Flow in a channel with pulsating walls. J Fluid Mech 88:273–288. https://doi.org/10.1017/S0022112078002104
Souter M, Nevill G, Forge A (1995) Postnatal development of membrane specialisations of gerbil outer hair cells. Hear Res 91:43–62. https://doi.org/10.1016/0378-5955(95)00163-8
Souter M, Nevill G, Forge A (1997) Postnatal maturation of the organ of Corti in gerbils: morphology and physiological responses. J Comp Neurol 386:635–651
Spicer SS, Schulte BA (1994) Ultrastructural differentiation of the first Hensen cell in the gerbil cochlea as a distinct cell type. Anat Rec 240:149–156. https://doi.org/10.1002/ar.1092400202
Spicer SS, Smythe N, Schulte BA (2003) Ultrastructure indicative of ion transport in tectal, Deiters, and tunnel cells: differences between gerbil and chinchilla basal and apical cochlea. Anat Rec 271A:342–359. https://doi.org/10.1002/ar.a.10041
Steele CR, Taber LA (1979) Comparison of WKB calculations and experimental results for three-dimensional cochlear models. J Acoust Soc 65:1007–1018. https://doi.org/10.1121/1.382570
Von Békésy G (1960) Experiments in hearing. McGraw, New York
Xia A, Udagawa T, Raphael PD et al (2018) Basilar membrane vibration after targeted removal of the third row of OHCs and Deiters cells. St Catharines, Canada p 020004. https://doi.org/10.1063/1.5038451
Yoon Y-J, Steele CR, Puria S (2011) Feed-forward and feed-backward amplification model from cochlear cytoarchitecture: an interspecies comparison. Biophys J 100:1–10. https://doi.org/10.1016/j.bpj.2010.11.039
Acknowledgements
We thank the MEE Otopathology staff and Anbuselvan Dharmarajan for preparing histology slides; Michael E. Ravicz and John J. Guinan, Jr., for technical support and assistance with cochlear-sensitivity measurements; Kevin N. O’Connor, Heidi H. Nakajima, John J. Guinan, Jr., and Rachel Chen for editing assistance; M. Charles Liberman and Jennifer T. O’Malley for helpful discussions regarding the histology measurements; and Andrew A. Tubelli for the OoC-structure drawings (Figs. 1b, d and 10). We thank Kovid T. Puria for help with statistical analyses.
Funding
This work was supported in part by Grant R01 DC07910 from the National Institute on Deafness and Other Communication Disorders (NIDCD) of the NIH, and the Amelia Peabody Charitable Fund.
Author information
Authors and Affiliations
Contributions
N.H.C. and S.P conceived of and designed the project. N.H.C developed the VibOCT software, conducted the experiments, and reconstructed the 3D OCT volumes. H.W. performed the segmentation and analysis of the 3D volumes from OCT and histology. S.P, N.H.C, and H.W. wrote and edited the manuscript, and S.P. supervised the project.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MPG 4750 KB)
Supplementary file2 (MPG 4318 KB)
Rights and permissions
About this article
Cite this article
Cho, N.H., Wang, H. & Puria, S. Cochlear Fluid Spaces and Structures of the Gerbil High-Frequency Region Measured Using Optical Coherence Tomography (OCT). JARO 23, 195–211 (2022). https://doi.org/10.1007/s10162-022-00836-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10162-022-00836-4