Abstract
Background
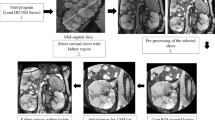
We assessed the effectiveness and convenience of a novel semi-automatic kidney volume (KV) measuring high-speed 3D-image analysis system SYNAPSE VINCENT® (Fuji Medical Systems, Tokyo, Japan) for autosomal dominant polycystic kidney disease (ADPKD) patients.
Methods
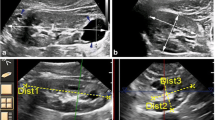
We developed a novel semi-automated KV measurement software for patients with ADPKD to be included in the imaging analysis software SYNAPSE VINCENT®. The software extracts renal regions using image recognition software and measures KV (VINCENT KV). The algorithm was designed to work with the manual designation of a long axis of a kidney including cysts. After using the software to assess the predictive accuracy of the VINCENT method, we performed an external validation study and compared accurate KV and ellipsoid KV based on geometric modeling by linear regression analysis and Bland–Altman analysis.
Results
Median eGFR was 46.9 ml/min/1.73 m2. Median accurate KV, Vincent KV and ellipsoid KV were 627.7, 619.4 ml (IQR 431.5–947.0) and 694.0 ml (IQR 488.1–1107.4), respectively. Compared with ellipsoid KV (r = 0.9504), Vincent KV correlated strongly with accurate KV (r = 0.9968), without systematic underestimation or overestimation (ellipsoid KV; 14.2 ± 22.0%, Vincent KV; − 0.6 ± 6.0%). There were no significant slice thickness-specific differences (p = 0.2980).
Conclusions
The VINCENT method is an accurate and convenient semi-automatic method to measure KV in patients with ADPKD compared with the conventional ellipsoid method.




Similar content being viewed by others
References
Chapman AB, Guay-Woodford L, Grantham JJ, et al. Renal structure in early autosomal dominant polycystic kidney disease: the Consortium for Radiologic Imaging Studies of Polycystic Kidney Disease (CRISP) cohort. Kidney Int. 2003;64:1035–45.
Bae KT, Zhu F, Chapman AB, et al. Magnetic resonance imaging evaluation of hepatic cysts in early autosomal-dominant polycystic kidney disease: the Consortium for Radiologic Imaging Studies of Polycystic Kidney Disease cohort. Clin J Am Soc Nephrol. 2006;1:64–9.
Grantham JJ, Torres VE, Chapman AB, et al. Volume progression in polycystic kidney disease. N Engl J Med. 2006;354:2122–30.
Chapman AB, Bost JE, Torres VE, et al. Kidney volume and functional outcomes in autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol. 2012;7:479–86.
Tokiwa S, Muto S, China T, Horie S. The relationship between renal volume and renal function in autosomal dominant polycystic kidney disease. Clin Exp Nephrol. 2011;15:539–45.
Bae KT, Grantham JJ. Imaging for the prognosis of autosomal dominant polycystic kidney disease. Nat Rev Nephrol. 2010;6:96–106.
King BF, Reed JE, Bergstralh EJ, Sheedy PF 2nd, Torres VE. Quantification and longitudinal trends of kidney, renal cyst, and renal parenchyma volumes in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 2000;11:1505–11.
Kistler AD, Poster D, Krauer F, et al. Increases in kidney volume in autosomal dominant polycystic kidney disease can be detected within 6 months. Kidney Int. 2009;75:235–41.
Bae KT, Tao C, Wang J, et al. Novel approach to estimate kidney and cyst volumes using mid-slice magnetic resonance images in polycystic kidney disease. Am J Nephrol. 2013;38:333–41.
Bae KT, Commean PK, Lee J. Volumetric measurement of renal cysts and parenchyma using MRI: phantoms and patients with polycystic kidney disease. J Comput Assist Tomogr. 2000;24:614–9.
Bae KT, Tao C, Zhu F, et al. Consortium for Radiologic Imaging Studies Polycystic Kidney Disease: MRI-based kidney volume measurements in ADPKD: reliability and effect of gadolinium enhancement. Clin J Am Soc Nephrol. 2009;4:719–25.
Irazabal MV, Rangel LJ, Bergstralh EJ, et al. A simple model for selecting patients for clinical trials. J Am Soc Nephrol. 2015;26:160–72.
Matsuo S, Imai E, Horio M, et al. Collaborators developing the Japanese equation for estimated GFR: revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
R Development Core Team. R. a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing, 2011.
Spithoven EM, van Gastel MD, Messchendorp AL, et al. DIPAK Consortium: estimation of total kidney volume in autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2015;66:792–801.
Cheong B, Muthupillai R, Rubin MF, Flamm SD. Normal values for renal length and volume as measured by magnetic resonance imaging. Clin J Am Soc Nephrol. 2007;2:38–45.
Mignani R, Corsi C, De Marco M, et al. Assessment of kidney volume in polycystic kidney disease using magnetic resonance imaging without contrast medium. Am J Nephrol. 2011;33:176–84.
Cohen BA, Barash I, Kim DC, Sanger MD, Babb JS, Chandarana H. Intraobserver and interobserver variability of renal volume measurements in polycystic kidney disease using a semiautomated MR segmentation algorithm. Am J Roentgenol. 2012;199:387–93.
Bae K, Park B, Sun H, et al. Segmentation of individual renal cysts from MR images in patients with autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol. 2013;8:1089–97.
Kim Y, Ge Y, Tao C, et al. Automated segmentation of kidneys from MR images in patients with autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol. 2016;11:576–84.
Mochizuki K, Takatsuki M, Soyama A, Hidaka M, Obatake M, Eguchi S. The usefulness of a high-speed 3D-image analysis system in pediatric living donor liver transplantation. Ann Transplant. 2012;17:31–4.
Acknowledgements
This study was supported in part by Grant-in-Aid for Intractable Renal Diseases Research, Research on rare and intractable diseases, Health and Labour Sciences Research Grants from the Ministry of Health, Labour and Welfare of Japan. We are grateful to Kyoko Suzuki and Jun Masumoto (Fuji Medical Systems, Tokyo, Japan) for developing novel software SYNAPSE VINCENT® to measure KV in patients with ADPKD.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted (IRB Approval number 16-033 and 16-188) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
About this article
Cite this article
Muto, S., Kawano, H., Isotani, S. et al. Novel semi-automated kidney volume measurements in autosomal dominant polycystic kidney disease. Clin Exp Nephrol 22, 583–590 (2018). https://doi.org/10.1007/s10157-017-1486-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-017-1486-6