Abstract
Background
This pilot study aimed to assess whether the perioperative infusion of donor bone marrow cells (DBMC) in renal allograft recipients can affect the appearance of peripheral regulatory T-cell subsets and the profile of cytokine-producing cells [interferon-gamma (IFN-γ), interleukin (IL)-17 and IL-10] 2 years after transplantation.
Methods
Fresh blood samples were collected from 14 kidney recipients who received infusion and from 13 kidney recipients without infusion who served as controls at the end of the second post-transplantation year. Initially the percentages of CD4+CD25+FoxP3+ T cells and CD3+CD8+CD28− T cells were quantified using flowcytometry. Thereafter, the frequencies of IL-10-, IL-17- and IFN-γ-producing cells were determined separately using the ELISPOT technique with peptides corresponding to mismatched donor HLA-DR molecules and phytohemagglutinin (PHA).
Results
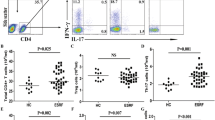
The mean numbers of IFN-γ- and IL-17-producing cells in response to PHA were lower in infused patients than in controls (P = 0.02 and P = 0.18, respectively); however, an increased frequency of IL-10-producing cells was observed compared to controls (P = 0.07). Furthermore, the ratio of IL-10/IFN-γ-producing cells was significantly higher in the DBMC-infused group versus controls (P = 0.01). There was a negative correlation between the percentage of CD3+CD8+CD28−T cells and IL-17-producing cells in the infused group (r = −0.539, P = 0.04). The mean levels and the frequency of microchimerism within the first post-transplantation year were also significantly higher in infused patients than in controls (P = 0.007 and P = 0.001, respectively).
Conclusion
Our findings suggest that DBMC infusion could partially stimulate the regulatory mechanisms against alloimmune responses in kidney allograft recipients.






Similar content being viewed by others
References
Yong Z, Chang L, Mei YX, Yi L. Role and mechanism of CD4+CD25+ regulatory T cells in the induction and maintenance of transplantation tolerance. Transpl Immunol. 2007;17:120–9.
Hendrikx TK, van Gurp EA, Sewgobind VD, Mol WM, Schoordijk W, Klepper M, et al. Generation of donor-specific regulatory T-cell function in kidney transplant patients. Transplantation. 2009;87:376–83.
Delis S, Ciancio G, Burke GW 3rd, Garcia-Morales R, Miller J. Donor bone marrow transplantation: chimerism and tolerance. Transpl Immunol. 2004;13:105–15.
Scandling JD, Busque S, Dejbakhsh-Jones S, Benike C, Millan MT, Shizuru JA, et al. Tolerance and chimerism after renal and hematopoietic-cell transplantation. N Engl J Med. 2008;358:362–8.
Monaco AP. The beginning of clinical tolerance in solid organ allografts. Exp Clin Transplant. 2004;2:153–61.
De Pauw L, Toungouz M, Goldman M. Infusion of donor-derived hematopoietic stem cells in organ transplantation: clinical data. Transplantation. 2003;75:46S–9S.
Garcia-Morales R, Esquenazi V, Zucker K, Gomez CI, Fuller L, Carreno M, et al. An assessment of the effects of cadaver donor bone marrow on kidney allograft recipient blood cell chimerism by a novel technique combining PCR and flow cytometry. Transplantation. 1996;62:1149–60.
Ciancio G, Miller J, Garcia-Morales RO, Carreno M, Burke GW III, Roth D, Kupin W, et al. Six-year clinical effect of donor bone marrow infusions in renal transplant patients. Transplantation. 2001;71:827–35.
Ciancio G, Burke GW, Garcia-Morales R, Suzart K, Rosen A, Ricordi C, et al. Effect of living related donor bone marrow infusion on chimerism and in vitro immunoregulatory activity in kidney transplant recipients. Transplantation. 2002;74:488–96.
Spitzer TR, Delmonico F, Tolkoff-Rubin N, McAfee S, Sackstein R, Saidman S, et al. Combined histocompatibility leukocyte antigen-matched donor bone marrow and renal transplantation for multiple myeloma with end stage renal disease: the induction of allograft tolerance through mixed lymphohematopoietic chimerism. Transplantation. 1999;68:480–4.
Bonilla WV, Geuking MB, Aichele P, Ludewig B, Hengartner H, Zinkernagel RM. Microchimerism maintains deletion of the donor cell-specific CD8+ T cell repertoire. J Clin Invest. 2006;116:156–62.
Mathew JM, Garcia-Morales RO, Carreno M, Jin Y, Fuller L, Blomberg B, et al. Immune responses and their regulation by donor bone marrow cells in clinical organ transplantation. Transpl Immunol. 2003;11:307–21.
Brown K, Wong W. Diagnostic value of regulatory T cells: a new facet of a much studied cell population. Transplantation. 2008;86:1485–91.
Adeegbe D, Bayer AL, Levy RB, Malek TR. Cutting edge: allogeneic CD4+CD25+Foxp3+ T regulatory cells suppress autoimmunity while establishing transplantation tolerance. J Immunol. 2006;176:7149–53.
Colovai AI, Mirza M, Vlad G, Wang S, Ho E, Cortesini R, et al. Regulatory CD8+CD28− T cells in heart transplant recipients. Hum Immunol. 2003;64:31–7.
Kingsley CI, Karim M, Bushell AR, Wood KJ. CD25+ CD4+ regulatory T cells prevent graft rejection: CTLA-4- and IL10-dependent immunoregulation of alloresponses. J Immunol. 2002;168:1080–6.
Akl A, Jones ND, Rogers N, Bakr MA, Mostafa A, El Shehawy el M, et al. An investigation to assess the potential of CD25highCD4 + T cells to regulate responses to donor alloantigens in clinically stable renal transplant recipients. Transpl Int. 2008;21:65–73.
Bestard O, Cruzado JM, Mestre M, Caldés A, Bas J, Carrera M, et al. Achieving donor-specific hyporesponsiveness is associated with FOXP3+ regulatory T cell recruitment in human renal allograft infiltrates. J Immunol. 2007;179:4901–9.
Mitchell P, Afzali B, Lombardi G, Lechler RI. The T helper 17-regulatory T cell axis in transplant rejection and tolerance. Curr Opin Organ Transplant. 2009;14:326–31.
Chadha R, Heidt S, Jones ND, Wood KJ. Th17: contributors to allograft rejection and a barrier to the induction of transplantation tolerance? Transplantation. 2011;91:939–45.
Ashton-Chess J, Dugast E, Colvin RB, Giral M, Foucher Y, Moreau A, et al. Regulatory, effector, and cytotoxic T cell profiles in long-term kidney transplant patients. J Am Soc Nephrol. 2009;20:1113–22.
Taflin C, Nochy D, Hill G, Frouget T, Rioux N, Vérine J, et al. Regulatory T cells in kidney allograft infiltrates correlate with initial inflammation and graft function. Transplantation. 2010;89:194–9.
Solgi G, Pourmand G, Mehrsai A, Taherimahmoudi M, Nicknam MH, Ebrahimi Rad MR, et al. Anti-HLA antibodies and kidney allograft outcomes in recipients with donor bone marrow cell infusion. Iran J Immunol. 2010;7:18–29.
Salama AD, Najafian N, Clarkson MR, Harmon WE, Sayegh MH. Regulatory CD25+ T cells in human kidney transplant recipients. J Am Soc Nephrol. 2003;14:1643–51.
Solgi G, Mytilineos J, Gadi VK, Biswajit P, Pourmand G, Mehrsai A, et al. Donor derived peripheral mononuclear cell DNA is associated with stable kidney allograft function: a randomized controlled trial. Chimerism. 2011; 2:102–110.
Solgi G, Amirzargar AA, Pourmand G, Mehrsai AR, Taherimahmoudi M, Baradaran N, et al. TH1/TH2 cytokines and soluble CD30 levels in kidney allograft patients with donor bone marrow cell infusion. Transpl Proc. 2009;41:2800–4.
Noris M, Azzollini N, Mister M, Pezzotta A, Piccinini G, Casiraghi F, et al. Peripheral donor leukocytes prolong survival of rat renal allograft. Kidney Int. 1999;56:1101–12.
Wekerle T, Kurtz J, Ito H, Ronquillo JV, Dong V, Zhao G, et al. Allogeneic bone marrow transplantation with co-stimulatory blockade induces macrochimerism and tolerance without cytoreductive host treatment. Nat Med. 2000;6:464–9.
Seung E, Mordes JP, Rossini AA, Greiner DL. Hematopoietic chimerism and central tolerance created by peripheral-tolerance induction without myeloablative conditioning. J Clin Invest. 2003;112:795–808.
Mathew JM, Garcia-Morales R, Fuller L, Rosen A, Ciancio G, Burke GW, et al. Donor bone marrow-derived chimeric cells present in renal transplant recipients infused with donor marrow. I. Potent regulators of recipient antidonor immune responses. Transplantation. 2000;70:1675–82.
Cirocco RE, Carreño MR, Mathew JM, Garcia-Morales RO, Fuller L, Esquenazi V, et al. FoxP3 mRNA transcripts and regulatory cells in renal transplant recipients 10 years after donor marrow infusion. Transplantation. 2007;83:1611–9.
Hara M, Kingsley CI, Niimi M, Read S, Turvey SE, Bushell AR, et al. IL-10 is required for regulatory T cells to mediate tolerance to alloantigens in vivo. J Immunol. 2001;166:3789–96.
Mittal SK, Sharma RK, Gupta A, Naik S. Increased interleukin-10 production without expansion of CD4+ CD25+ T-regulatory cells in early stable renal transplant patients on calcineurin inhibitors. Transplantation. 2009;88:435–41.
Najafian N, Salama AD, Fedoseyeva EV, Benichou G, Sayegh MH. Enzyme-linked immunosorbent spot assay analysis of peripheral blood lymphocyte reactivity to donor HLA-DR peptides: potential novel assay for prediction of outcomes for renal transplant recipients. J Am Soc Nephrol. 2002;13:252–9.
Tsaur I, Gasser M, Aviles B, Lutz J, Lutz L, Grimm M, et al. Donor antigen-specific regulatory T-cell function affects outcome in kidney transplant recipients. Kidney Int. 2011;79:1005–12.
Kawai T, Cosimi AB, Spitzer TR, Tolkoff-Rubin N, Suthanthiran M, Saidman SL, et al. HLA-mismatched renal transplantation without maintenance immunosuppression. N Engl J Med. 2008;358:353–61.
Lin YX, Wang LL, Yan LN, Cai P, Li B, Wen TF, et al. Analysis of CD8+ CD28− T-suppressor cells in living donor liver transplant recipients. Hepatobiliary Pancreat Dis Int. 2009;8:241–6.
Joffre O, Santolaria T, Calise D, Al Saati T, Hudrisier D, Romagnoli P, et al. Prevention of acute and chronic allograft rejection with CD4+ CD25+ Foxp3+ regulatory T lymphocytes. Nat Med. 2008;14:88–92.
Acknowledgments
We are grateful to the staff of the transplantation ward at Sina University Hospital for their kind collaboration in taking the blood samples and collecting clinical data.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ranjbar, M., Solgi, G., Mohammadnia, M. et al. Regulatory T-cell subset analysis and profile of interleukin (IL)-10, IL-17 and interferon-gamma cytokine-producing cells in kidney allograft recipients with donor cells infusion. Clin Exp Nephrol 16, 636–646 (2012). https://doi.org/10.1007/s10157-012-0591-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-012-0591-9