Abstract
Introduction
The aim of this study was to investigate the factors influencing serum parathyroid (PTH) levels, including medications for treating chronic kidney disease–mineral and bone metabolism disorder (CKD-MBD) in patients with end-stage renal disease.
Methods
We enrolled 1,076 patients in nine Japanese facilities who had begun hemodialysis (HD) due to ESRD. We investigated the relationships between intact PTH (iPTH) levels and clinical parameters and medications just prior to beginning HD.
Results
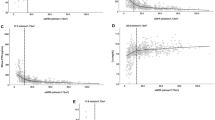
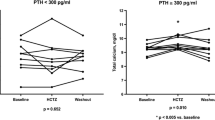
Significant decreases in serum iPTH levels were seen in males, in the presence of diabetes mellitus (DM), and with administration of renin–angiotensin system inhibitors (RASIs). Significant correlations were found between serum calcium and iPTH levels. In the patients administered RASIs, there was a significant decrease in serum iPTH levels with DM, male gender, and administration of active vitamin D sterols (VDs) compared with those not administered RASIs, although serum-corrected calcium levels were not different. Multiple regression analysis found gender, age, presence of DM, and serum calcium and phosphate levels to be significant contributing factors. In addition, administration of angiotensin II receptor blockers (ARBs) may also be a contributing factor to iPTH levels at the beginning HD (p = 0.050).
Conclusions
In this study, serum iPTH levels were related to administration of ARBs besides gender, age, the presence of DM and serum calcium levels. Our study suggests that the RA system involve serum iPTH levels in uremic patients.
Similar content being viewed by others
References
Craver L, Marco MP, Martinez I, Rue M, Borras M, Martin ML, et al. Mineral metabolism parameters throughout chronic kidney disease stages 1–5: achievement of K/DOQI target ranges. Nephrol Dial Transplant. 2007;22:1171–6.
Levin A, Bakris GL, Molitch M, Smulders M, Tian J, Williams LA, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 2007;71:31–8.
Isakova T, Gutierrez O, Shah A, Castaldo L, Holmes J, Lee H, et al. Postprandial mineral metabolism and secondary hyperparathyroidism in early CKD. J Am Soc Nephrol. 2008;19:615–23.
Ben Dov IZ, Galitzer H, Lavi Moshayoff V, Goetz R, Kuro OM, Mohammadi M, et al. The parathyroid is a target organ for FGF23 in rats. J Clin Invest. 2007;117:4003–8.
Smith DH, Johnson ES, Thorp ML, Yang X, Neil N. Hyperparathyroidism in chronic kidney disease: a retrospective cohort study of costs and outcomes. J Bone Miner Metab. 2009;27:287–94.
Kovesdy CP, Ahmadzadeh S, Anderson JE, Kalantar Zadeh K. Secondary hyperparathyroidism is associated with higher mortality in men with moderate to severe chronic kidney disease. Kidney Int. 2008;73:1296–302.
Hase H, Tsunoda T, Tanaka Y, Takahashi Y, Imamura Y, Ishikawa H, et al. Risk factors for de novo acute cardiac events in patients initiating hemodialysis with no previous cardiac symptom. Kidney Int. 2006;70:1142–8.
National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42(Suppl 3):S1–202.
KDIGO Clinical practice guideline for the diagnosis, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int. 2009;76(Suppl 113):S70–S89.
Guideline Working Group, Japanese Society for Dialysis Therapy. Clinical practice guideline for the management of secondary hyperparathyroidism in chronic dialysis patients. Ther Apher Dial. 2008;12:514–25.
Melamed ML, Eustace JA, Plantinga LC, Jaar BG, Fink NE, Parekh RS, et al. Third-generation parathyroid hormone assays and all-cause mortality in incident dialysis patients: the CHOICE study. Nephrol Dial Transplant. 2007;23:1650–8.
Tanaka H, Hamano T, Fujii N, Tomida I, Matsui S, Mikami Y, et al. The impact of diabetes mellitus on vitamin D metabolism in predialysis patients. Bone. 2009;45:949–55.
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Renal study investigators, et al. Effect of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:851–60.
Hou FF, Zhang X, Zhang GH, Xie D, Chen PY, Zhang WR, et al. Efficacy and safety of benazepril for advanced chronic renal insufficiency. N Engl J Med. 2006;354:131–40.
Mizobuchi M, Morrissey J, Finch JL, Martin DR, Liapis H, Akizawa T, et al. Combination therapy with an angiotensin-converting enzyme inhibitor and a vitamin D analog suppresses the progression of renal insufficiency in uremic rat. J Am Soc Nephrol. 2007;18:1796–806.
Porter L, Conlin PR, Scott J, Brown EM, El-Hajj Fuleihan G. Calcium modulation of the renin–aldosterone axis. J Endocrinol Invest. 1999;22:115–21.
Grant FD, Mandel SJ, Brown EM, Williams GH, Seely EW. Interrelationships between the renin–angiotensin–aldosterone and calcium homeostatic systems. J Clin Endocrinol Metab. 1992;75:988–92.
Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin–angiotensin system. J Clin Invest. 2002;110:229–38.
Szeto CC, Chow KM, Kwan BC, Chung KY, Leung CB, Li PK. Oral calcitriol for the treatment of persistent proteinuria in immunoglobulin A nephropathy: an uncontrolled trial. Am J Kidney Dis. 2008;51:724–31.
Zhang Y, Deb DK, Kong J, Ning G, Wang Y, Li G, et al. Long-term therapeutic effect of vitamin D analog doxercalciferol on diabetic nephropathy: strong synergism with AT1 receptor antagonist. Am J Physiol Renal Physiol. 2009;297:F791–801.
Kimata N, Albert JM, Akiba T, Yamazaki S, Kawaguchi Y, Fukuhara S, et al. Association of mineral metabolism factors with all-cause and cardiovascular mortality in hemodialysis patients: the Japan dialysis outcomes and practice patterns study. Hemodial Int. 2007;11:340–8.
Acknowledgments
The authors thank the following investigators, members of the STudy group of Assessing initiation of Renal replacement Therapy (START) who participated in this study: Toshihiko Yamaka (Social Insurance Center General Hospital), Hiroyuki Sekihara (Showa University Fujigaoka Hospital), Moriaki Osaka (St. Luke’s International Hospital), Hiroyuki Miyakawa (Jikei University Aoto Hospital), Okamoto Hiromi (Toho University Ohashi Medical Center), and Kazuhiko Kitano (Musashino Red-cross Hospital).
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Koiwa, F., Komukai, D., Hirose, M. et al. Influence of renin–angiotensin system on serum parathyroid hormone levels in uremic patients. Clin Exp Nephrol 16, 130–135 (2012). https://doi.org/10.1007/s10157-011-0534-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-011-0534-x