Abstract
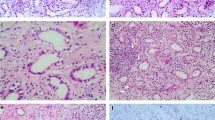
A 40-year-old man was transferred to our hospital because of severe anasarca. He was a heavy drinker for more than 20 years, and diagnosed with diabetes mellitus 8 years earlier and treated with retinal photocoagulation 8 months earlier. He reported loss of appetite after divorce 10 months prior to admission. On admission, he presented with systemic edema and dyspnea. Chest radiography showed massive pleural effusion and cardiomegaly. Serum total protein was 5.6 g/dl, albumin 2.6 g/dl, and urinary protein excretion was 5.3 g/day. Glucose tolerance test showed normal pattern. Ultrafiltration and continuous hemofiltration resulted in loss of 40 kg body weight in 5 days. Echocardiography revealed high-output heart failure and blood tests showed low serum thiamine level of 12 ng/ml (normal, >28 ng/ml). Accordingly, the diagnosis was established as beriberi heart disease complicated with nephrotic syndrome. Treatment with 50 mg/day thiamine intravenously and 80 mg/day furosemide resulted in increase in urine output, decrease in cardiac output, resolution of pulmonary effusion, and about 70 kg body weight loss. Percutaneous renal biopsy showed nodular glomerulosclerosis, mesangial matrix expansion, and thickening of glomerular basement membrane (GBM). Immunofluorescence study showed no glomerular deposition of immunoglobulin or complement. Electron microscopy showed GBM thickening and mesangial matrix deposition without electron-dense deposits or fibrils. These findings were compatible with diabetic glomerulosclerosis. In this patient, extreme malnutrition altered glucose tolerance but, on the other hand, nephrotic syndrome associated with diabetic nephropathy made the diagnosis of beriberi heart disease difficult.


Similar content being viewed by others
References
Koike H, Iijima M, Sugiura M, Mori K, Hattori N, Ito H, et al. Alcoholic neuropathy is clinicopathologically distinct from thiamine-deficiency neuropathy. Ann Neurol. 2003;54:19–29.
Kawano H, Hayashi T, Koide Y, Toda G, Yano K, et al. Histopathological changes of biopsied myocardium in Shoshin beriberi. Int Heart J. 2005;46:751–9.
Wakabayashi A, Yui Y, Kawai C. A clinical study on thiamine deficiency. Jpn Circ J. 1979;43:995–9.
Thomson AD, Jeyasingham MD, Pratt OE. Possible role of toxins in nutritional deficiency. Am J Clin Nutr. 1987;45:1351–60.
Kawai C, Wakabayashi A, Matsumura T, Yui Y. Reappearance of beriberi heart disease in Japan. A study of 23 cases. Am J Med. 1980;69:383–6.
Russell RM. Vitamin and trace mineral deficiency and excess, in Harrison’s principles of internal medicine. 16th ed. New York: McGraw-Hill; 2005. p. 403–4.
Wooley JA. Characteristics of thiamine and its relevance to the management of heart failure. Nutr Clin Pract. 2008;23:487–93.
McCandless DW. Energy metabolism in the lateral vestibular nucleus in pyrithiamine-induced thiamine deficiency. Ann N Y Acad Sci. 1982;378:355–64.
Bakker SJ, Leunissen KM. Hypothesis on cellular ATP depletion and adenosine release as causes of heart failure and vasodilatation in cardiovascular beriberi. Med Hypotheses. 1995;45:265–7.
Seligmann H, Halkin H, Rauchfleisch S, Kaufmann N, Motro M, Vered Z, et al. Thiamine deficiency in patients with congestive heart failure receiving long-term furosemide therapy: a pilot study. Am J Med. 1991;91:151–5.
Blacket RB, Palmer AJ. Haemodynamic studies in high output beriberi. Br Heart J. 1960;22:483–501.
Anderson SH, Charles TJ, Nicol AD. Thiamine deficiency at a district general hospital: Report of five cases. Q J Med. 1985;55:15–32.
Kazama I, Matsubara M, Ejima Y, Michimata M, Suzuki M, Miyama N, et al. Steroid resistance in prolonged type I membranoproliferative glomerulonephritis and accelerated disease remission after steroid withdrawal. Clin Exp Nephrol. 2005;9:62–8.
Preud’homme JL, Aucouturier P, Touchard G, Striker L, Khamlichi AA, Rocca A, et al. Monoclonal immunoglobulin deposition disease: relation with structural abnormalities of immunoglobulin chains. Kidney Int. 1994;46:965–72.
Yoshimura M, Kida H, Saito Y, Yokoyama H, Tomosugi N, Abe T, et al. Peculiar glomerular lesions in Takayasu’s arteritis. Clin Nephrol. 1985;24:120–7.
Sanders PW, Herrera G, Kirk KA, Old CW, Galla JH. Spectrum of glomerular and tubulointerstitial renal lesions associated with monotypical immunoglobulin light chain deposition. Lab Invest. 1991;64:527–37.
Eriksson KF, Lindgarde F. Prevention of type 2 (non-insulin-dependent) diabetes mellitus by diet and physical exercise. The 6-year Malmo feasibility study. Diabetologia. 1991;34:891–8.
Rubenstein AH, Mako ME, Horwitz DL. Insulin and the kidney. Nephron. 1975;15:306–26.
Imamura Y, Yokono K, Shii K, Hari J, Sakai H, Baba S. Plasma levels of proinsulin, insulin and C-peptide in chronic renal, hepatic and muscular disorders. Jpn J Med. 1984;23:3–8.
Acknowledgments
We thank Mr. M. Munakata (Fukuoka Red Cross Hospital) and Ms H. Hayashida (Kyushu University Hospital) for the excellent technical assistance. We also thank Dr. F.G. Issa (http://www.word-medex.com.au) for careful reading and editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Toyonaga, J., Masutani, K., Tsuruya, K. et al. Severe anasarca due to beriberi heart disease and diabetic nephropathy. Clin Exp Nephrol 13, 518–521 (2009). https://doi.org/10.1007/s10157-009-0189-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-009-0189-z