Abstract
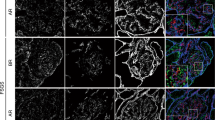
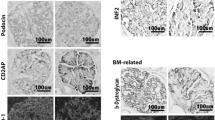
Several recent studies have demonstrated that the slit diaphragm of the glomerular epithelial cell (podocyte) is the structure likely to be the principal barrier in the glomerular capillary wall. Nephrin identified as a gene product mutated in congenital nephrotic syndrome located at the outer leaflet of plasma membranes of the slit diaphragm. The anti-nephrin antibody is capable of inducing massive proteinuria, which indicates that nephrin is a key functional molecule in the slit diaphragm. Expression of nephrin was reduced in glomeruli of minimal change nephrotic syndrome. Some recent studies demonstrated that podocin, CD2-associated protein and NEPH1 are also functional molecules in the slit diaphragm, and their expressions are altered in membranous nephropathy and also in focal glomerulosclerosis. These observations suggested that the alteration of the molecular arrangement in the slit diaphragm is involved in the development of proteinuria in several kinds of glomerular diseases. Recent studies of our group have demonstrated that type 1 receptor-mediated angiotensin II action reduced the expression of the slit diaphragm-associated molecules and that type 1 receptor blockade ameliorated proteinuria by preventing the function of angiotensin II on the slit diaphragm. By the subtraction hybridization techniques using glomerular cDNA of normal and proteinuric rats, we detected that synaptic vesicle protein 2B and ephrin B1 are involved in the maintenance of the barrier function of the slit diaphragm.

Similar content being viewed by others
References
Pavenstadt H, Kriz W, Kretzler M. Cell biology of the glomerular podocyte. Physiol Rev. 2003;83:253–307.
Kawachi H, Shimizu F. Molecular composition and function of the slit diaphragm: nephrin, the molecule responsible for proteinuria. Clin Exp Nephrol. 2000;4:161–72.
Mundel P, Shankland SJ. Podocyte biology and response to injury. J Am Soc Nephrol. 2002;13:3005–15.
Arakawa M. A scanning electron microscopy of the glomerulus of normal and nephrotic rats. Lab Invet. 1970;23:489–97.
Yamada E. The fine structure of the renal glomerulus of the mouse. J Biophys Biochem Cytol. 1955;1:551–6.
Farquhar MG, Wissig SL, Palade GE. Glomerular permeability I. Ferritin transfer across the normal glomerular capillary wall. J Exp Med. 1961;113:47–66.
Rodewald R, Karnovsky MJ. Porous substructure of the glomerular slit diaphragm in the rat and mouse. J Cell Biol. 1974;60:423–33.
Orikasa M, Matsui K, Oite T, Shimizu F. Massive proteinuria induced in rats by a single intravenous injection of a monoclonal antibody. J Immunol. 1988;141:807–14.
Kestilä M, Lenkkeri U, Männikkö M, Lamerdin J, McCready P, Putaala H, et al. Positionally cloned gene for a novel glomerular protein-nephrin- is mutated in congenital nephrotic syndrome. Mol Cell. 1998;1:572–8.
Ruotsalainen V, Ljungberg P, Wartiovaara J, Lenkkeri U, Kestilä M, Jalanko H, et al. Nephrin is specifically located at the site of the slit diaphragm of glomerular podocytes. Proc Natl Acad Sci USA. 1999;96:7962–7.
Boute N, Gribouval O, Roselli S, Benessy F, Lee H, Fuchshuber A, et al. NPHS2, encoding the glomerular protein podocin, is mutated in autosomal recessive steroid-resisitant nephrotic syndrome. Nature Genet. 2000;24:349–54.
Shih NY, Li J, Karpitskii V, Nguyen A, Dustin ML, Kanagawa O, et al. Congenital nephrotic syndrome in mice lacking CD2-associated protein. Science. 1999;286:312–5.
Shih NY, Li J, Karpitskii V, Nguyen A, Dustin ML, Kanagawa O, et al. Proteinuria and perinatal lethality in mice lacking NEPH1, a novel protein with homology to NEPHRIN. Mol Cell Biol. 2001;21:4829–36.
Huber TB, Schmidts M, Gerke P, Schermer B, Zahn A, Hartleben B, et al. NEPH1 difines a novel family of podocin-interacting proteins. FASEB J. 2003;17:115–7.
Winn MP, Conlon PJ, Lynn KL, Farrington MK, Creazzo T, Hawkins AF, et al. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science. 2005;308:1801–4.
Reiser J, Polu KR, Möller CC, Kenlan P, Altintas MM, Wei C, et al. TRPC6 is a glomerular slit diaphragm-associated channel required for normal renal function. Nat Genet. 2005;37:739–44.
Ahola H, Wang S-X, Luimula P, Solin M-L, Holzman LB, Holthofer H. Cloning and expression of the rat nephrin homologue. Am J Pathol. 1999;155:907–13.
Kawachi H, Koike H, Kurihara H, Yaoita E, Orikasa M, Shia MA, et al. Cloning of rat nephrin: expression in developing glomeruli and in proteinuric states. Kidney Int. 2000;57:1949–61.
Furness PN, Hall LL, Shaw JA, Pringle JH. Glomerular expression of nephrin is decreased in acquired human nephrotic syndrome. Nephrol Dial Transplant. 1999;14:1234–7.
Kawachi H, Koike H, Kurihara H, Sakai T. Shimizu F: Cloning of rat homologue of podocin: expression in proteinuric states and in developing glomeruli. J. Am Soc Nephrol. 2003;14:46–56.
Doublier S, Ruotsalainen V, Salvidio G, Lupia E, Biancone L, Conaldi PG, et al. Nephrin redistribution on podocytes is a potential mechanism for proteinuria in patients with primary acquired nephrotic syndrome. Am J Pathol. 2001;158:1723–31.
Horinouchi I, Nakazato H, Kawano T, Iyama K, Furuse A, Arizono K, et al. In situ evaluation of podocin in normal and glomerular diseases. Kidney Int. 2003;64:2092–9.
Wernerson A, Dunér F, Pettersson E, Widholm SM, Berg U, Ruotsalainen V, et al. Altered ultrastructural distribution of nephrin in minimal change nephrotic syndrome. Nephrol Dial Transplant. 2003;18:70–6.
Yuan H, Takeuchi E, Taylor GA, McLaughlin M, Brown D, Salant DJ. Nephrin dissociates from actin, and its expression is reduced in early experimental membranous nephropathy. J Am Soc Nephrol. 2002;13:946–56.
Nakatsue T, Koike H, Han GD, Suzuki K, Miyauchi N, Yuan H, et al. Nephrin and podocin dissociate at the onset of proteinuria in experimental membranous nephropathy. Kidney Int. 2005;67:2239–53.
Otaki Y, Miyauchi N, Higa M, Takada A, Kuroda T, Gejyo F, et al. The dissociation of NEPH1 from nephrin is involved in the development of rat model of focal segmental glomerulosclerosis. Am J Physiol Renal Physiol. 2008. in press.
Matsusaka T, Xin J, Niwa S, Kobayashi K, Akatsuka A, Hashizume H, et al. Genetic engineering of glomerular sclerosis in the mouse via control of onset and severity of podocyte-specific injury. J Am Soc Nephrol. 2005;16:1013–23.
Sawai K, Mori K, Mukoyama M, Sugawara A, Suganami T, Koshikawa M, et al. Angiogenic protein Cyr61 is expressed by podocytes in anti-Thy-1 glomerulonephritis. J Am Soc Nephrol. 2003;14:1154–63.
Morioka Y, Koike H, Ikezumi Y, Ito Y, Oyanagi A, et al. Podocyte injuries exacerbate mesangial proliferative glomerulonephritis. Kidney Int. 2001;60:2192–204.
Han GD, Koike H, Nakatsue T, Suzuki K, Yoneyama H, Narumi S, et al. IFN-inducible protein-10 has a differential role in podocyte during Thy 1.1 glomerulonephritis. J Am Soc Nephrol. 2003;14:3111–26.
Hara M, Yanagihara T, Takada T, Itoh M, Matsuno M, Yamamoto T, et al. Urinary excretion of podocytes reflects disease activity in children with glomerulonephritis. Nephron. 1998;18:35–41.
Maschio G, Alberti D, Janin G, Locatelli F, Mann JF, Motolese M, et al. Effect of the angiotensin-converting-enzyme inhibitor benazepril on the progression of chronic renal insufficiency. N Eng J Med. 1996;334:939–45.
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, et al. RENAAL study investigators:effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861–9.
Benigni A, Tomasoni S, Gagliardini E, Zoja C, Grunkemeyer JA, Kalluri R, et al. Blocking angiotensin II synthesis/activity preserves glomerular nephrin in rats with severe nephrosis. J Am Soc Nephrol. 2001;12:941–8.
Bonnet F, Cooper ME, Kawachi H, Allen TJ, Boner G, Cao Z. Irbesartan normalises the deficiency in glomerular nephrin expression in a model of diabetes and hypertension. Diabetologia. 2001;44:874–7.
Kawachi H, Kurihara H, Topham PS, Brown D, Shia MA, Orikasa M, et al. Slit diaphragm-reactive nephritogenic MAb 5-1-6 alters expression of ZO-1 in rat podocytes. Am J Physiol. 1997;273:F984–93.
Suzuki K, Han GD, Miyauchi N, Hashimoto T, Nakatsue T, Fujioka Y, et al. Angiotensin II type 1 and type 2 receptors play opposite roles in regulating the barrier function of kidney glomerular capillary wall. Am J Pathol. 2007;170:1841–53.
Miyauchi N, Saito A, Karasawa T, Harita Y, Suzuki K, Koike H, et al. Synaptic vesicle protein 2B is expressed in podocyte, and its expression is altered in proteinuric glomeruli. J Am Soc Nephrol. 2006;17:2748–59.
Hashimoto T, Karasawa T, Saito A, Miyauchi N, Han GD, Hayasaka K, et al. Ephrin-B1 localizes at the slit diaphragm of the glomerular podocyte. Kidney Int. 2007;72:954–64.
Janz R, Goda Y, Geppert M, Missler M, Sudhof TC. SV2A and SV2B function as redundant Ca2+ regulators in neurotransmitter release. Neuron. 1999;24:1003–16.
Heese K, Nagai Y, Sawada T. Identification of a new synaptic vesicle protein 2B mRNA transcript which is up-regulated in neurons by amyloid peptide fragment (1–42). Biochem Biophys Res Commun. 2001;289:924–8.
Lazzell DR, Belizaire R, Thakur P, Sherry DM, Janz R. SV2B regulates synaptotagmin 1 by direct interaction. J Biol Chem. 2004;279:52124–31.
Wang MM, Janz R, Belizaire R, Frishman LJ, Sherry DM. Differential distribution and developmental expression of synaptic vesicle protein 2 isoforms in the mouse retina. J Comp Neurol. 2003;460:106–22.
Clegg N, Ferguson C, True LD, Arnold H, Moorman A, Quinn JE, et al. Molecular characterization of prostatic small-cell neuroendocrine carcinoma. Prostate. 2003;55:55–64.
Hayashi M, Yamamoto A, Yatsushiro S, Yamada H, Futai M, Yamaguchi A, et al. Synaptic vesicle protein SV2B, but not SV2A, is predominantly expressed and associated with microvesicles in rat pinealocytes. J Neurochem. 1998;71:356–65.
Rastaldi MP, Armelloni S, Berra S, Li M, Pesaresi M, Poczewski H, et al. Glomerular podocytes possess the synaptic vesicle molecule Rab3A and its specific effector rabphilin-3a. Am J Pathol. 2003;163:889–99.
Eph Nomenclature Committee. Unified nomenclature for Eph family receptors and their ligands, the ephrins. Cell. 1997;90:403–4.
Kullander K, Klein R. Mechanisms and functions of Eph and ephrin signaling. Nat Rev Mol Cell Biol. 2002;3:475–86.
Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462–75.
Martinez A, Soriano E. Functions of ephrin/Eph interactions in the development of the nervous system: emphasis on the hippocampal system. Brain Res Rev. 2005;49:211–26.
Poliakov A, Cotrina M, Wilkinson DG. Diverse roles of eph receptors and ephrins in the regulation of cell migration and tissue assembly. Dev Cell. 2002;7:465–80.
Cheng N, Brantley DM, Chen J. The ephrins and Eph receptors in angiogenesis. Cytokine Growth Factor Rev. 2002;13:75–85.
Eph Nomenclature Committee. Unified nomenclature for Eph family receptors and their ligands, the ephrins. Cell. 1997;90:403–4.
Kullander K, Klein R. Mechanisms and functions of Eph and ephrin signaling. Nat Rev Mol Cell Biol. 2002;3:475–86.
Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462–75.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented at the 36th Eastern Regional Meeting of the Japanese Society of Nephrology.
About this article
Cite this article
Kawachi, H., Suzuki, K., Miyauchi, N. et al. Slit diaphragm dysfunction in proteinuric states: identification of novel therapeutic targets for nephrotic syndrome. Clin Exp Nephrol 13, 275–280 (2009). https://doi.org/10.1007/s10157-009-0162-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-009-0162-x