Abstract
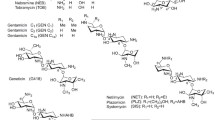
The critical resistance mechanisms of aminoglycoside antibiotics in bacteria of clinical importance are the enzymatic N-acetylation, O-phosphorylation, and O-nucleotidylation that generally result in the inactivation of aminoglycosides. To overcome such resistance mechanisms, dibekacin (3′,4′-dideoxykanamycin B) was developed as the first rationally designed semisynthetic aminoglycoside, based on the enzymatic 3′-O-phosphorylation of kanamycin. Subsequently, amikacin, netilmicin, and isepamicin were developed by introducing (S)-4-amino-2-hydroxybutyryl (AHB), ethyl, and (S)-3-amino-2-hydroxypropionyl side chains into the 1-amino group of kanamycin, sisomicin, and gentamicin B, respectively. These side chains are believed to block the access of a variety of aminoglycoside-modifying enzymes to their target sites. The latest semisynthetic aminoglycoside of clinical use in Japan is arbekacin (1-N-AHB-dibekacin), which has been extensively used since its approval as an anti-methicillin-resistant Staphylococcus aureus (MRSA) agent in 1990. Although it has several possible modification sites for aminoglycoside acetyltransferases (AACs), arbekacin-resistant MRSA strains that have emerged in the past 8 years have been those with a low or moderate level of resistance, due to a bifunctional enzyme, AAC(6′)/APH(2″), at low incidence. To overcome AAC(6′)/APH(2″)-dependent arbekacin-resistant MRSA strains, 2″-amino-2″-deoxyarbekacin and its 5-epiamino derivative have been already synthesized. However, simulative modification studies using AACs from aminoglycoside-producing Streptomyces strains have revealed that AAC(3) and AAC(2′) converted arbekacin to 3″-N-acetyl and 2′-N-acetyl derivatives, respectively, which retain high antibiotic activity. By contrast, the same acetylations of amikacin (3″-N-) and dibekacin (3-N-) resulted in their inactivation. Thus, these new findings confirmed the steric hindrance effect of the 1-N-acyl side chain and illuminated the novel aspect of arbekacin distinct from the other semisynthetic aminoglycosides, indicating that MRSA strains cannot be arbekacin-resistant even if they have acquired the aac(3) or aac(2′) gene.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: November 26, 1998 / Accepted: December 9, 1998
About this article
Cite this article
Kondo, S., Hotta, K. Semisynthetic aminoglycoside antibiotics: Development and enzymatic modifications. J Infect Chemother 5, 1–9 (1999). https://doi.org/10.1007/s101560050001
Issue Date:
DOI: https://doi.org/10.1007/s101560050001