Abstract
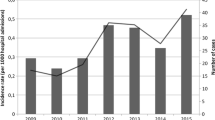
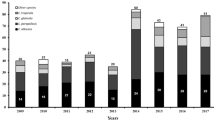
Invasive fungal infections have increased significantly in the past few decades because of the increase in high-risk populations. To investigate the distribution and drug susceptibilities of such infections, we analyzed all 152 Candida isolates causing candidemia from 2004 to 2006 at the China Medical University Hospital, a medical center in central Taiwan. Candida albicans was the most common species, accounting for 52.6 % of the isolates, followed by C. tropicalis (19.7 %), C. parapsilosis (14.5 %), C. glabrata (8.6 %), C. guilliermondii (3.9 %), and C. pelliculosa (0.7 %). All isolates were susceptible to amphotericin B, anidulafungin, micafungin, and voriconazole according to minimum inhibitory concentrations (MICs) after a 24-h incubation; 0.7 %, 6.6 %, and 7.9 % of isolates were resistant to amphotericin B, fluconazole, and voriconazole, respectively, after 48-h incubation. Both C. albicans and C. parapsilosis had high degrees of agreement for azoles between 24- and 48-h incubation periods, whereas C. glabrata (38.5–46.2 %) and C. tropicalis (56.7–63.3 %) did not. The majority of the isolates with high azole MICs displayed a trailing growth phenotype. Hence, the MICs of different drugs after 24-h incubation may be considered for prognosis of candidemia.
Similar content being viewed by others
References
Pappas PG, Kauffman CA, Andes D, Benjamin DK Jr, Calandra TF, Edwards JE Jr, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;48:503–35.
Pfaller MA, Diekema DJ. Epidemiology of invasive mycoses in North America. Crit Rev Microbiol. 2010;36:1–53.
Yang YL, Lo HJ. Mechanisms of antifungal agent resistance. J Microbiol Immunol Infect. 2001;34:79–86.
Chen PL, Lo HJ, Wu CJ, Lee HC, Chang CM, Lee NY, et al. Species distribution and antifungal susceptibility of blood Candida isolates at a tertiary hospital in southern Taiwan, 1999–2006. Mycoses. 2011;54:e17–23.
Chen S, Slavin M, Nguyen Q, Marriott D, Playford EG, Ellis D, et al. Active surveillance for candidemia, Australia. Emerg Infect Dis. 2006;12:1508–16.
Cheng MF, Yu KW, Tang RB, Fan YH, Yang YL, Hsieh KS, et al. Distribution and antifungal susceptibility of Candida species causing candidemia from 1996 to 1999. Diagn Microbiol Infect Dis. 2004;48:33–7.
Pfaller MA, Diekema DJ. Epidemiology of invasive candidiasis: a persistent public health problem. Clin Microbiol Rev. 2007;20:133–63.
Warnock DW. Trends in the epidemiology of invasive fungal infections. Nippon Ishinkin Gakkai Zasshi. 2007;48:1–12.
Yang YL, Cheng MF, Wang CW, Wang AH, Cheng WT, Lo HJ, et al. The distribution of species and susceptibility of amphotericin B and fluconazole of yeast pathogens isolated from sterile sites in Taiwan. Med Mycol. 2010;48:328–34.
Chuang YC, Chen YC, Chang SC, Sun CC, Chang YY, Chen ML, et al. Secular trends of healthcare-associated infections at a teaching hospital in Taiwan, 1981–2007. J Hosp Infect. 2010;76:143–9.
Colombo AL, Nucci M, Park BJ, Nouer SA, Arthington-Skaggs B, da Matta DA, et al. Epidemiology of candidemia in Brazil: a nationwide sentinel surveillance of candidemia in eleven medical centers. J Clin Microbiol. 2006;44:2816–23.
Edwards EJJ. Candida species. In: Mandell GL, Bennett JE, Dolin R, editors. Principles and practice of infectious diseases. New York: Churchill Livingstone; 1995. p. 2289–306.
Ruan SY, Hsueh PR. Invasive candidiasis: an overview from Taiwan. J Formos Med Assoc. 2009;108:443–51.
Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis. 2004;39:309–17.
Almirante B, Rodriguez D, Park BJ, Cuenca-Estrella M, Planes AM, Almela M, et al. Epidemiology and predictors of mortality in cases of Candida bloodstream infection: results from population-based surveillance, Barcelona, Spain, from 2002 to 2003. J Clin Microbiol. 2005;43:1829–35.
Marchetti O, Bille J, Fluckiger U, Eggimann P, Ruef C, Garbino J, et al. Epidemiology of candidemia in Swiss tertiary care hospitals: secular trends, 1991–2000. Clin Infect Dis. 2004;38:311–20.
Hajjeh RA, Sofair AN, Harrison LH, Lyon GM, Arthington-Skaggs BA, Mirza SA, et al. Incidence of bloodstream infections due to Candida species and in vitro susceptibilities of isolates collected from 1998 to 2000 in a population-based active surveillance program. J Clin Microbiol. 2004;42:1519–27.
Chen LY, Liao SY, Kuo SC, Chen SJ, Chen YY, Wang FD, et al. Changes in the incidence of candidaemia during 2000–2008 in a tertiary medical centre in northern Taiwan. J Hosp Infect. 2011;78:50–3.
Liu CY, Liao CH, Chen YC, Chang SC. Changing epidemiology of nosocomial bloodstream infections in 11 teaching hospitals in Taiwan between 1993 and 2006. J Microbiol Immunol Infect. 2010;43:416–29.
Wu CJ, Lee HC, Lee NY, Shih HI, Ko NY, Wang LR, et al. Predominance of gram-negative bacilli and increasing antimicrobial resistance in nosocomial bloodstream infections at a university hospital in southern Taiwan, 1996–2003. J Microbiol Immunol Infect. 2006;39:135–43.
Johnson EM. Rare and emerging Candida species. Curr Fungal Infect Rep. 2009;3:152–9.
Richardson MD. Opportunistic and pathogenic fungi. J Antimicrob Chemother. 1991;28(suppl A):1–11.
Sanglard D, Odds FC. Resistance of Candida species to antifungal agents: molecular mechanisms and clinical consequences. Lancet Infect Dis. 2002;2:73–85.
White TC, Marr KA, Bowden RA. Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev. 1998;11:382–402.
Yang YL, Cheng HH, Lo HJ. Distribution and antifungal susceptibility of Candida species isolated from different age populations in Taiwan. Med Mycol. 2006;44:237–42.
Yang YL, Hsieh LY, Wang AH, Lo HJ. Characterization of Candida species from different populations in Taiwan. Mycopathologia. 2011;172:131–9.
Lo HJ, Ho AH, Ho M. Factors accounting for mis-identification of Candida species. J Microbiol Immunol Infect. 2001;34:171–7.
Yang YL, Li SY, Cheng HH, Lo HJ. Susceptibilities to amphotericin B and fluconazole of Candida species in TSARY 2002. Diagn Microbiol Infect Dis. 2005;51:179–83.
Yang YL, Wang AH, Wang CW, Cheng WT, Li SY, Lo HJ, et al. Susceptibilities to amphotericin B and fluconazole of Candida species in TSARY 2006. Diagn Microbiol Infect Dis. 2008;61:175–80.
Leaw SN, Chang HC, Barton R, Bouchara JP, Chang TC. Identification of medically important Candida and non-Candida yeast species by an oligonucleotide array. J Clin Microbiol. 2007;45:2220–9.
Leaw SN, Chang HC, Sun HF, Barton R, Bouchara JP, Chang TC. Identification of medically important yeast species by sequence analysis of the internal transcribed spacer regions. J Clin Microbiol. 2006;44:693–9.
Yang YL, Ho YA, Cheng HH, Ho M, Lo HJ. Susceptibilities of Candida species to amphotericin B and fluconazole: the emergence of fluconazole resistance in Candida tropicalis. Infect Control Hosp Epidemiol. 2004;25:60–4.
Clinical Laboratory Standard Institute. Reference method for broth dilution antifungal susceptibility testing of yeasts; approved standard-third edition. CLSI document M27-A3. Wayne, PA: Clinical Laboratory Standard Institute; 2008.
Pfaller MA, Diekema DJ. Progress in antifungal susceptibility testing of Candida spp. by use of Clinical and Laboratory Standards Institute broth microdilution methods, 2010 to 2012. J Clin Microbiol. 2012;50:2846–56.
Pfaller MA, Diekema DJ, Andes D, Arendrup MC, Brown SD, Lockhart SR, et al. Clinical breakpoints for the echinocandins and Candida revisited: integration of molecular, clinical, and microbiological data to arrive at species-specific interpretive criteria. Drug Resist Update. 2011;14:164–76.
Peman J, Canton E, Linares-Sicilia MJ, Rosello EM, Borrell N, Ruiz-Perez-de-Pipaon MT, et al. Epidemiology and antifungal susceptibility of bloodstream fungal isolates in pediatric patients: a Spanish multicenter prospective survey. J Clin Microbiol. 2011;49:4158–63.
Arthington-Skaggs BA, Lee-Yang W, Ciblak MA, Frade JP, Brandt ME, Hajjeh RA, et al. Comparison of visual and spectrophotometric methods of broth microdilution MIC end point determination and evaluation of a sterol quantitation method for in vitro susceptibility testing of fluconazole and itraconazole against trailing and nontrailing Candida isolates. Antimicrob Agents Chemother. 2002;46:2477–81.
Yang YL, Chen HT, Lin CC, Chu WL, Lo HJ, TSARY Hospitals. Species distribution and drug susceptibilities of Candida isolates in TSARY 2010. Diagn Microbiol Infect Dis. 2013;76:182–6.
Moretti ML, Trabasso P, Lyra L, Fagnani R, Resende MR, de Oliveira Cardoso LG, et al. Is the incidence of candidemia caused by Candida glabrata increasing in Brazil? Five-year surveillance of Candida bloodstream infection in a university reference hospital in southeast Brazil. Med Mycol. 2013;51:225–30.
Pfaller M, Neofytos D, Diekema D, Azie N, Meier-Kriesche HU, Quan SP, et al. Epidemiology and outcomes of candidemia in 3648 patients: data from the Prospective Antifungal Therapy (PATH Alliance(R)) registry, 2004–2008. Diagn Microbiol Infect Dis. 2012;74:323–31.
Krcmery V, Barnes AJ. Non-albicans Candida spp. causing fungaemia: pathogenicity and antifungal resistance. J Hosp Infect. 2002;50:243–60.
Cheng MF, Yang YL, Yao TJ, Lin CY, Liu JS, Tang RB, et al. Risk factors for fatal candidemia caused by Candida albicans and non-albicans Candida species. BMC Infect Dis. 2005;5:22.
Pfaller MA, Andes D, Diekema DJ, Espinel-Ingroff A, Sheehan D. Wild-type MIC distributions, epidemiological cutoff values and species-specific clinical breakpoints for fluconazole and Candida: time for harmonization of CLSI and EUCAST broth microdilution methods. Drug Resist Update. 2010;13:180–95.
Pfaller MA, Andes D, Arendrup MC, Diekema DJ, Espinel-Ingroff A, Alexander BD, et al. Clinical breakpoints for voriconazole and Candida spp. revisited: review of microbiologic, molecular, pharmacodynamic, and clinical data as they pertain to the development of species-specific interpretive criteria. Diagn Microbiol Infect Dis. 2011;70:330–43.
Acknowledgments
We thank Bristol Myers Squibb for providing amphotericin B, Pfizer for providing anidulafungin, fluconazole, and voriconazole, and Astellas Pharms for micafungin. This work was supported in part by grants 00A1-ID-PP-04-014 and 01A1-IV-PP-04-014 from National Health Research Institutes.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
T.-P. Chang and M.-W. Ho contributed equally to this article.
About this article
Cite this article
Chang, TP., Ho, MW., Yang, YL. et al. Distribution and drug susceptibilities of Candida species causing candidemia from a medical center in central Taiwan. J Infect Chemother 19, 1065–1071 (2013). https://doi.org/10.1007/s10156-013-0623-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10156-013-0623-8