Abstract
Background
Histone deacetylase (HDAC) is strongly associated with epigenetic regulation and carcinogenesis, and its inhibitor can induce the differentiation or apoptosis of cancer cells.
Methods
We investigated the anticancer effects of the HDAC inhibitor valproic acid (VPA) in combination with gemcitabine (GEM), an antimetabolic, and pegylated interferon-α2b (PEG-IFN-α2b) in a human pancreatic cancer cell line using a cell proliferation assay. The gene expressions of HDAC1, MTA1, p21Waf1, and HIF-1 were evaluated by reverse transcription-PCR.
Results
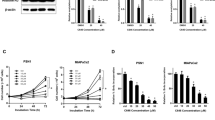
Valproic acid at 0.5 mM when used alone did not suppress cell proliferation. PEG–IFN-α2b at 102 E/ml weakly suppressed cell proliferation in both the BxPC3 (by 28%) and SUIT-2 (by 17%) human pancreatic cancer cell lines. GEM at 5 nM when used alone suppressed cell proliferation by 36 and 61% in the BxPC3 and SUIT-2 cell lines, respectively. The combination treatment of GEM + PEG–IFN-α2b strongly suppressed cell proliferation in the SUIT-2 (82%) and BxPC3 (51%) cell lines, which was further reinforced by the addition of VPA up to 88 and 67%, respectively. The combination treatment of GEM + PEG–IFN-α2b enhanced the expression of p21Waf1, which was also reinforced by VPA.
Conclusion
VPA augmented the inhibitory effects of PEG–IFN-α2b alone or in combination with PEG–IFN-α2b and GEM on cell proliferation. Such inhibitory effects may be due to the up-regulation of p21Waf1 expression.




Similar content being viewed by others
References
Hidalgo M (2010) Pancreatic cancer. N Engl J Med 362:1605–1617
Burris HA III, Moore MJ, Andersen J et al (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15:2403–2413
Haberland M, Montgomery RL, Olson EN (2009) The many roles of histone deacetylases in development and physiology: implications for disease and therapy. Nat Rev Genet 10:32–42
Minucci S, Pelicci PG (2006) Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer 6:38–51
Glozak MA, Seto E (2007) Histone deacetylases and cancer. Oncogene 26:5420–5432
Patra SK, Patra A, Dahiya R (2001) Histone deacetylase and DNA methyltransferase in human prostate cancer. Biochem Biophys Res Commun 287:705–713
Zhu P, Martin E, Mengwasser J et al (2004) Induction of HDAC2 expression upon loss of APC in colorectal tumorigenesis. Cancer Cell 5:455–463
Lin RJ, Nagy L, Inoue S et al (1998) Role of the histone deacetylase complex in acute promyelocytic leukaemia. Nature 391:811–814
Miyake K, Yoshizumi T, Imura S et al (2008) Expression of hypoxia-inducible factor-1alpha, histone deacetylase 1, and metastasis-associated protein 1 in pancreatic carcinoma: correlation with poor prognosis with possible regulation. Pancreas 36:e1–e9
Bolden JE, Peart MJ, Johnstone RW (2005) Anticancer activities of histone deacetylase inhibitors. Nat Rev Drug Discov 5:769–784
Warrell RP Jr, He LZ, Richon V et al (1998) Therapeutic targeting of transcription in acute promyelocytic leukemia by use of an inhibitor of histone deacetylase. J Natl Cancer Inst 90:1621–1625
Göttlicher M, Minucci S, Zhu P et al (2001) Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. EMBO J 20:6969–6978
Xia Q, Sung J, Chowdhury W et al (2006) Chronic administration of valproic acid inhibits prostate cancer cell growth in vitro and in vivo. Cancer Res 66:7237–7244
Catalano MG, Fortunati N, Pugliese M et al (2005) Valproic acid induces apoptosis and cell cycle arrest in poorly differentiated thyroid cancer cells. J Clin Endocrinol Metab 90:1383–1389
Baron S, Dianzani F (1994) The interferons: a biological system with therapeutic potential in viral infections. Antiviral Res 24:97–110
Hertzog PJ, Hwang SY, Kola I (1994) Role of interferons in the regulation of cell proliferation, differentiation and development. Mol Reprod Dev 39:226–232
Gutterman JU (1994) Cytokine therapeutics: lessons from interferon-α. Proc Natl Acad Sci USA 91:1198–1205
Krown SE (1988) Interferons in malignancy: biological products or biological response modifiers? J Natl Cancer Inst (Bethesda) 80:306–309
Miyake K, Tsuchida K, Sugino H et al (2007) Combination therapy of human pancreatic cancer implanted in nude mice by oral fluoropyrimidine anticancer agent (S-1) with interferon-alpha. Cancer Chemother Pharmacol 59:113–126
Baker DE (2003) Pegylated interferon plus ribavirin for the treatment of chronic hepatitis C. Rev Gastroenterol Disord 3:93–109
Yamada T, Furukawa K, Yokoi K et al (2010) Effects of irinotecan and 5-FU combination therapy in gastric cancer—is combination therapy synergic (in Japanese)? Gan To Kagaku Ryoho 37:2125–2129
Yoo YG, Kong G, Lee MO (2006) Metastasis-associated protein 1 enhances stability of hypoxia-inducible factor-1alpha protein by recruiting histone deacetylase 1. EMBO J 25:1231–1241
Kelly WK, O’Connor OA, Krug LM et al (2005) Phase I study of an oral histone deacetylase inhibitor, suberoylanilide hydroxamic acid, in patients with advanced cancer. J Clin Oncol 23:3923–3931
Fuino L, Bali P, Wittmann S et al (2003) Histone deacetylase inhibitor LAQ824 down-regulates Her-2 and sensitizes human breast cancer cells to trastuzumab, taxotere, gemcitabine, and epothilone B. Mol Cancer Ther 2:971–984
Rundall BK, Denlinger CE, Jones DR (2005) Suberoylanilide hydroxamic acid combined with gemcitabine enhances apoptosis in non-small cell lung cancer. Surgery 138:360–367
El-Deiry WS, Tokino T, Velculescu VE et al (1993) WAF1, a potential mediator of p53 tumor suppression. Cell 75:817–825
El-Deiry WS, Harper JW, O’Connor PM et al (1994) WAF1/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res 54:1169–1174
Deng C, Zhang P, Harper JW et al (1995) Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 82:675–684
Schwaller J, Koeffler HP, Niklaus G et al (1995) Posttranscriptional stabilization underlies p53-independent induction of p21WAF1/CIP1/SDI1 in differentiating human leukemic cells. J Clin Invest 95:973–979
DiGiuseppe JA, Redston MS, Yeo CJ et al (1995) p53-independent expression of the cyclin-dependent kinase inhibitor p21 in pancreatic carcinoma. Am J Pathol 147:884–888
Huang L, Sowa Y, Sakai T et al (2000) Activation of the p21WAF1/CIP1 promoter independent of p53 by the histone deacetylase inhibitor suberoylanilide hydroxamic acid (SAHA) through the Sp1 sites. Oncogene 19:5712–5719
Das CM, Aguilera D, Vasquez H et al (2007) Valproic acid induces p21 and topoisomerase-II (alpha/beta) expression and synergistically enhances etoposide cytotoxicity in human glioblastoma cell lines. J Neurooncol 85:159–170
Donadelli M, Costanzo C, Faggioli L et al (2003) Trichostatin A, an inhibitor of histone deacetylases, strongly suppresses growth of pancreatic adenocarcinoma cells. Mol Carcinog 38:59–69
Richon VM, Sandhoff TW, Rifkind RA et al (2000) Histone deacetylase inhibitor selectively induces p21WAF1 expression and gene-associated histone acetylation. Proc Natl Acad Sci USA 97:10014–10019
Nichole B, Nohea A, Jason G et al (2007) The histone deacetylase inhibitor suberoylanilide hydroxamic acid induces growth inhibition and enhances gemcitabine-induced cell death in pancreatic cancer. Clin Cancer Res 13:18–26
Tagliaferri P, Caraglia M, Budillon A et al (2005) New pharmacokinetic and pharmacodynamic tools for interferon-alpha (IFN-alpha) treatment of human cancer. Cancer Immunol Immunother 54:1–10
Toh Y, Pencil SD, Nicolson GL (1994) A novel candidate metastasis-associated gene, mta1, differentially expressed in highly metastatic mammary adenocarcinoma cell lines. cDNA cloning, expression, and protein analyses. J Biol Chem 269:22958–22963
Kumar R (2003) Another tie that binds the MTA family to breast cancer. Cell 113:142–143
Bowen NJ, Fujita N, Kajita M et al (2004) Mi-2/NuRD: multiple complexes for many purposes. Biochim Biophys Acta 1677:52–57
Picozzi VJ, Kozarek RA, Traverso LW (2003) Interferon based adjuvant chemoradiation therapy after pancreaticoduodenectomy for pancreatic adenocarcinoma. Am J Surg 185:4767–4780
Solorzano CC, Hwang R, Baker CH et al (2003) Administration of optimal biological dose and schedule of interferon alpha combined with gemcitabine induces apoptosis in tumor-associated endothelial cells and reduces growth of human pancreatic carcinoma implanted orthotopically in nude mice. Clin Cancer Res 9:1858–1867
Perez-Zincer F, Olencki T, Budd GT et al (2002) A phase I trial of weekly gemcitabine and subcutaneous interferon alpha in patients with refractory renal cell carcinoma. Invest New Drugs 20:305–310
Fuxius S, Mross K, Mansouri K et al (2002) Gemcitabine and interferon-alpha 2b in solid tumors: a phase I study in patients with advanced or metastatic non-small cell lung, ovarian, pancreatic or renal cancer. Anticancer Drugs 13:899–905
Amato RJ, Khan M (2008) A phase I clinical trial of low-dose interferon-alpha-2A, thalidomide plus gemcitabine and capecitabine for patients with progressive metastatic renal cell carcinoma. Cancer Chemother Pharmacol 61:1069–1073
Krepler C, Certa U, Wacheck V et al (2004) Pegylated and conventional interferon-alpha induce comparable transcriptional responses and inhibition of tumor growth in a human melanoma SCID mouse xenotransplantation model. J Invest Dermatol 123:664–669
Blaheta RA, Michaelis M, Driever PH et al (2005) Evolving anticancer drug valproic acid: insights into the mechanism and clinical studies. Med Res Rev 25:383–397
Faiella A, Wernig M, Consalez GG et al (2000) A mouse model for valproate teratogenicity: parental effects, homeotic transformations, and altered HOX expression. Hum Mol Genet 9:227–236
Acknowledgments
Grant support was provided by the Research Support Foundation of The University of Tokushima and TAIHO Pharmaceutical Co., Ltd., and the Grants-in-Aid for Scientific Researches of the Japan Society for the Promotion of Science (Grant-in-Aid for Scientific Research B: No. 20390359). We would like to thank Ms. Harada for providing technical assistance.
Conflict of interest
M. Shimada has received research funding from Merck Sharp and Dohme Corp. The other authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iwahashi, S., Shimada, M., Utsunomiya, T. et al. Histone deacetylase inhibitor augments anti-tumor effect of gemcitabine and pegylated interferon-α on pancreatic cancer cells. Int J Clin Oncol 16, 671–678 (2011). https://doi.org/10.1007/s10147-011-0246-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-011-0246-y