Abstract
Background
We aimed to evaluate the feasibility and efficacy of hypofractionated involved-field radiation therapy (IFRT) omitting elective nodal irradiation (ENI) with concurrent chemotherapy for locally advanced non-small-cell lung cancer (NSCLC).
Methods
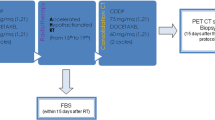
Between July 2004 and July 2006, ten patients with locally advanced NSCLC were included in this study. One had stage IIIA and 9 had stage IIIB disease. The treatment consisted of IFRT in fractions of 2.5 Gy and weekly carboplatin (CBDCA)/paclitaxel (PTX). Hypofractionated IFRT with a median total dose of 65 Gy with median percent total lung volume exceeding 20 Gy (V20) of 20.2%, and a median of five courses of chemotherapy with weekly CBDCA (area under the curve, 1.5−2.0)/PTX (30−35 mg/m2) were given to all patients.
Results
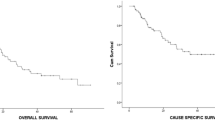
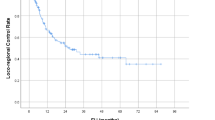
The median survival time and the 1-, 2-, and 3-year overall survival rates were 29.5 months and 90.0%, 58.3%, and 43.8%, respectively. No elective nodal failure was encountered during the median follow up of 18.2 months. No acute or late toxicities of grade 3 or worse were observed. No in-field recurrence occurred in the group with a total dose of 67.5 Gy or more, but there was such recurrence in 83.3% of those in the group with less than 67.5 Gy.
Conclusion
Hypofractionated IFRT with weekly CBDCA/PTX was a feasible treatment regimen. Hypofractionated IFRT with a total dose of 67.5 Gy or more could be a promising modality to improve the treatment results in patients with locally advanced NSCLC.
Similar content being viewed by others
References
Pfister DG, Johson DH, Azzoli CG, et al. (2004) American Society of Clinical Oncology treatment of unresectable non-small-cell lung cancer guideline: update 2003. J Clin Oncol 22:330–353
Curran WJ, Scott CB, Langer CJ, et al. (2003) Long-term benefit is observed in a phase III comparison of sequential vs concurrent chemoradiation for patients with unresected stage III NSCLC: RTOG 9410. Proc Am Soc Clin Oncol 22:621 (abstract 2499)
Zatloukal P, Petruzelka L, Zemanova M, et al. (2004) Concurrent versus sequential chemoradiotherapy with cisplatin and vinorelbine in locally advanced non-small-cell lung cancer. A randomized study. Lung Cancer 46:87–98
Belani CP, Choy H, Bonomi P, et al. (2005) Combined chemoradiotherapy regimens of paclitaxel and carboplatin for locally advanced non-small-cell lung cancer: a randomized phase II locally advanced multi-modality protocol. J Clin Oncol 23:5883–5891
Pierre F, Gilles R, Pascal T, et al. (2005) Randomized phase III trial of sequential chemoradiotherapy compared with concurrent chemoradiotherapy in locally advanced non-small-cell lung cancer: Groupe Lyon-Saint-Etienne d’Oncologie Thoracique-Groupe Français de Pneumo-Cancérologie NPC 95-01 Study. J Clin Oncol 25:5910–5917
Saunders M, Dische S, Barrett A, et al. (1999) Continuous hyperfractionated accelerated radiotherapy (CHART) versus conventional radiotherapy in non-small cell lung cancer: mature data from a randomised multicentre trial. Radiother Oncol 52:137–148
Schaake-Koning C, van den Bogaert W, Dalesio O, et al. (1992) Effects of concomitant cisplatin and radiotherapy on inoperable non-small-cell lung cancer. N Engl J Med 326:524–530
Senan S, Burgers S, Samson MJ, et al. (2002) Can elective nodal irradiation be omitted in stage III non-small-cell lung cancer? Analysis of recurrences in a phase II study of induction chemotherapy and involved-field radiotherapy. Int J Radiat Oncol Biol Phys 54:999–1006
Rosenzweig KE, Sim SE, Mychalczak B, et al. (2001) Elective nodal irradiation in the treatment of non-small-cell lung cancer with three-dimensional conformal radiation therapy. Int J Radiat Oncol Biol Phys 50:681–685
Kimura T, Hirokawa Y, Murakami Y, et al. (2004) The preliminary results of accelerated hyperfractionated radiotherapy with involved-field omitted elective nodal irradiation (ENI) for inoperable advanced non-small cell lung cancer (in Japanese with English abstract). J Jpn Soc Ther Radiol Oncol 16:79–84
Graham MV, Purdy JA, Emami B, et al. (1999) Clinical dose-volume histogram analysis for pneumonitis after 3D treatment for non-small cell lung cancer (NSCLC). Int J Radiat Oncol Biol Phys 45:323–329
Tsujino K, Hirota S, Endo M, et al. (2003) Predictive value of dose-volume histogram parameters for predicting radiation pneumonitis after concurrent chemoradiation for lung cancer. Int J Radiat Oncol Biol Phys 55:110–115
Bradley JD, Graham MV, Winter KW, et al. (2003) Acute and late toxicity results of RTOG 9311: a dose escalation study using 3D conformal radiation therapy in patients with inoperable non-small cell lung cancer. Int J Radiat Oncol Biol Phys 57:137–138 (abstract 23)
Ohashi N, Arita K, Daga H, et al. (2003) Sequential phase I studies of paclitaxel +/− carboplatin and hyperfractionated radiation therapy (HFX RT) for advanced non-small cell lung cancer (NSCLC). Proc Am Soc Clin Oncol 22:680 (abstract 2736).
Uematsu M, Shioda A, Suda A, et al. (2001) Computed tomography-guided frameless stereotactic radiotherapy for stage I nonsmall cell lung cancer: a 5-year experience. Int J Radiat Oncol Biol Phys 51:666–670
Onishi H, Araki T, Shirato H, et al. (2004) Stereotactic hypofractionated high-dose irradiation for stage I nonsmall cell lung carcinoma: clinical outcomes in 245 subjects in a Japanese multiinstitutional study. Cancer 101:1623–1631
Nagata Y, Takayama K, Matsuo Y, et al. (2005) Clinical outcomes of a phase I/II study of 48 Gy of stereotactic body radiotherapy in four fractions for primary lung cancer using a stereotactic body frame. Int J Radiat Oncol Biol Phys 63:1427–1431
Chang JY, Bradley JD, Govindan R, et al. (2008) Thoracic tumors. In: Halperin EC, Perez CA, Brady LW (eds) Perez and Brady’s principles and practice of radiation oncology, 5th edn. Lippincott Williams & Wilkins, Philadelphia, PA, pp 1076–1108
Rosenzweig KE, Sura S, Jackson A, et al. (2007) Involved-field radiation therapy for inoperable non-small-cell lung cancer. J Clin Oncol 35:5557–5561
Yuan S, Sun X, Li M, et al. (2007) A randomized study of involved-field irradiation versus elective nodal irradiation in combination with concurrent chemotherapy for inoperable stage III nonsmall cell lung cancer. Am J Clin Oncol 30:239–244
Bradley JD, Graham MV, Winter K, et al. (2005) Toxicity and outcome results of RTOG 9311: a phase I-II dose-escalation study using three-dimensional conformal radiotherapy in patients with inoperable non-small-cell lung carcinoma. Int J Radiat Oncol Biol Phys 61:318–328
Bradley JD, Graham M, Suzanne S, et al. (2005) Phase I results of RTOG L-0117; a phase I/II dose intensification study using 3DCRT and concurrent chemotherapy for patients with Inoperable NSCLC. J Clin Oncol 23:636 (abstract 7063)
Schild SE, McGinnis WL, Graham D, et al. (2006) Results of a phase I trial of concurrent chemotherapy and escalating doses of radiation for unresectable non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 15:1106–1111
Socinski MA, Blackstock AW, Bogart JA, et al. (2008) Randomized phase II trial of induction chemotherapy followed by concurrent chemotherapy and dose-escalated thoracic conformal radiotherapy (74 Gy) in stage III non-small-cell lung cancer: CALGB 30105. J Clin Oncol 26:2457–2463
Blackstock AW, Govindan R (2007) Definitive chemoradiation for the treatment of locally advanced non-small-cell lung cancer. J Clin Oncol 25:4146–4152
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Matsuura, K., Kimura, T., Kashiwado, K. et al. Results of a preliminary study using hypofractionated involved-field radiation therapy and concurrent carboplatin/paclitaxel in the treatment of locally advanced non-small-cell lung cancer. Int J Clin Oncol 14, 408–415 (2009). https://doi.org/10.1007/s10147-009-0889-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-009-0889-0