Abstract
Background
Gene polymorphisms of the UDP-glucuronosyltransferase 1 family, polypeptide A1 (UGT1A1) contribute to individual variations in adverse events among patients administered irinotecan, and the distribution of the polymorphisms shows large interethnic differences. Variation in the solute carrier organic anion-transporter family, member 1B1 (SLCO1B1) gene also has a significant effect on the disposition of irinotecan in Asian cancer patients. In the present study, we evaluated the association of genetic polymorphisms of UGT1A1 and SLCO1B1 with irinotecanrelated neutropenia in Japanese cancer patients.
Methods
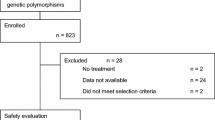
One hundred and thirty-five consecutive patients treated with irinotecan were enrolled. Genotypes of UGT1A1 (*60, *28, *6, and *27) and SLCO1B1 (*1b, *5, and haplotype *15) were determined by direct sequencing. Severe neutropenia refers to events observed during the first cycle of irinotecan treatment.
Results
Severe neutropenia was observed in 29 patients (22%). Six patients were homozygous and 48 heterozygous for UGT1A1*6. Only 1 patient was homozygous for UGT1A1*28. Homozygosity for UGT1A1*6 was associated with a high risk of severe neutropenia (odds ratio [OR], 7.78; 95% confidence interval [CI], 1.36 to 44.51). No significant association was found between severe neutropenia and other UGT1A1 polymorphisms or SLCO1B1 polymorphisms.
Conclusion
These findings suggest that the UGT1A1*6 polymorphism is a potential predictor of severe neutropenia caused by irinotecan in Japanese cancer patients.
Similar content being viewed by others
References
Meyerhardt JA, Mayer RJ (2005) Systemic therapy for colorectal cancer. N Engl J Med 352:476–487
Noda K, Nishiwaki Y, Kawahara M, et al. (2002) Irinotecan plus cisplatin compared with etoposide plus cisplatin for extensive small-cell lung cancer. N Engl J Med 346:85–91
Hahn KK, Wolff JJ, Kolesar JM (2006) Pharmacogenetics and irinotecan therapy. Am J Health Syst Pharm 63:2211–2217
Innocenti F, Undevia SD, Iyer L, et al. (2004) Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol 22:1382–1388
Ando Y, Saka H, Ando M, et al. (2000) Polymorphisms of UDPglucuronosyltransferase gene and irinotecan toxicity: a pharmacogenetic analysis. Cancer Res 60:6921–6926
Ramchandani RP, Wang Y, Booth BP, et al. (2007) The role of SN-38 exposure, UGT1A1*28 polymorphism, and baseline bilirubin level in predicting severe irinotecan toxicity. J Clin Pharmacol 47:78–86
Monaghan G, Ryan M, Seddon R, et al. (1996) Genetic variation in bilirubin UDP-glucuronosyltransferase gene promoter and Gilbert’s syndrome. Lancet 347:578–581
Ando Y, Chida M, Nakayama K, et al. (1998) The UGT1A1*28 allele is relatively rare in a Japanese population. Pharmacogenetics 8:357–360
Kaniwa N, Kurose K, Jinno H, et al. (2005) Racial variability in haplotype frequencies of UGT1A1 and glucuronidation activity of a novel single nucleotide polymorphism 686C>T (P229L) found in an African-American. Drug Metab Dispos 33:458–465
Sai K, Saeki M, Saito Y, et al. (2004) UGT1A1 haplotypes associated with reduced glucuronidation and increased serum bilirubin in irinotecan-administered Japanese patients with cancer. Clin Pharmacol Ther 75:501–515
Minami H, Sai K, Saeki M, et al. (2007) Irinotecan pharmacokinetics/ pharmacodynamics and UGT1A genetic polymorphisms in Japanese: roles of UGT1A1*6 and *28. Pharmacogenet Genomics 17:497–504
Araki K, Fujita K, Ando Y, et al. (2006) Pharmacogenetic impact of polymorphisms in the coding region of the UGT1A1 gene on SN-38 glucuronidation in Japanese patients with cancer. Cancer Sci 97:1255–1259
Han JY, Lim HS, Shin ES, et al. (2006) Comprehensive analysis of UGT1A polymorphisms predictive for pharmacokinetics and treatment outcome in patients with non-small-cell lung cancer treated with irinotecan and cisplatin. J Clin Oncol 24:2237–2244
Charasson V, Bellott R, Meynard D, et al. (2004) Pharmacogenetics of human carboxylesterase 2, an enzyme involved in the activation of irinotecan into SN-38. Clin Pharmacol Ther 76:528–535
de Jong FA, Scott-Horton TJ, Kroetz DL, et al. (2007) Irinotecaninduced diarrhea: functional significance of the polymorphic ABCC2 transporter protein. Clin Pharmacol Ther 81:42–49
Nozawa T, Minami H, Sugiura S, et al. (2005) Role of organic anion transporter OATP1B1 (OATP-C) in hepatic uptake of irinotecan and its active metabolite, 7-ethyl-10-hydroxycamptothecin: in vitro evidence and effect of single nucleotide polymorphisms. Drug Metab Dispos 33:434–439
Xiang X, Jada SR, Li HH, et al. (2006) Pharmacogenetics of SLCO1B1 gene and the impact of *1b and *15 haplotypes on irinotecan disposition in Asian cancer patients. Pharmacogenet Genomics 16:683–691
Akaba K, Kimura T, Sasaki A, et al. (1999) Neonatal hyperbilirubinemia and a common mutation of the bilirubin uridine diphosphate-glucuronosyltransferase gene in Japanese. J Hum Genet 44:22–25
Maruo Y, Nishizawa K, Sato H, et al. (1999) Association of neonatal hyperbilirubinemia with bilirubin UDP-glucuronosyltransferase polymorphism. Pediatrics 103:1224–1227
Bancroft JD, Kreamer B, Gourley GR (1998) Gilbert syndrome accelerates development of neonatal jaundice. J Pediatr 132:656–660
Ciotti M, Basu N, Brangi M, et al. (1999) Glucuronidation of 7-ethyl-10-hydroxycamptothecin (SN-38) by the human UDPglucuronosyltransferases encoded at the UGT1 locus. Biochem Biophys Res Commun 260:199–202
Gagne JF, Montminy V, Belanger P, et al. (2002) Common human UGT1A polymorphisms and the altered metabolism of irinotecan active metabolite 7-ethyl-10-hydroxycamptothecin (SN-38). Mol Pharmacol 62:608–617
Fujita K, Ando Y, Nagashima F, et al. (2007) Genetic linkage of UGT1A7 and UGT1A9 polymorphisms to UGT1A1*6 is associated with reduced activity for SN-38 in Japanese patients with cancer. Cancer Chemother Pharmacol 60:515–522
Hoskins, JM, Godberg, RM, Qu, P, et al. (2007) UGT1A1*28 genotype and irinotecan-induced neutropenia. J Natl Cancer Inst 99:1290–1295
Nishizato Y, Ieiri I, Suzuki H, et al. (2003) Polymorphisms of OATP-C (SLC21A6) and OAT3 (SLC22A8) genes: consequences for pravastatin pharmacokinetics. Clin Pharmacol Ther 73:554–565
Tokui T, Nakai D, Nakagomi R, et al. (1999) Pravastatin, an HMG-CoA reductase inhibitor, is transported by rat organic anion transporting polypeptide, oatp2. Pharm Res 16:904–908
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Onoue, M., Terada, T., Kobayashi, M. et al. UGT1A1*6 polymorphism is most predictive of severe neutropenia induced by irinotecan in Japanese cancer patients. Int J Clin Oncol 14, 136–142 (2009). https://doi.org/10.1007/s10147-008-0821-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-008-0821-z