Abstract
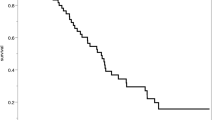
The need to pursue long-term follow-up in patients treated for a ruptured aneurysm remains debated. New aneurysms development is a crucial element to consider but remains scarcely analyzed especially after a mean follow-up longer than 10 years. Our study was designed to provide rates of newly developed aneurysms in patients who have undergone prior clipping who were not followed with serial imaging. Patients were included if they were (1) treated more than 10 years ago by clipping of a ruptured aneurysm, (2) independent at time of discharge, (3) presently younger than 65 years, and if (4) they agreed to undergo a late digital subtraction angiography (DSA) control or to transmit results of a recent one performed elsewhere. Twenty patients were included with a mean delay between aneurysm treatment and late DSA of 18.0 years (10–26.5 years). Out of these patients, six (30%) harbored new aneuryms. Of these six individuals, four (66.6%) presented multiple aneurysms with a total of 15 newly discovered aneurysms. Aneurysm sizes ranged from 1 to 10 mm. One patient suffered from a de novo aneurysm rupture. Multiple aneurysms at the time of the first hemorrhage were a risk factor in developing de novo aneurysm (p = 0.0175). In conclusion, based on a 30% rate of new aneurysm formation in patients clipped more than a decade ago, close screening on a very long-term perspective is encouraged. This study suggests aneurysm formation to be a continuous process.


Similar content being viewed by others
References
Akyuz M, Tuncer R, Yilmaz S, Sindel T (2004) Angiographic follow-up after surgical treatment of intracranial aneurysms. Acta Neurochir (Wien) 146(3):245–250, discussion 250
Brinjikji W, Lanzino G, Cloft HJ, Rabinstein A, Kallmes DF (2010) Endovascular treatment of very small (3 mm or smaller) intracranial aneurysms: report of a consecutive series and a meta-analysis. Stroke 41(1):116–121
Bromberg JE, Rinkel GJ, Algra A, Greebe P, van Duyn CM, Hasan D, Limburg M, ter Berg HW, Wijdicks EF, van Gijn J (1995) Subarachnoid haemorrhage in first and second degree relatives of patients with subarachnoid haemorrhage. BMJ 311(7000):288–289
Cloft HJ, Joseph GJ, Dion JE (1999) Risk of cerebral angiography in patients with subarachnoid hemorrhage, cerebral aneurysm, and arteriovenous malformation: a meta-analysis. Stroke 30(2):317–320
David CA, Vishteh AG, Spetzler RF, Lemole M, Lawton MT, Partovi S (1999) Late angiographic follow-up review of surgically treated aneurysms. J Neurosurg 91(3):396–401
Edner G, Almqvist H (2007) The Stockholm 20-year follow-up of aneurysmal subarachnoid hemorrhage outcome. Neurosurgery 60(6):1017–1023, discussion 1023–1014
(2000) Epidemiology of aneurysmal subarachnoid hemorrhage in Australia and New Zealand: incidence and case fatality from the Australasian Cooperative Research on Subarachnoid Hemorrhage Study (ACROSS). Stroke 31(8):1843–1850
Feuerberg I, Lindquist C, Lindqvist M, Steiner L (1987) Natural history of postoperative aneurysm rests. J Neurosurg 66(1):30–34
Juvela S (2001) Risk of subarachnoid hemorrhage from a de novo aneurysm. Stroke 32(8):1933–1934
Juvela S, Porras M, Heiskanen O (1993) Natural history of unruptured intracranial aneurysms: a long-term follow-up study. J Neurosurg 79(2):174–182
Juvela S, Poussa K, Porras M (2001) Factors affecting formation and growth of intracranial aneurysms: a long-term follow-up study. Stroke 32(2):485–491
Lubicz B, Levivier M, Francois O, Thoma P, Sadeghi N, Collignon L, Baleriaux D (2007) Sixty-four-row multisection CT angiography for detection and evaluation of ruptured intracranial aneurysms: interobserver and intertechnique reproducibility. AJNR Am J Neuroradiol 28(10):1949–1955
Matheus MG, Castillo M (2003) Development of de novo intracranial aneurysm in three months: case report and literature review. AJNR Am J Neuroradiol 24(4):709–710
Molyneux AJ, Kerr RS, Birks J, Ramzi N, Yarnold J, Sneade M, Rischmiller J (2009) Risk of recurrent subarachnoid haemorrhage, death, or dependence and standardised mortality ratios after clipping or coiling of an intracranial aneurysm in the International Subarachnoid Aneurysm Trial (ISAT): long-term follow-up. Lancet Neurol 8(5):427–433
Nakase H, Kamada Y, Aoki H, Goda K, Morimoto T, Sakaki T (2000) Clinical study on recurrent intracranial aneurysms. Cerebrovasc Dis 10(4):255–260
Obray R, Clatterbuck R, Olvi A, Tamargo R, Murphy KJ, Gailloud P (2003) De novo aneurysm formation 6 and 22 months after initial presentation in two patients. AJNR Am J Neuroradiol 24(9):1811–1813
Pechlivanis I, Koenen D, Engelhardt M, Scholz M, Koenig M, Heuser L, Harders A, Schmieder K (2008) Computed tomographic angiography in the evaluation of clip placement for intracranial aneurysm. Acta Neurochir (Wien) 150(7):669–676
Rinkel GJ (2005) Intracranial aneurysm screening: indications and advice for practice. Lancet Neurol 4(2):122–128
Rinkel GJ, Djibuti M, Algra A, van Gijn J (1998) Prevalence and risk of rupture of intracranial aneurysms: a systematic review. Stroke 29(1):251–256
Romijn M, Gratama van Andel HA, van Walderveen MA, Sprengers ME, van Rijn JC, van Rooij WJ, Venema HW, Grimbergen CA, den Heeten GJ, Majoie CB (2008) Diagnostic accuracy of CT angiography with matched mask bone elimination for detection of intracranial aneurysms: comparison with digital subtraction angiography and 3D rotational angiography. AJNR Am J Neuroradiol 29(1):134–139
Ronkainen A, Niskanen M, Rinne J, Koivisto T, Hernesniemi J, Vapalahti M (2001) Evidence for excess long-term mortality after treated subarachnoid hemorrhage. Stroke 32(12):2850–2853
Sagara Y, Kiyosue H, Hori Y, Sainoo M, Nagatomi H, Mori H (2005) Limitations of three-dimensional reconstructed computerized tomography angiography after clip placement for intracranial aneurysms. J Neurosurg 103(4):656–661
Sakaki T, Tominaga M, Miyamoto K, Tsunoda S, Hiasa Y (1993) Clinical studies of de novo aneurysms. Neurosurgery 32(4):512–516, discussion 516–517
Sprengers ME, van Rooij WJ, Sluzewski M, Rinkel GJ, Velthuis BK, de Kort GA, Majoie CB (2009) MR angiography follow-up 5 years after coiling: frequency of new aneurysms and enlargement of untreated aneurysms. AJNR Am J Neuroradiol 30(2):303–307
Sundt TM Jr, Kobayashi S, Fode NC, Whisnant JP (1982) Results and complications of surgical management of 809 intracranial aneurysms in 722 cases. Related and unrelated to grade of patient, type of aneurysm, and timing of surgery. J Neurosurg 56(6):753–765
Tonn J, Hoffmann O, Hofmann E, Schlake HP, Sorensen N, Roosen K (1999) “De novo” formation of intracranial aneurysms: who is at risk? Neuroradiology 41(9):674–679
Tsutsumi K, Ueki K, Kirino T (2001) Recurrent subarachnoid hemorrhage. J Neurosurg 94(3):541–542
Tsutsumi K, Ueki K, Morita A, Usui M, Kirino T (2001) Risk of aneurysm recurrence in patients with clipped cerebral aneurysms: results of long-term follow-up angiography. Stroke 32(5):1191–1194
Tsutsumi K, Ueki K, Usui M, Kwak S, Kirino T (1998) Risk of recurrent subarachnoid hemorrhage after complete obliteration of cerebral aneurysms. Stroke 29(12):2511–2513
Tsutsumi K, Ueki K, Usui M, Kwak S, Kirino T (1999) Risk of subarachnoid hemorrhage after surgical treatment of unruptured cerebral aneurysms. Stroke 30(6):1181–1184
van der Schaaf IC, Brilstra EH, Rinkel GJ, Bossuyt PM, van Gijn J (2002) Quality of life, anxiety, and depression in patients with an untreated intracranial aneurysm or arteriovenous malformation. Stroke 33(2):440–443
van der Schaaf IC, Velthuis BK, Wermer MJ, Frenkel NJ, Majoie CB, Witkamp TD, de Kort G, Freling NJ, Rinkel GJ (2006) Multislice computed tomography angiography screening for new aneurysms in patients with previously clip-treated intracranial aneurysms: feasibility, positive predictive value, and interobserver agreement. J Neurosurg 105(5):682–688
van der Schaaf IC, Velthuis BK, Wermer MJ, Majoie C, Witkamp T, de Kort G, Freling NJ, Rinkel GJ (2005) New detected aneurysms on follow-up screening in patients with previously clipped intracranial aneurysms: comparison with DSA or CTA at the time of sah. Stroke 36(8):1753–1758
van Rooij WJ, Sprengers ME, de Gast AN, Peluso JP, Sluzewski M (2008) 3D rotational angiography: the new gold standard in the detection of additional intracranial aneurysms. AJNR Am J Neuroradiol 29(5):976–979
Wermer MJ, Buskens E, van der Schaaf IC, Bossuyt PM, Rinkel GJ (2004) Yield of screening for new aneurysms after treatment for subarachnoid hemorrhage. Neurology 62(3):369–375
Wermer MJ, Greebe P, Algra A, Rinkel GJ (2005) Incidence of recurrent subarachnoid hemorrhage after clipping for ruptured intracranial aneurysms. Stroke 11:2394–2399
Wermer MJ, Rinkel GJ, Greebe P, Albrecht KW, Dirven CM, Tulleken CA (2005) Late recurrence of subarachnoid hemorrhage after treatment for ruptured aneurysms: patient characteristics and outcomes. Neurosurgery 2:197–204, discussion 197–204
Wermer MJ, van der Schaaf IC, Velthuis BK, Algra A, Buskens E, Rinkel GJ (2005) Follow-up screening after subarachnoid haemorrhage: frequency and determinants of new aneurysms and enlargement of existing aneurysms. Brain 128(Pt 10):2421–2429
Wiebers DO, Whisnant JP, Huston J 3rd, Meissner I, Brown RD Jr, Piepgras DG, Forbes GS, Thielen K, Nichols D, O’Fallon WM, Peacock J, Jaeger L, Kassell NF, Kongable-Beckman GL, Torner JC (2003) Unruptured intracranial aneurysms: natural history, clinical outcome, and risks of surgical and endovascular treatment. Lancet 362(9378):103–110
Yamakawa H, Sakai N, Takenaka K, Yoshimura S, Andoh T, Yamada H, Ohkuma A, Takada M, Funakoshi T (1997) Clinical analysis of recurrent subarachnoid hemorrhage after neck clipping surgery. Neurol Med Chir (Tokyo) 37(5):380–385, discussion 385–386
Yasargil MG Sr, Young PH, Teddy PJ (1984) Microneurosurgery: II. Clinical considerations, surgery of the intracranial aneurysms and results. Thieme, Stutgart
Yasuhara T, Tamiya T, Sugiu K, Inoue S, Ohmoto T (2002) De novo formation and rupture of an aneurysm. Case report. J Neurosurg 97(3):697–700
Yoneoka Y, Takeda N, Akira I, Ibuchi Y, Kumagai T, Sugai T, Takeda KI, Ueda K (2004) Ruptured de novo intracranial aneurysms. Acta Neurochir (Wien) 146(9):979–981, discussion 981
Zhang LJ, Wu SY, Niu JB, Zhang ZL, Wang HZ, Zhao YE, Chai X, Zhou CS, Lu GM (2010) Dual-energy CT angiography in the evaluation of intracranial aneurysms: image quality, radiation dose, and comparison with 3D rotational digital subtraction angiography. AJR Am J Roentgenol 194(1):23–30
Acknowledgments
We are grateful to Patrick Fransen, MD, for providing information regarding digital subtraction angiographies performed in his institution (Clinique du Parc Léopold, Brussels, Belgium); Christian Melot, MD, PhD, MSciBiostat (Department of Emergency Medicine, Erasme Hospital, Brussels, Belgium) for his assistance in statistical analysis; and Jeroen Coppens, MD (Department of Neurosurgery, Saint Louis, MO) for his help in grammatical corrections.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Elke R. Gizewski, Giessen, Germany
For a long time, patients with cerebral artery aneurysms have been treated using surgical clipping. The method is reported to be the most definite treatment of aneurysms; therefore, the debate of long-term follow-up in this group was and is controversial. There is a high clinical impact in patients suffering from a cerebral artery aneurysm in which way the neurosurgeons interpret this pathology: a focal vessel wall defect or more general vessel wall instability.
As clearly summarized in this article, many patients treated by clipping after an SAH were considered cured with no reason for follow-up. However, there are studies reporting a higher risk of recurrent SAH in those patients even if all aneurysms at the time of the first event were successfully treated. This article also presents the two many possible findings in a rebleeding: regrowth of the initial aneurysm (e.g., at the neck) or de novo aneurysms; both pathologies indicating that the aneurysm is not a focal defect and a finding that is also reported for endovascular-treated aneurysms (1). As the risk of de novo aneurysms increases over the time, the authors present an interesting and very important evaluation of the value of long-term follow-up diagnostics in patients after aneurysm clipping.
The here presented study and the review of the literature have an important impact on the clinical management of patients after clip therapy of a cerebral artery aneurysm. The authors present an evaluation of the DSA in 20 patients after clipping. The strength of this study is the follow-up time more than 10 years after the first surgery (10–26 years). The authors could find one recurrent aneurysm at the neck of a clipped aneurysm and de novo aneurysms in six patients of whom four had multiple de novo aneurysms. Within this study, there is a relatively high rate of new aneurysms (de novo aneurysm/patient-year, 4.15%). However, one shortcoming is that the statistical interpretations are partly preliminary as the included patient number was low. As the authors very clearly summarize in Table 2, there are further studies with much lower de novo rates. One argument is the follow-up of less than 10 years in most studies, however, two studies also had a follow-up comparable to the here presented one and a 1% de novo aneurysm/patient-year with groups of 40 and 89 patients, respectively. Besides the total number of patients included, the authors discuss correctly the possible overestimation in their study due to one patient with seven de novo aneurysms. Apart from this main topic, the authors present an interesting discussion concerning the point of “true de novo” aneurysms and possible overseen aneurysms in the first examination. Furthermore, CTA and DSA are compared in respect of sensitivity especially for small aneurysms. This point is very important for the method of follow-up; on one hand, the CTA has the advantage of a non-invasive technique, on the other hand, it is known that even small aneurysms (maybe not detectable in CTA) can cause a bleeding in patients with SAH in history.
In total, due to the small group, the authors cannot identify any risk factors for de novo aneurysms apart from SAH in history. However, they come to the conclusion that a long-term follow-up is important for such patients which is a message with high clinical impact. Further studies with larger patient groups or even multi-center registers are necessary to evaluate further risk factors but also the best choice of the method. The higher sensitivity of DSA might be useless if many patients are “lost” because they would not agree to this invasive procedure or clinics could have logistic problems (in recent times also cost restrictions). As newer CT developments (dual source technique) could be even used to analyze the clipped aneurysm, this non-invasive method could be an acceptable alternative (2).
References
1. Plowman RS, Clarke A, Clarke M, Byrne JV (2011) Sixteen-year single-surgeon experience with coil embolization for ruptured intracranial aneurysms: recurrence rates and incidence of late rebleeding. Clinical Article. J Neurosurg 114(3):863–874
2. Fahrendorf DM, Goericke SL, Oezkan N, Breyer T, Hussain S, Sandalcioglu EI, Sure U, Forsting M, Gizewski ER (2011) The value of dual-energy CTA for control of surgically clipped aneurysms. Eur Radiol. doi:10.1007/s00330-011-2147-x
Kiyohiro Houkin, Sapporo, Japan
We empirically know that de novo cerebral aneurysms are occasionally seen in the long-term follow-up after the surgical treatment of aneurysms. However, there are quite few systemic studies on this clinical question since these studies require well-designed long-term observation.
This study revealed the high incidence of the de novo aneurysms seen in patients with multiple aneurysms that is quite intuitively acceptable for neurosurgeons. The conclusion that 30% rate of new aneurysm formation in patients clipped more than a decade ago invokes us that the careful follow-up is indispensable even in 10 years after the surgery.
However, this study includes some drawbacks. Firstly, the possibility of missing of the detection of the aneurysm in the first event is not always ruled out. Secondly, we neurosurgeons, often detect very small aneurysms during surgery that are not correctly diagnosed in the angiography. Indeed, true “de novo” aneurysm is not always clearly discriminated from the missing small aneurysm in the first procedures. However, in any case, the high rate of the new formation of the cerebral aneurysm detected in the long-term follow-up is quite an important suggestion for us. We have to carefully watch the treated patients with cerebral aneurysm considering high incidence of new formation of the cerebral aneurysm in particular when the patient is young and has multiple aneurysms. In addition, we might have to consider some new guidelines for the follow-up of the patients with previously clipped aneurysms based on the similar studies. I sincerely expect that more studies with more number of patients on this issue will be launched in many major institutions.
Marc Sindou, Lyon, France
This study pleads for following the patients with previously ruptured intracranial aneurysms (and also those with asymptomatic aneurysms), especially when they are multiple or developed in dysplastic arteries. For practical reasons, perhaps imaging monitoring should only be CT angiography or MR angiography. Secondary DSA through direct femoral route should be limited to the exploration of patients’ candidates to endovascular or surgical treatment, or when images are difficult to interpret.
Rights and permissions
About this article
Cite this article
Bruneau, M., Rynkowski, M., Smida-Rynkowska, K. et al. Long-term follow-up survey reveals a high yield, up to 30% of patients presenting newly detected aneurysms more than 10 years after ruptured intracranial aneurysms clipping. Neurosurg Rev 34, 485–496 (2011). https://doi.org/10.1007/s10143-011-0332-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-011-0332-7