Abstract
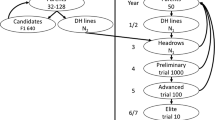
Selective breeding programs for salmonids typically aim to improve traits associated with growth and disease resistance. It has been established that stressors common to production environments can adversely affect these and other traits which are important to producers and consumers. Previously, we employed phenotypic selection to create families that exhibit high or low plasma cortisol concentrations in response to crowding stress. Subsequent crosses of high × low phenotypes founded a multigenerational breeding scheme with the aim of dissecting the genetic basis for variation underlying stress response through the identification of quantitative trait loci (QTL). Multiple methods of QTL analyses differing in their assumptions of homozygosity of the causal alleles in the grandparental generation yielded similar results in the F1 generation, and the analysis of two stress response phenotype measurement indexes were highly correlated. In the current study, we conducted a genome scan with microsatellites to detect QTL in the F2 generation of two families created through phenotypic selection and having larger numbers of offspring than families screened in the previous generation. Seven suggestive and three significant QTL were detected, seven of which were not previously detected in the National Center for Cool and Cold Water Aquaculture germplasm, bringing the total number of chromosomes containing significant and suggestive stress response QTL to 4 and 15, respectively. One significant QTL which peaks at 7 cM on chromosome Omy12 spans 12 cM and explains 25 % of the phenotypic variance in family 2008052 particularly warrants further investigation. Five QTL with significant parent-of-origin effects were detected in family 2008052, including two QTL on Omy12. The 95 % confidence intervals for the remaining QTL we detected were broad, requiring validation and fine mapping with other genotyping approaches and mapping strategies. These results will facilitate identification of potential casual alleles that can be employed in strategies aimed at better understanding the genetic and physiological basis of stress responses to crowding in rainbow trout aquaculture production.

Similar content being viewed by others
Abbreviations
- QTL:
-
Quantitative trait loci
- NCCCWA:
-
National Center for Cool and Cold Water Aquaculture
- BLUP:
-
Best linear unbiased prediction
- EBV:
-
Estimated breeding value
- MSD:
-
Mendelian segregation distortion
- Omy1:
-
Oncorhynchus mykiss chromosome 1
References
Backstrom T, Schjolden J, Overli O, Thornqvist PO, Winberg S (2011) Stress effects on AVT and CRF systems in two strains of rainbow trout (Oncorhynchus mykiss) divergent in stress responsiveness. Horm Behav 59:180–186. doi:10.1016/j.yhbeh.2010.11.008
Boutin-Ganache I, Raposo M, Raymond M, Deschepper CF (2001) M13-tailed primers improve the readability and usability of microsatellite analyses performed with two different allele-sizing methods. Biotechniques 31(24–6):28
De Koning DJ, Rattink AP, Harlizius B, Van Arendonk JM, Brascamp EW, Groenen MM (2000) Genome-wide scan for body composition in pigs reveals important role of imprinting. Proc Natl Acad Sci U S A 97:7947–7950
De Koning DJ, Bovenhuis H, Van Arendonk JM (2002) On the detection of imprinted quantitative trait loci in experimental crosses of outbred species. Genetics 161:931–938
De Koning DJ, Windsor D, Hocking PM, Burt DW, Law A, Haley CS, Morris A, Vincent J, Griffin H (2003) Quantitative trait locus detection in commercial broiler lines using candidate regions. J Anim Sci 81:1158–1165
Desautes C, Bidanelt JP, Milant D, Iannuccelli N, Amigues Y, Bourgeois F, Caritez JC, Renard C, Chevalet C, Mormede P (2002) Genetic linkage mapping of quantitative trait loci for behavioral and neuroendocrine stress response traits in pigs. J Anim Sci 80:2276–2285
Fevolden SE, Refstie T, Røed KH (1992) Disease resistance in rainbow trout (Oncorhynchus mykiss) selected for stress response. Aquaculture 104:19–29
Fevolden S-E, Røed KH, Fjalestad KT (2002) Selection response of cortisol and lysozyme in rainbow trout and correlation to growth. Aquaculture 205:61–75
Fevolden S-E, Røed KH, Fjalestad K (2003) A combined salt and confinement stress enhances mortality in rainbow trout (Oncorhynchus mykiss) selected for high stress responsiveness. Aquaculture 216:67–76
Freyer G, Kuhn C, Weikard R, Zhang Q, Mayer M, Hoeschele I (2002a) Multiple QTL on chromosome six in dairy cattle affecting yield and content traits. J Anim Breed Genet 119:69–82
Freyer G, Stricker C, Kuhn C (2002b) Comparison of estimated breeding values and daughter yield deviations used in segregation and linkage analyses. Czech Journal of Animal Science 47:247–252
Gentleman RC, Carey VJ, BatesDM BB, Dettling M, Dudoit S, Ellis B, Gautier L, Ge YC, Gentry J, Hornik K, Hothorn T, Huber W, Iacus S, Irizarry R, Leisch F, Li C, Maechler M, Rossini AJ, Sawitzki G, Smith C, Smyth G, Tierney L, Yang JYH, Zhang JH (2004) Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 5(10):R80
Gilmour AR, Gogel BJ, Cullis BR, Thompson R (2006) ASReml User Guide Release 2.0. VSN, Hemel Hempstead
Gjedrem T (1992) Breeding plans for rainbow-trout. Aquaculture 100:73–83
Goll MG, Halpern ME (2011) DNA methylation in zebrafish. Prog Mol Biol Transl Sci 101:193–218. doi:10.1016/B978-0-12-387685-0.00005-6
Haffray P, Vandeputte M, Petit V, Pincent C, Chatain B, Chapuis H, Meriaux JC, Coudurier B, Quillet E, Dupont-Nivet M (2012) Minimizing maternal effect in salmonid families mixed since eyed stages and a posteriori DNA-pedigreed. Livest Sci 150:170–178. doi:10.1016/j.livsci.2012.08.017
Hager R, Cheverud JM, Wolf JB (2008) Maternal effects as the cause of parent-of-origin effects that mimic genomic imprinting. Genetics 178:1755–1762. doi:10.1534/genetics.107.080697
Haley CS, Knott SA, Elsen JM (1994) Mapping quantitative trait loci in crosses between outbred lines using least-squares. Genetics 136:1195–1207
Henryon M, Berg P, Olesen NJ, Kjaer TE, Slierendrecht WJ, Jokumsen A, Lund I (2005) Selective breeding provides an approach to increase resistance of rainbow trout (Oncorhynchus mykiss) to the diseases, enteric redmouth disease, rainbow trout fry syndrome, and viral haemorrhagic septicaemia. Aquaculture 250:621–636. doi:10.1016/j.aquaculture.2004.12.022
Hoskonen P, Pirhonen J (2006) Effects of repeated handling, with or without anaesthesia, on feed intake and growth in juvenile rainbow trout, Oncorhynchus mykiss (Walbaum). Aquac Res 37:409–415
Imumorin IG, Kim EH, Lee YM, De Koning DJ, Van Arendonk JA, De Donato M, Taylor JF, Kim JJ (2011) Genome scan for parent-of-origin QTL effects on bovine growth and carcass traits. Front Genet 2:44. doi:10.3389/fgene.2011.00044
Janhunen M, Kause A, Vehvilainen H, Jarvisalo O (2012) Genetics of microenvironmental sensitivity of body weight in rainbow trout (Oncorhynchus mykiss) selected for improved growth. PLoS One 7:e38766. doi:10.1371/journal.pone.0038766
Janss LLG (2008) iBay manual version 1.46. Janss Biostatistics, Leiden, P.O. Box 535
Johansen IB, Sandvik GK, Nilsson GE, Bakken M, Overli O (2011) Cortisol receptor expression differs in the brains of rainbow trout selected for divergent cortisol responses. Comp Biochem Physiol Part D Genomics Proteomics 6:126–132. doi:10.1016/j.cbd.2010.11.002
Kause A, Paananen T, Ritola O, Koskinen H (2007) Direct and indirect selection of visceral lipid weight, fillet weight, and fillet percentage in a rainbow trout breeding program. J Anim Sci 85:3218–3227. doi:10.2527/jas.2007-0332
Knott SA (2005) Regression-based quantitative trait loci mapping: robust, efficient and effective. Philosophical Transactions of the Royal Society B-Biological Sciences 360:1435–1442. doi:10.1098/rstb.2005.1671
Knott SA, Elsen JM, Haley CS (1996) Methods for multiple-marker mapping of quantitative trait loci in half-sib populations. Theor Appl Genet 93:71–80
Knott SA, Neale DB, Sewell MM, Haley CS (1997) Multiple marker mapping of quantitative trait loci in an outbred pedigree of loblolly pine. Theor Appl Genet 94:810–820
Knott SA, Marklund L, Haley CS, Andersson K, Davies W, Ellegren H, Fredholm M, Hansson I, Hoyheim B, Lundstrom K, Moller M, Andersson L (1998) Multiple marker mapping of quantitative trait loci in a cross between outbred wild boar and large white pigs. Genetics 149:1069–1080
Lankford SE, Weber GM (2006) Associations between plasma growth hormone, insulin-like growth factor-I, and cortisol with stress responsiveness and growth performance in a selective breeding program for rainbow trout. N Am J Aquacult 68:151–159. doi:10.1577/A05-014.1
Leach RJ, Craigmile SC, Knott SA, Williams JL, Glass EJ (2010) Quantitative trait loci for variation in immune response to a Foot-and-Mouth Disease virus peptide. BMC Genet. doi:10.1186/1471-2156-11-107
Leeds TD, Silverstein JT, Weber GM, Vallejo RL, Palti Y, Rexroad CE 3rd, Evenhuis J, Hadidi S, Welch TJ, Wiens GD (2010) Response to selection for bacterial cold water disease resistance in rainbow trout. J Anim Sci 88:1936–1946. doi:10.2527/jas.2009-2538
Lefèvre F, Bugeon J, Aupérin B, Aubin J (2008) Rearing oxygen level and slaughter stress effects on rainbow trout flesh quality. Aquaculture 284:81–89
Matise TC, Perlin M, Chakravarti A (1994) Automated construction of genetic linkage maps using an expert system (MultiMap): a human genome linkage map. Nat Genet 6:384–390. doi:10.1038/ng0494-384
Mcgowan RA, Martin CC (1997) DNA methylation and genome imprinting in the zebrafish, Danio rerio: some evolutionary ramifications. Biochem Cell Biol 75:499–506
Merkin GV, Roth B, Gjerstad C, Dahl-Paulsen E, Nortvedt R (2010) Effect of pre-slaughter procedures on stress responses and some quality parameters in sea-farmed rainbow trout (Oncorhynchus mykiss). Aquaculture 309:231–235
Navarro-Martin L, Vinas J, Ribas L, Diaz N, Gutierrez A, Di Croce L, Piferrer F (2011) DNA methylation of the gonadal aromatase (cyp19a) promoter is involved in temperature-dependent sex ratio shifts in the European sea bass. PLoS Genet 7:e1002447. doi:10.1371/journal.pgen.1002447
Ott J (1999) Analysis of human genetic linkage. The John Hopkins University Press, Baltimore
Overli O, Pottinger TG, Carrick TR, Overli E, Winberg S (2001) Brain monoaminergic activity in rainbow trout selected for high and low stress responsiveness. Brain Behav Evol 57:214–224
Øverli Ø, Sørensen C, Kiessling A, Pottinger TG, Gjøen HM (2006) Selection for improved stress tolerance in rainbow trout (Oncorhynchus mykiss) leads to reduced feed waste. Aquaculture 261:776–781
Overturf K, Bullock D, Lapatra S, Hardy R (2004) Genetic selection and molecular analysis of domesticated rainbow trout for enhanced growth on alternative diet sources. Environ Biol Fish 69:409–418
Pemmasani JK, Pottinger TG, Cairns MT (2011) Analysis of stress-induced hepatic gene expression in rainbow trout (Oncorhynchus mykiss) selected for high- and low-responsiveness to stress. Comp Biochem Physiol Part D Genomics Proteomics 6:406–419. doi:10.1016/j.cbd.2011.09.001
Pickering AD (1992) Rainbow trout husbandry: management of the stress response. Aquaculture 100:125–139
Poli BM, Parisi G, Scappini F, Zampacavallo G (2005) Fish welfare and quality as affected by pre-slaughter and slaughter management. Aquacult Int 13:29–49
Pottinger TG (2006) Context dependent differences in growth of two rainbow trout (Oncorhynchus mykiss) lines selected for divergent stress responsiveness. Aquaculture 256:140–147
Pottinger TG, Carrick TR (1999) Modification of the plasma cortisol response to stress in rainbow trout by selective breeding. Gen Comp Endocrinol 116:122–132. doi:10.1006/gcen.1999.7355
Pottinger TG, Carrick TR (2000) Indicators of reproductive performance in rainbow trout Oncorhynchus mykiss (Walbaum) selected for high and low responsiveness to stress. Aquac Res 31:367–375. doi:10.1046/j.1365-2109.2000.00450.x
Pottinger TG, Carrick TR (2001a) ACTH does not mediate divergent stress responsiveness in rainbow trout. Comp Biochem Physiol A Mol Integr Physiol 129:399–404
Pottinger TG, Carrick TR (2001b) Stress responsiveness affects dominant–subordinate relationships in rainbow trout. Horm Behav 40:419–427. doi:10.1006/hbeh.2001.1707
Rebl A, Verleih M, Kollner B, Korytar T, Goldammer T (2012a) Duplicated NELL2 genes show different expression patterns in two rainbow trout strains after temperature and pathogen challenge. Comp Biochem Physiol B Biochem Mol Biol 163:65–73. doi:10.1016/j.cbpb.2012.05.001
Rebl A, Verleih M, Korytar T, Kuhn C, Wimmers K, Kollner B, Goldammer T (2012b) Identification of differentially expressed protective genes in liver of two rainbow trout strains. Vet Immunol Immunopathol 145:305–315. doi:10.1016/j.vetimm.2011.11.023
Rexroad CE 3rd, Palti Y, Gahr SA, Vallejo RL (2008) A second generation genetic map for rainbow trout (Oncorhynchus mykiss). BMC Genet 9:74. doi:10.1186/1471-2156-9-74
Rexroad CE, Vallejo RL, Liu S, Palti Y, Weber GM (2012) A survey of QTL affecting stress response to crowding in rainbow trout in a single broodstock population. BMC Genet 13:97
Rowe SJ, Pong-Wong R, Haley CS, Knott SA, De Koning DJ (2009) Detecting parent of origin and dominant QTL in a two-generation commercial poultry pedigree using variance component methodology. Genet Sel Evol. doi:10.1186/1297-9686-41-6
Sae-Lim P, Komen H, Kause A, Van Arendonk JM, Barfoot AJ, Martin KE, Parsons JE (2012) Defining desired genetic gains for rainbow trout breeding objective using analytic hierarchy process. J Anim Sci 90:1766–1776. doi:10.2527/Jas2011-4267
Sandor C, Georges M (2008) On the detection of imprinted quantitative trait loci in line crosses: effect of linkage disequilibrium. Genetics 180:1167–1175. doi:10.1534/genetics.108.092551
Sas (2007) SAS 9.1.3 help and documentation. SAS Institute, Cary
Schjolden J, Backstrom T, Pulman KG, Pottinger TG, Winberg S (2005) Divergence in behavioural responses to stress in two strains of rainbow trout (Oncorhynchus mykiss) with contrasting stress responsiveness. Horm Behav 48:537–544. doi:10.1016/j.yhbeh.2005.04.008
Seaton G, Hernandez J, Grunchec JA, White I, Allen J, De Koning D J, Wei W, Berry D, Haley C & Knott S (2006) GridQTL: A Grid Portal for QTL mapping of compute intensive datasets. 8th World Congress on Genetics Applied to Livestock Production. Belo Horizonte, Brazil
Silverstein JT, Rexroad CE, King TL (2004) Genetic variation measured by microsatellites among three strains of domesticated rainbow trout (Oncorhynchus mykiss, Walbaum). Aquac Res 35:40–48
Thomsen H, Reinsch N, Xu N, Looft C, Grupe S, Kuhn C, Brockmann GA, Schwerin M, Leyhe-Horn B, Hiendleder S, Erhardt G, Medjugorac I, Russ I, Forster M, Brenig B, Reinhardt F, Reents R, Blumel J, Averdunk G, Kalm E (2001) Comparison of estimated breeding values, daughter yield deviations and de-regressed proofs within a whole genome scan for QTL. J Anim Breed Genet 118:357–370
Trenzado CE, Carrick TR, Pottinger TG (2003) Divergence of endocrine and metabolic responses to stress in two rainbow trout lines selected for differing cortisol responsiveness to stress. Gen Comp Endocrinol 133:332–340
Tuiskula-Haavisto M, De Koning D-J, Honkatukia M, Schulman NF, Mäki-Tanila A, Vilkki J (2004) Quantitative trait loci with parent-of-origin effects in chicken. Genetics Research 84:57–66. doi:10.1017/S0016672304006950
Uddin MJ, Duy DN, Cinar MU, Tesfaye D, Tholen E, Juengst H, Looft C, Schellander K (2011) Detection of quantitative trait loci affecting serum cholesterol, LDL, HDL, and triglyceride in pigs. BMC Genet. doi:10.1186/1471-2156-12-62
Vallejo RL, Rexroad CE 3rd, Silverstein JT, Janss LL, Weber GM (2009) Evidence of major genes affecting stress response in rainbow trout using Bayesian methods of complex segregation analysis. J Anim Sci 87:3490–3505. doi:10.2527/jas.2008-1616
Venturelli PA, Shuter BJ, Murphy CA (2009) Evidence for harvest-induced maternal influences on the reproductive rates of fish populations. P R Soc B 276:919–924. doi:10.1098/rspb.2008.1507
Visscher PM, Thompson R, Haley CS (1996) Confidence intervals in QTL mapping by bootstrapping. Genetics 143:1013–1020
Weber GM, Silverstein JT (2007) Evaluation of a stress response for use in a selective breeding program for improved growth and disease resistance in rainbow trout. N Am J Aquacult 69:69–79. doi:10.1577/A05-103.1
Weber GM, Vallejo RL, Lankford SE, Silverstein JT, Welch TJ (2008) Cortisol response to a crowding stress: heritability and association with disease resistance to Yersinia ruckeri in rainbow trout. N Am J Aquacult 70:425–433. doi:10.1577/A07-059.1
Weller JI (2009) Quantitative trait analysis in animals. CABI, Cambridge
Wendelaar Bonga SE (1997) The stress response in fish. Physiol Rev 77:591–625
Acknowledgments
We would like to acknowledge the following individuals for providing excellent technical assistance including Roseanna Long, Kristy Shewbridge, and Brian Smith for genotyping; Lisa Radler, Jill Birkett, Mark Hostuttler and David Payne for phenotyping fish for stress response; and Josh Kretzer, Jim Everson, Kyle Jenkins, Jenea McGowan, Kevin Melody for animal care. We also thank Guangtu Gao for processing the genotype data files. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Author information
Authors and Affiliations
Corresponding author
Additional information
Caird E. Rexroad and Roger L. Vallejo contributed equally to this work.
Rights and permissions
About this article
Cite this article
Rexroad, C.E., Vallejo, R.L., Liu, S. et al. Quantitative Trait Loci Affecting Response to Crowding Stress in an F2 Generation of Rainbow Trout Produced Through Phenotypic Selection. Mar Biotechnol 15, 613–627 (2013). https://doi.org/10.1007/s10126-013-9512-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-013-9512-5