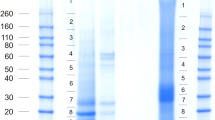
Abstract
Shell nacre is laid upon an organic cell-free matrix, part of which, paradoxically, is water soluble and displays biological activities. Proteins in the native shell also constitute an insoluble network and offer a model for studying supramolecular organization as a means of self-ordering. Consequently, difficulties are encountered in extraction and purification strategies for protein characterization. In this work, water-soluble proteins and the insoluble conhiolin residue of the nacre of Pinctada margaritifera matrix were analyzed via a proteomics approach. Two sequences homologous to nacre matrix proteins of other Pinctada species were identified in the water-soluble extract. One of them is known as a fundamental component of the insoluble organic matrix of nacre. In the conchiolin, the insoluble residue, four homologs of Pinctada nacre matrix proteins were found. Two of them were the same as the molecules characterized in the water-soluble extract. Results established that soluble and insoluble proteins of the nacre organic matrix share constitutive material. Surprisingly, a peptide in the conchiolin residue was found homologous to a prismatic matrix protein of Pinctada fucata, suggesting that prismatic and nacre matrices may share common proteins. The insoluble properties of shell matrix proteins appear to arise from structural organization via multimerization. The oxidative activity, found in the water-soluble fraction of the nacre matrix, is proposed as a leading process in the transformation of transient soluble proteins into the insoluble network of conchiolin during nacre growth.






Similar content being viewed by others
References
Albeck S, Addadi L, Weiner S (1996) Regulation of calcite crystal morphology by intracrystalline acidic proteins and glycoproteins. Connect Tissue Res 35, 365–370
Bédouet L, Rusconi F, Rousseau M, Duplat D, Marie A, Dubost L, Le Ny K, Berland S, Péduzzi J, Lopez E (2006) Low molecular weight molecules as new components of the nacre organic matrix. Comp Biochem Physiol B 144, 532–543
Claus H (2004) Laccases: structure, reactions, distribution. Micron 35, 93–96
Conyers SM, Kidwell DA (1991) Chromogenic substrates for horseradish peroxidase. Anal Biochem 192, 207–211
Gotliv B, Addadi L, Weiner S (2003) Mollusk shell acidic proteins: in search of individual functions. Chem Bio Chem 4, 522–529
Hattan SJ, Laue TM, Chasteen ND (2001) Purification and characterization of a novel calcium-binding protein from the extrapallial fluid mollusc, Mytilus edulis. J Biol Chem 276, 4461–4468
Kono M, Hayashi N, Samata T (2000) Molecular mechanism of the nacreous layer formation in Pinctada maxima. Biochem Biophys Res Commun 269, 213–218
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685
Lamghari M, Almeida MJ, Berland S, Huet H, Laurent A, Milet C, Lopez E (1999) Stimulation of bone marrow cells and bone formation by nacre in vivo and in vitro studies. Bone 25 (Suppl 1), 91S–94S
Marin F, Luquet G (2004) Molluscan shell protein. C R Palevol 6–7, 469–492
Marin F, Amons R, Guichard N, Stitger M, Hecker A, Luquet G, Layrolle P, Alcaraz G, Riondet C, Westbroek P (2005) Caspartin and calprismin, two proteins of the shell calcitic prisms of the mediterranean fan mussel Pinna nobilis. J Biol Chem 280, 33895–33908
Marxen JC, Nimtz M, Becker W, Mann K (2003) The major soluble 196 kDa protein of the organic shell matrix of the freshwater snail Biomphalaria glabrata is an N-glycosylated dermatopontin. Biochim Biophys Acta 1650, 92–98
Mayer AM, Staples RC (2002) Laccases: new functions for an old enzyme. Phytochemistry 60, 551–565
Michenfelder M, Fu G, Lawrence C, Weaver JC, Wustman BA, Taranto L, Evans JS, Morse DE (2003) Characterization of two molluscan crystal-modulating biomineralization proteins and identification of putative mineral binding domains. Biopolymers 70, 522–533
Monsigny M, Petit C, Roche AC (1988) Colorimetric determination of neutral sugars by a resorcinol sulfuric acid micromethod. Anal Biochem 175, 525–530
Mouries LP, Almeida MJ, Milet C, Berland S, Lopez E (2002) Bioactivity of nacre water-soluble organic matrix from the bivalve mollusk Pinctada maxima in three mammaliancell types: fibroblasts, bone marrow stromal cells and osteoblasts. Comp Biochem Physiol B, 132, 217–229
Pereira-Mouriès L, Almeida MJ, Ribeiro C, Peduzzi J, Barthélemy M, Milet C, Lopez E (2002) Soluble silk-like organic matrix in the nacreous layer of the bivalve Pinctada maxima A new insight in the biomineralization field. Eur J Biochem 269, 4994–5003
Rousseau M, Pereira-Mouriès L, Almeida MJ, Milet C, Lopez E (2003) The water-soluble matrix fraction extracted from the nacre of Pinctada maxima produces earlier mineralization of MC3T3-E1 mouse pre-osteoblasts. Comp Biochem Physiol A 135, 271–278
Samata T (2004) Recent advances in studies on nacreous layer biomineralization. Molecular and cellular aspects. Thalassas 20, 25–44
Shen X, Belcher AM, Hansma PK, Stucky GD, Morse DE (1997) Molecular cloning and characterization of lustrin A, a matrix protein from the shell and pearl nacre of Haliotis rufescens. J Biol Chem 272, 32472–32481
Sudo S, Fujikawa T, Nagakura T, Ohkubo K, Sakaguchi K, Tanaka M, Nakashima K, Takahashi T (1997) Structures of mollusc shell framework proteins. Nature 387, 563–564
Suzuki M, Murayama E, Inoue H, Ozaki N, Tohse H, Kogure T, Nagasawa H (2004) Charaterization of Prismalin-14, a novel matrix protein from the prismatic layer of the Japanese pearl oyster (Pinctada fucata). Biochem J 382, 205–213
Weiner S (1986) Organization of extracellularly mineralized tissues: a comparative study of biological crystal growth. CRC Crit Rev Biochem 20, 365–408
Westbroek P, Marin F (1998) A marriage of bone and nacre. Nature (News and views) 392, 861–862
Xu F, Koch DE, Kong IC, Hunter RP, Bhandari A (2005) Peroxidase-mediated oxidative coupling of 1-naphthol: characterization of polymerization products. Water Res 39, 2358–2368
Zhang C, Zhang R (2006) Matrix proteins in the outer shells of molluscs. Mar Biotechnol 8, 572–586
Zhang C, Xie L, Huang J, Liu X, Zhang R (2006) A novel matrix protein family participating in the prismatic layer framework formation of pearl oyster, Pinctada fucata. Biochem Biophys Res Commun 344, 735–740
Acknowledgments
The authors thank Sandrine Borzeix (Muséum National d’Histoire Naturelle, Département des Milieux et Peuplements Aquatiques, UMR 5178, CNRS-MNHN Biologie des Organismes Marins et Ecosystèmes) for the artwork, and SIERA S.A. for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bédouet, L., Marie, A., Dubost, L. et al. Proteomics Analysis of the Nacre Soluble and Insoluble Proteins from the Oyster Pinctada margaritifera . Mar Biotechnol 9, 638–649 (2007). https://doi.org/10.1007/s10126-007-9017-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-007-9017-1