Abstract
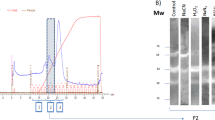
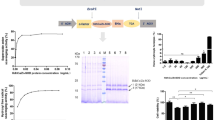
Copper/zinc superoxide dismutase was cloned from the zebrafish (Danio rerio). The full coding region of the zebrafish superoxide dismutase (ZSOD) complementary DNA was ligated with pET-20b(+) and successfully expressed in Escherichia coli strain AD494(DE3)pLysS. The active enzyme was purified by His tagging. The ZSOD yield was 6 mg from 0.2 L of E. coli culture, and the specific activity was 2000 U/mg as assayed using a RANSOD kit. The enzyme stability was characterized by reaction to temperature, pH, and detergent treatment. The results showed enzyme activity was still active after heat treatment at 70°C for 10 minutes, resistant to pH treatment from 2.3 to 12, and resistant to treatment with sodium dodecyl sulfate (SDS) under 4%. In addition, the recombinant ZSOD was used to protect fish from 100 ppm of paraquat-induced oxidative injury by soaking fish larva in 55 µg/ml SOD enzyme. The results were significant.







Similar content being viewed by others
References
C. Beauchamp I. Fridovich (1971) ArticleTitleImproved assays and an assay applicable to acrylamide gel. Anal Biochem 44 276–287 Occurrence Handle1:CAS:528:DyaE38XjtFKhsg%3D%3D Occurrence Handle4943714
B.J. Day M. Patel L. Calavetta L.Y. Chang J.S. Stamler (1999) ArticleTitleA mechanism of paraquat toxicity involving nitric oxide synthase. Proc Natl Acad Sci USA 96 IssueID22 12760–12765 Occurrence Handle10.1073/pnas.96.22.12760 Occurrence Handle1:CAS:528:DyaK1MXnt1yqsbk%3D Occurrence Handle10535996
M.St. Dimitrova V. Tishinova V. Velcheva (1994) ArticleTitleCombined effect of zinc and lead on the hepatic superoxide dismutase-catalase system in carp (cyprinus carpio). Comp Biochem Physiol 108C 43–46
A.D. Dodge (1971) ArticleTitleThe mode of action of the bipyridylium herbicides, paraquat and diquat. Endeavour 30 IssueID111 130–135 Occurrence Handle1:CAS:528:DyaE3MXkslGisL8%3D Occurrence Handle4110469
H.J. Forman I. Fridovich (1973) ArticleTitleOn the stability of bovine superoxide dismutase: the effects of metals. J Biol Chem 248 2645–2649 Occurrence Handle1:CAS:528:DyaE3sXktVWqsr8%3D Occurrence Handle4697386
A. Garcia-Gonzalez J.L. Ochoa (1990) ArticleTitleAnti-inflammatory activity of Debaryomyces hansenii Cu, Zn-SOD. Arch Med Res 30 69–73 Occurrence Handle10.1016/S0188-0128(98)00005-0
R.A. Hallewell K.C. Imlay P. Lee N.M. Fong C. Gallegos E.D. Getzoff J.A. Tainer D.E. Cabelli P. Tekamp-Olson G.T. Mullenbach (1991) ArticleTitleThermostabilization of recombinant human and bovine CuZn superoxide dismutases by replacement of free cysteines. Biochem Biophys Res Commun 181 IssueID1 474–480 Occurrence Handle1:CAS:528:DyaK3sXjsFShsw%3D%3D Occurrence Handle1958215
B. Halliwell J.M. Gutteridge (1984) ArticleTitleLipid peroxidation, oxygen radicals, cell damage, and antioxidant therapy. Lancet 1 1396–1397 Occurrence Handle1:STN:280:BiuB2cvmslM%3D Occurrence Handle6145845
J.J. Hu N. Dubin D. Kurland B.L. Ma G.C. Roush (1995) ArticleTitleThe effects of hydrogen peroxide on DNA repair activities. Mutat Res 336 193–201 Occurrence Handle10.1016/0921-8777(94)00054-A Occurrence Handle1:CAS:528:DyaK2MXjvVelur4%3D Occurrence Handle7885389
InstitutionalAuthorNameIPCS (1984) Paraquat and Diquat, Environmental Health Criteria. WHO Geneva 39:13–128
M.J. Kelner R. Bagnell M. Montoya L. Estes S.F. Uglik P. Cerutti (1995) ArticleTitleTransfection with human copper-zinc superoxide dismutase induces bidirectional alterations in other antioxidant enzymes, proteins, growth factor response, and paraquat resistance. Free Radic Biol Med 18 497–506 Occurrence Handle10.1016/0891-5849(94)00167-I Occurrence Handle1:CAS:528:DyaK2MXjsFyhu7g%3D Occurrence Handle9101240
C.F. Ken J.F. Shaw J.L. Wu C.T. Lin (1998) ArticleTitleMolecular cloning of a cDNA coding for a Cu/Zn-superoxide dismutase from zebrafish and overexpression in Escherichia coli. J Agri Food Chem 46 2863–2867 Occurrence Handle10.1021/jf970763e Occurrence Handle1:CAS:528:DyaK1cXktFKrtLo%3D
F. Komada K. Nishiguchi Y. Tanigawara T. Akamatsu X.Y. Wu S. Iwakawa K. Okumura (1996) ArticleTitleEffect of transfection with superoxide dismutase expression plasmid on superoxide anion induced cytotoxicity in cultured rat lung cells. Biol Pharm Bull 19 IssueID2 274–279 Occurrence Handle1:CAS:528:DyaK28Xht1Ogsrs%3D Occurrence Handle8850321
T. Kondo H. Terajima T. Todoroki T. Hirano Y. Ito T. Usia K. Messmer (1999) ArticleTitlePrevention of hepatic ischemia-reperfusion injury by SOD-DIVEMA conjugate. J Surg Res 85 IssueID1 26–36 Occurrence Handle10.1006/jsre.1999.5591 Occurrence Handle1:CAS:528:DyaK1MXktVSltLw%3D Occurrence Handle10383834
C.T. Lin M.T. Lin Y.T. Chen J.F. Shaw (1995) ArticleTitleSubunit interaction enhances enzyme activity and stabilities of sweet potato cytosolic Cu/Zn-superoxide dismutase purified by a His-tagged recombinant protein method. Plant Mol Biol 28 303–311 Occurrence Handle1:CAS:528:DyaK2MXmvVGqs74%3D Occurrence Handle7599315
C.T. Lin T.J. Kuo J.F. Shaw M.C. Kao (1999) ArticleTitleCharacterization of the dimer-monomer equilibrium of the papaya Cu/Zn superoxide dismutase and its equilibrium shift by a single amino acid mutation. J Agri Food Chem 47 2944–2949 Occurrence Handle10.1021/jf981355t Occurrence Handle1:CAS:528:DyaK1MXjsFajtrs%3D
M.T. Lin T.J. Kuo C.T. Lin (1998) ArticleTitleMolecular cloning of a cDNA encoding copper/zinc-superoxide dismutase from papaya fruit and overexpression in Escherichia coli. J Agri Food Chem 46 344–348 Occurrence Handle10.1021/jf9706843 Occurrence Handle1:CAS:528:DyaK1cXhs1Ggtw%3D%3D
D.P. Malinowski I. Fridovich (1979) ArticleTitleSubunit association and side-chain reactivities of bovine erythrocyte superoxide dismutase in denaturing solvents. Biochemistry 18 5055–5060 Occurrence Handle1:CAS:528:DyaE1MXmtVKgtbs%3D Occurrence Handle115491
E. Mather-Mihaich R.T. Diguilio (1986) ArticleTitleAntioxidant enzyme activities and malondialdehyde, glutathione and methemoglobin concentration in channel catfish exposed to DFF and N-butylmercaptan. Comp Biochem Physiol 85C 427–432 Occurrence Handle1:CAS:528:DyaL2sXnvF2qsQ%3D%3D
E. McLean R. Ash (1990) ArticleTitleModified uptake of the protein antigen horseradish peroxidase (HRP), following oral delivery to rainbow trout, Oncorhynchus mykiss. Aquaculture 87 373–379 Occurrence Handle1:CAS:528:DyaK3MXjslSr
D.E. McRee S.M. Redford E.D. Getzoff J.R. Lepock R.A. Hallewell J.A. Tainer (1990) ArticleTitleChanges in crystallographic structure and thermostability of a Cu,Zn superoxide dismutase mutant resulting from the removal of a buried cysteine. J Biol Chem 265 14234–14241 Occurrence Handle1:CAS:528:DyaK3cXls1yjsr8%3D Occurrence Handle2387847
T. Ogata S. Manabe (1990) ArticleTitleCorrelation between lipid peroxidation and morphological manifestation of paraquat-induced lung injury in rats. Arch Toxicol 64 IssueID1 7–13 Occurrence Handle1:CAS:528:DyaK3cXhtlCitrc%3D Occurrence Handle2306198
W.C. Orr R.S. Sohal (1994) ArticleTitleExtension of life-span by overexpression of superoxide dismutase and catalase in Drosophila melanogaster. Science 263 1128–1130 Occurrence Handle1:CAS:528:DyaK2cXhvVOlsr4%3D Occurrence Handle8108730
J.I. Park C.M. Grant M.J. Davies I.W. Dawes (1998) ArticleTitleThe cytoplasmic Cu,Zn superoxide dismutase of saccharomyces cerevisiae is required for resistance to freeze-thaw stress: generation of free radicals during freezing and thawing. J Biol Chem 273 22921–22928 Occurrence Handle10.1074/jbc.273.36.22921 Occurrence Handle1:CAS:528:DyaK1cXlvFalurw%3D Occurrence Handle9722512
J.R. Pedrajas J. Peinado J. Lopez-Barea (1993) ArticleTitlePurification of Cu, Zn-superoxide dismutase isoenzymes from fish liver: appearance of new isoforms as a consequence of pollution. Free Rad Comms 19 29–41 Occurrence Handle1:CAS:528:DyaK2cXnt1Sltw%3D%3D
A.A.R. Radi B. Marcovics (1988) ArticleTitleEffects of metal ions on the antioxidant enzyme activities, protein contents and lipid peroxidation of carp tissues. Comp Biochem Physiol 90C 69–72 Occurrence Handle1:CAS:528:DyaL1cXltVKjsb8%3D
P. Smith D. Heath (1976) ArticleTitleParaquat. Crit Rev Toxicol 4 411–445 Occurrence Handle1:CAS:528:DyaE2sXltFWhsw%3D%3D
J.A. Tainer E.D. Getzoff K.M. Beem J.S. Richardson D.C. Richardson (1982) ArticleTitleDetermination and analysis of the 2 A structure and mechanism of copper/zinc superoxide dismutase. J Mol Biol 160 181–217 Occurrence Handle7175933
H. Wiseman B. Halliwell (1996) ArticleTitleDamage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem J 313 17–29 Occurrence Handle1:CAS:528:DyaK28XivVOgtA%3D%3D Occurrence Handle8546679
S.M. Wu P.P. Hwang C.L. Hew J.L. Wu (1998) ArticleTitleEffect of antifreeze protein on cold tolerance in juveniles of tilapia (Oreochromis mossambicus Peters) and milkfish (Chanos chanos Forsskal). Zool Stud 37 39–44 Occurrence Handle1:CAS:528:DyaK1cXitlChsL8%3D
H.Y. Yoo S.S. Kim H.M. Rho (1999) ArticleTitleOverexpression and simple purification of human superoxide dismutase (SOD1) in yeast and its resistance to oxidative stress. J Biotechnol 68 29–35 Occurrence Handle10.1016/S0168-1656(98)00188-6 Occurrence Handle1:CAS:528:DyaK1MXksFKjug%3D%3D Occurrence Handle10036768
Acknowledgements
This work was partially supported by the National Science Council of the Republic of China under Grant NSC 89-2313-B-019-066 to C.-T. L. and supported by the Council of Agriculture, Executive Yuan under Grant 90-BT-2.1-FID-F1 (3) to C.-T. L.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ken, CF., Lin, CT., Shaw, JF. et al. Characterization of Fish Cu/Zn–Superoxide Dismutase and Its Protection from Oxidative Stress . Mar. Biotechnol. 5, 167–173 (2003). https://doi.org/10.1007/s10126-002-0058-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10126-002-0058-1