Abstract:
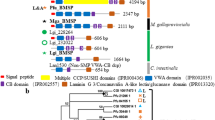
The complete primary structure of MSP-1, a major water-soluble glycoprotein in the foliated calcite shell layer of the scallop Patinopecten yessoensis, is reported. The full-length complementary DNA for MSP-1 isolated by polymerase chain reaction contained a sequence for a signal peptide of 20 amino acids followed by a polypeptide of 820 amino acids with calculated molecular mass of 74.5 kDa. The deduced amino acid sequence of MSP-1 includes a high proportion of Ser (32%), Gly (25%), and Asp (20%), and the predicted isoelectric point is 3.2; in these respects, MSP-1 is a typical acidic glycoprotein of mineralized tissues. A repeated modular structure characterizes MSP-1, with a sequence unit between 158 and 177 amino acids in length being repeated 4 times in tandem in the middle part of the protein. The repeated unit comprises 3 modules (SG, D, and K domains), each having a distinct amino acid composition and sequence. The SG domain is almost exclusively composed of Ser and Gly residues. The D domain is rich in Asp residues, potential N-glycosylation and phosphorylation sites. The K domain is rich in Gly residues and has a core of basic residues. The Asp residues are arranged more or less regularly in the D domains, exhibiting some repeated motifs such as Asp-Gly-Ser-Asp and Asp-Ser-Asp. Further, the 4 D domains indicate remarkable overall sequence similarities to each other. These observations suggest that the regular arrangements of COO− groups in the D domain side chains may be important for specific control of crystal growth.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received September 19, 2000; accepted February 9, 2001
Rights and permissions
About this article
Cite this article
Sarashina, I., Endo, K. The Complete Primary Structure of Molluscan Shell Protein 1 (MSP-1), an Acidic Glycoprotein in the Shell Matrix of the Scallop Patinopecten yessoensis . Mar. Biotechnol. 3, 362–369 (2001). https://doi.org/10.1007/s10126-001-0013-6
Issue Date:
DOI: https://doi.org/10.1007/s10126-001-0013-6