Abstract
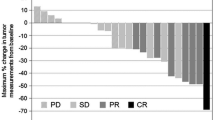
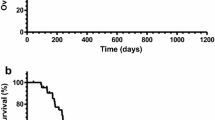
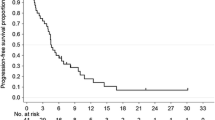
Several chemotherapy regimens used against advanced gastric cancer have been studied extensively over the decades in an attempt to further improve the prognosis of patients. To date, no standard chemotherapeutic regimens have been established worldwide. S-1 (TS-1®), a combination of ftorafur and two modulators, gimestat (CDHP) and oxonic acid, in a molar ratio of 1 : 0.4 : 1, has been widely used in Japan for the treatment of advanced gastric cancer, and much attention has been paid to attempts to increase its antitumor effect by combining it with other chemotherapeutic drugs. We treated 12 patients with advanced gastric cancer with 80 mg/m2 of S-1 for 21 days and 60 mg/m2 of cisplatin (CDDP) on day 8 every 5 weeks. The treatment was continued until disease progression, unacceptable toxicity, or the patient's refusal. Eight out of 12 evaluable patients achieved a partial response (PR), with a response rate of 66.7%. The incidence of grade 3 or 4 adverse effects, including myelosuppression and gastrointestinal toxicities, was 16.6%. None of the patients treated with this regimen died of adverse effects and none required hospitalization for the toxicity. We conclude that the combination of S-1 and CDDP seems to have a high therapeutic index, enhancing the antitumor effect of S-1 while maintaining modest adverse effects, thus suggesting the possible use of this combination based at the outpatient clinic (apart from a short stay in hospital during the infusion of CDDP with hydration). Further study with a large number of patients may be needed to confirm the combination of S-1 and CDDP to be an appropriate first-line chemotherapy for gastric cancer.
Article PDF
Similar content being viewed by others
References
Mayer RJ. Newer cytotoxic agents for advanced colon cancer. ASCO Educational Book. 2000. p. 625–30
GC Heggie JP Sommadossi DS Cross WJ Huster RB Diasio (1987) ArticleTitleClinical pharmacokinetics of 5-fluorouracil and its metabolites in plasma, urine, and bile Cancer Res 47 2003–6
RB Diasio (1998) ArticleTitleThe role of dihydropyrimidine dehydrogenase (DPD) modulation in 5-FU pharmacology Oncology 12 23–7 Occurrence Handle9830621 Occurrence Handle1:STN:280:DyaK1M%2FkvVeisQ%3D%3D
T Shirasaka K Nakano T Takechi H Satake J Uchida A Fujioka et al. (1996) ArticleTitleAntitumor activity of 1 M tegafur-0.4 M 5-chloro-2,4-dihydroxypyridine-1 M potassium oxonate (S-1) against human colon carcinoma orthotopically implanted into nude rats Cancer Res 56 2602–6 Occurrence Handle8653704 Occurrence Handle1:CAS:528:DyaK28XjtlCgurg%3D
T Shirasaka Y Shimamoto M Fukushima (1993) ArticleTitleInhibition by oxonic acid of gastrointestinal toxicity of 5-fluorouracil without loss of its antitumor activity in rats Cancer Res 53 4004–9 Occurrence Handle7689420 Occurrence Handle1:CAS:528:DyaK3sXmtFKqsrk%3D
T Takechi K Nakano J Uchida A Mita K Toko S Takeda et al. (1997) ArticleTitleAntitumor activity and low intestinal toxicity of S-1, a new formulation of oral tegafur, in experimental tumor models in rats Cancer Chemother Pharmacol 39 205–11 Occurrence Handle8996521 Occurrence Handle1:CAS:528:DyaK2sXis1ajsL4%3D
K Hirata N Horikoshi K Aibe M Okazaki R Denno K Sasaki et al. (1999) ArticleTitlePharmacokinetic study of S-1, a novel oral fluorouracil antitumor drug Clin Cancer Res 5 2000–5 Occurrence Handle10473078 Occurrence Handle1:STN:280:DyaK1MvgtVSmtQ%3D%3D
H Takiuchi JA Ajani (1998) ArticleTitleUracil-tegafur in gastric carcinoma: a comprehensive review J Clin Oncol 16 2877–85 Occurrence Handle9704742 Occurrence Handle1:CAS:528:DyaK1cXlsFCnsr0%3D
T Taguchi Y Inuyama R Kanamaru K Hasegawa S Akazawa H Niitani et al. (1997) ArticleTitlePhase I study of S-1 Jpn J Cancer Chemother 24 2253–64 Occurrence Handle1:STN:280:DyaK1c%2Fotl2rug%3D%3D
Y Sakata A Ohtsu N Horikoshi K Sugimachi Y Mitachi T Taguchi (1998) ArticleTitleLate phase II study of novel oral fluoropyrimidine anticancer drug S-1 (1 M tegafur–0.4 M gimestat–1 M otastat potassium) in advanced gastric cancer patients Eur J Cancer 34 1715–20 Occurrence Handle9893658 Occurrence Handle10.1016/S0959-8049(98)00211-1 Occurrence Handle1:CAS:528:DyaK1cXntlShurw%3D
K Sugimachi Y Maehara N Horikoshi Y Shimada Y Sakata Y Mitachi et al. (1999) ArticleTitleAn early phase II study of oral S-1, a newly developed 5-fluorouracil derivative for advanced and recurrent gastrointestinal cancers Oncology 57 202–10 Occurrence Handle10545788 Occurrence Handle10.1159/000012032 Occurrence Handle1:CAS:528:DyaK1MXmvF2msr0%3D
W Koizumi M Kurihara S Nakano K Hasegawa (2000) ArticleTitleThe S-1 Gastrointestinal Cancer Study Group. Phase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer Oncology 58 191–7 Occurrence Handle10765119 Occurrence Handle10.1159/000012099 Occurrence Handle1:CAS:528:DC%2BD3cXjvVeitr4%3D
A Ohtsu N Boku F Nagashima W Koizumi S Tanabe K Saigenji et al. (2001) ArticleTitleA phase I/II study of S-1 plus cisplatin (CDDP) in patients with advanced gastric cancer (abstract) Proc ASCO 20 165
Japan Society (1993) ArticleTitlefor Cancer Therapy. Criteria for the evaluation of the clinical effects of solid cancer chemotherapy J Jpn Soc Cancer Ther 28 101–30
InstitutionalAuthorNameThe Meta-Analysis Group in Cancer (1998) ArticleTitleEfficacy of intravenous continuous infusion of fluorouracil compared with bolus administration in advanced colorectal cancer J Clin Oncol 16 301–8
AM Lowy PF Mansfield SD Leach R Pazdur P Dumas JA Ajani (1999) ArticleTitleResponse to neoadjuvant chemotherapy best predicts survival after curative resection of gastric cancer Ann Surg 229 303–8 Occurrence Handle10077040 Occurrence Handle10.1097/00000658-199903000-00001 Occurrence Handle1:STN:280:DyaK1M7nsVKmug%3D%3D
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baba, H., Yamamoto, M., Endo, K. et al. Clinical efficacy of S-1 combined with cisplatin for advanced gastric cancer. Gastric Cancer 6 (Suppl 1), 45–49 (2003). https://doi.org/10.1007/s10120-003-0222-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10120-003-0222-y