Abstract
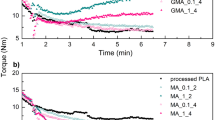
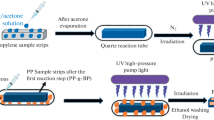
Poly(lactic acid) (PLA) is one of the most important bio-plastics, and chemical modification of the already-polymerized poly(lactic acid) chains may enable optimization of its material properties and expand its application areas. In this study, we demonstrated that poly(lactic acid) can be readily dissolved in acrylic acid at room temperature, and acrylic acid can be graft-polymerized onto poly(lactic acid) chains in solution with the help of photoinitiator benzophenone under 254 nm ultraviolet (UV) irradiation. Similar photo-grafting polymerization of acrylic acid (PAA) has only been studied before in the surface modification of polymer films. The graft ratio could be controlled by various reaction parameters, including irradiation time, benzophenone content, and monomer/polymer ratios. This photo-grafting reaction resulted in high graft ratio (graft ratio PAA/PLA up to 180%) without formation of homopolymers of acrylic acid. When the PAA/PLA graft ratio was higher than 100%, the resulting PLA-g-PAA polymer was found dispersible in water. The pros and cons of the photo-grafting reaction were also discussed.
Similar content being viewed by others
References
Di Lorenzo, M. L.; Androsch R. (eds.) Synthesis, structure and properties of poly(lactic acid). Springer, Switzerland, 2018, p. 1.
Aurus, R.; Lim, L.; Selke, S. E. M.; Tsuji, H. (eds.) Poly(lactic acid): Synthesis, structures, properties, processing, and Applications. Wiley, Canada, 2010, p. 141.
Krishnan, S.; Pandey, P.; Mohanty, S.; Nayak, S. K. Toughening of poly(lactic acid): An overview of research progress. Polym. Plastic. Tech. Eng.2016, 55, 1623–1652.
Ramot, Y.; Haim-Zada, M.; Domb, A. J.; Nyska, A. Biocompatibility and safety of PLA and its copolymers. Adv. Drug Deliver. Rev.2016, 107, 153–159.
Tyler, B.; Gullotti, D.; Mangraviti, A.; Utsuki, T.; Brem, H. Poly(lactic acid) (PLA) controlled delivery carriers for biomedical applications. Adv. Drug Deliver. Rev.2016, 107, 163–168.
Rasal, R. M.; Janorkar, A. V.; Hirt, D. E. Poly(lactic acid) modifications. Prog. Polym. Sci.2010, 35, 338-356.
Critechfield, F. E.; Koleske, J. V. US Patent 3, 760, 034, 1973.
Wu, C. Improving polylactide/starch biocomposites by grafting polylactide with acrylic acid—Characterization and biodegradability assessment. Macromol. Biosci.2005, 5, 352–361.
Dorman, G.; Nakamura, H.; Pulsipher, A.; Prestwich, G. D. The life of pi star: Exploring the exciting and forbidden worlds of the benzophenone photophore. Chem. Rev.2016, 116, 15284–15398.
Yang, W. T.; Ranby, B. Bulk surface photografting process and its applications. I. Reactions and kinetics. J. Appl. Polym. Sci.1996, 62, 533–543.
Li, Y.; DeSimone, J. M.; Poon, C.; Samulski, E. T. Photoinduced graft polymerization of styrene onto polypropylene substrates. J. Appl. Polym. Sci.1997, 64, 883–889.
Ranby, B.; Yang, W. T.; Tretinnikov, V.; Tokarev, V.; Xu, Y. H. Lamination of polymer films by bulk surface photografting process and properties. Chinese J. Polym. Sci.2001, 19, 123-127.
Yang, W. T.; Ranby, B. Bulk surface photografting process and its applications. II. Principal factors affecting surface photografting. J. Appl. Polym. Sci.1996, 62, 545–555.
Ma, H.; Davis, R. H.; Bowman, C. N. A novel sequential photoinduced living graft polymerization. Macromolecules2000, 33, 331–335.
Janorkar, A. V.; Metters, A. T.; Hirt, D. E. Modification of poly(lactic acid) films: Enhanced wettability from surface-confined photografting and increased degradation rate due to an artifact of the photografting process. Macromolecules2004, 37, 9151–9159.
Ikada, E. Photo- and bio-degradable polyesters, photodegradation behaviours of aliphatic polyesters. J. Photopolym. Sci. Tech.1997, 10, 265–269.
Tsuji, H.; Echizen, Y.; Nishimura, Y. Photodegradation of biodegradable polyesters: A comprehensive study on poly(Llactide) and poly(ε-caprolactone). Polym. Degrad. Stab.2006, 91, 1128–1137.
Li, L.; Raghupathi, K.; Song, C.; Prasad, P.; Thayumanavan, S. Selfassembly of random copolymers. Chem. Commun.2014, 50, 13417–13432.
Zhou, Y.; Liu, B.; Wang, X. Self-assembly of homopolymers through strong dipole-dipole interaction in their aqueous solutions. Polymer2016, 97, 1–10.
Choucair, A.; Lavigueur, C.; Eisenberg, A. Polystyrene-poly(acrylic acid) vesicle size control using solution properties and hydrophilic block length. Langmuir2004, 20, 3894–3900.
McNeill, I. C.; Sadeghi, S. M. T. Thermal-stability and degradation mechanisms of poly(acrylic acid) and its salts. I. Poly(acrylic acid). Polym. Degrad. Stab.1990, 29, 233.
Chen, L.; Hu, K.; Sun, S.; Jiang, H.; Huang, D.; Zhang, K.; Pan, L.; Li, Y. Toughening poly(lactic acid) with imidazolium-based elastomeric ionomers. Chinese J. Polym. Sci.2018, 36, 1342–1352.
Zhang, J.; Jia, J.; Kim, J. P.; Shen, H.; Yang, F.; Zhang, Q.; Xu, M.; Bi, W.; Wang, X.; Yang, J.; Wu, D. Ionic colloidal molding as a biomimetic scaffolding strategy for uniform bone tissue regeneration. Adv. Mater.2017, 29, 1605546–1605554.
Acknowledgments
The work was financially supported by the National Natural Science Foundation of China (No. 21434008). We thank Professor Lei Tao and his laboratory for the GPC characterization.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, JX., Huang, YB. & Yang, WT. Photo-grafting Poly(acrylic acid) onto Poly(lactic acid) Chains in Solution. Chin J Polym Sci 38, 137–142 (2020). https://doi.org/10.1007/s10118-019-2308-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-019-2308-y