Abstract
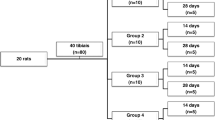
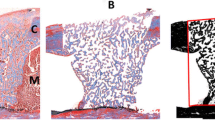
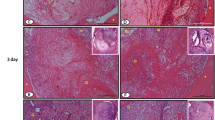
The goal of this study was to analyze the role of cyclo-oxygenase-2 following bone repair in rats submitted to low-level laser therapy. A total of 48 rats underwent surgery to inflict bone defects in their tibias having been randomly distributed into two groups: negative control and laser exposed group, i.e., the animals were treated with low-level laser therapy by means of gallium arsenide laser at 16 J/cm2. The animals were killed after 48 h, 7 days, 14 days, or 21 days. The tibias were removed for morphological, morphometric, and immunohistochemistry analysis for cyclo-oxygenase-2. Statistical significant differences (P < 0.05) were observed in the quality of bone repair and quantity of formed bone between groups 14 days after surgery in the laser exposed group. In the same way, cyclo-oxygenase-2 immunoreactivity was more intense in bone cells for intermediate periods evaluated in this group. Taken together, such results suggest that low-level laser therapy is able to improve bone repair in the tibia of rats after 14 days of surgery as a result of an up-regulation for cyclo-oxygenase-2 expression in bone cells.








Similar content being viewed by others
References
Hammond A (2004) Rehabilitation in rheumatoid arthritis: a critical review. Musculoskelet Care 2:135–151
Cetiner S, Kahraman SA, Yucetas S (2006) Evaluation of low-level laser therapy in the treatment of temporomandibular disorders. Photomed Laser Surg 24:637–641
Faria Amorim JC, Sousa GR, Silveira Lde B, Prates RA, Pinotti M, Ribeiro MS (2006) Clinical study of the gingiva healing after gingivectomy and low-level laser therapy. Photomed Laser Surg 24:588–594
Landthaler M, Hohenleutner U (2006) Laser therapy of vascular lesions. Photodermatol Photoimmunol Photomed 22:324–332
Turner J, Jode L (1999) Low level laser therapy. Prima Books, Stockholm
Kipshidge N, Nikolaychik V, Keelan MH (2001) Low-power helium:neon laser irradiation enhances production of vascular endothelial growth factor and promotes growth of endothelial cells in vitro. Lasers Surg Med 28:355–364
Chen WR, Liu H, Ritchey JW, Bartels KE, Lucroy MD, Nordquist RE (2002) Effect of different components of laser immunotherapy in treatment of metastatic tumors in rats. Cancer Res 62:4295–4299
Dube A, Bansal H, Gupta PK (2003) Modulation of macrophage structure and function by low level He-Ne laser irradiation. Photochem Photobiol Sci 2:851–855
Lan CC, Wu CS, Chiou MH, Hsieh PC, Yu HS (2006) Low-energy helium-neon laser induces locomotion of the immature melanoblasts and promotes melanogenesis of the more differentiated melanoblasts: recapitulation of vitiligo repigmentation in vitro. J Invest Dermatol 126:2119–2126
Bayat M, Vasheghani MM, Razavi N, Taheri S, Rakhshan M (2005) Effect of low-level laser therapy on the healing of second-degree burns in rats: a histological and microbiological study. J Photochem Photobiol B 78:171–177
Nissan J, Assif D, Gross MD, Yaffe A, Binderman I (2006) Effect of low intensity laser irradiation on surgically created bony defects in rats. J Oral Rehabil 33:619–924
da Silva RV, Camilli JA (2006) Repair of bone defects treated with autogenous bone graft and low-power laser. J Craniofac Surg 17:297–301
Heckman JD, Sarasohn-Kahn J (1997) The economics of treating tibia fractures. The cost of delayed unions. Bull Hosp Joint Dis 56:63–72
Nicola RA, Jorgetti V, Rigau J, Pacheco MT, dos Reis LM, Zângaro RA (2003) Effect of low-power GaAlAs laser (660 nm) on bone structure and cell activity: an experimental animal study. Lasers Med Sci 18:89–94
Lirani-Galvao AP, Jorgetti V, da Silva OL (2006) Comparative study of how low-level laser therapy and low-intensity pulsed ultrasound affect bone repair in rats. Photomed Laser Surg 24:735–740
Kargman S, Charleson S, Cartwright M, Frank J, Riendeau D, Mancini J, Evans J, O’Neill G (1996) Characterization of prostaglandin G/H synthase 1 and 2 in rat, dog, monkey, and human gastrointestinal tracts. Gastroenterology 111:445–454
Dempke W, Rie C, Grothey A, Schmoll HJ (2001) Cyclooxygenase-2: a novel target for cancer chemotherapy? J Cancer Res Clin Oncol 127:411–417
Miranda SR, Filho HN, Marques Padovan LE, Ribeiro DA, Nicolielo D, Matsumoto MA (2006) Use of platelet-rich plasma under autogenous onlay bone grafts. Clin Oral Implant Res 17:694–699
Gerbi ME, Pinheiro AL, Marzola C, Limeira Júnior Fde A, Ramalho LM, Ponzi EA, Soares AO, Carvalho LC, Lima HV, Gonçalves TO (2005) Assessment of bone repair associated with the use of organic bovine bone and membrane irradiated at 830 nm. Photomed Laser Surg 23:382–388
Khadra M, Kasem N, Haanaes HR, Ellingsen JE, Lyngstadaas SP (2004) Enhancement of bone formation in rat calvarial bone defects using low-level laser therapy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 97:693–700
Coombe AR, Ho CT, Darendeliler MA, Hunter N, Philips JR, Chapple CC, Yum LW (2001) The effects of low level laser irradiation on osteoblastic cells. Clin Orthod Res 4:3–14
Khadra M (2005) The effect of low level laser irradiation on implant-tissue interaction. In vivo and in vitro studies. Swed Dent J Suppl 172:1–63
Ozawa Y, Shimizu N, Kariya G, Abiko Y (1998) Low-energy laser irradiation stimulates bone nodule formation at early stages of cell culture in rat calvarial cells. Bone 22:347–354
Shibata M, Kodani I, Osaki M, Araki K, Adachi H, Ryoke K, Ito H (2005) Cyclo-oxygenase-1 and -2 expression in human oral mucosa, dysplasias and squamous cell carcinomas and their pathological significance. Oral Oncol 41:304–312
Tsujii M, Kawano S, Tsuji S, Sawaoka H, Hori M, DuBois RN (1998) Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell 93:705–716
Wang W, Bergh A, Damber JE (2007) Increased expression of CCAAT/enhancer-binding protein beta in proliferative inflammatory atrophy of the prostate: relation with the expression of COX-2, the androgen receptor, and presence of focal chronic inflammation. Prostate 67:1238–1246
Sato Y, Arai N, Negishi A, Ohya K (1997) Expression of cyclooxygenase genes and involvement of endogenous prostaglandin during osteogenesis in the rat tibial bone marrow cavity. J Med Dent Sci 44:81–92
Zhang X, Schwarz EM, Young DA, Puzas E, Rosier RN, O’Keefe RJ (2002) Cyclooxygenase-2 regulates mesenchymal cell differentiation into the osteoblast lineage and is critically involved in bone repair. J Clin Invest 109:1405–1415
Li J, Burr DB, Turner CH (2002) Suppression of prostaglandin synthesis with NS-398 has different effects on endocortical and periosteal bone formation induced by mechanical loading. Calcif Tissue Int 70:320–329
Forwood MR (1996) Inducible cyclo-oxygenase (COX-2) mediates the induction of bone formation by mechanical loading in vivo. J Bone Miner Res 11:1688–1693
Acknowledgments
The authors are grateful to Maira Cristina Rondina Couto for histology assistance and Wilson Aparecido Orcini for morphometry assistance. R V Ferino is a recipient of a Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) fellowship [Programa Institucional de Bolsas de Iniciação Científica (PIBIC)].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsumoto, M.A., Ferino, R.V., Monteleone, G.F. et al. Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med Sci 24, 195–201 (2009). https://doi.org/10.1007/s10103-008-0544-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-008-0544-4