Abstract
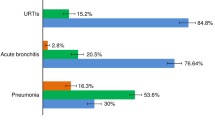
C-reactive protein (CRP) is a biomarker widely used for disease severity assessment and treatment of inflammatory conditions. Point-of-care testing (POCT) devices should ideally be rapid and provide similar results to standard tests done in laboratories. Two thousand nine hundred twenty-two serum samples were obtained from adult patients presenting to primary care with symptoms of lower respiratory infection in a European diagnostic study. The analytic performance of the CRP QuikRead POCT device (Orion Diagnostica) was evaluated by comparing results with a central laboratory method (Dimension Vista, Siemens), with both tests performed in a laboratory setting. For a CRP cut-off concentration of ≥ 30 mg/L, the QuikRead test had a sensitivity of 92.2%, and specificity of 99.4%. The mean difference between the QuikRead and the central lab test was 0.4 mg/L. The slope of the Passing-Bablok regression was 0.94 (95% CI 0.93–0.95) indicating an underestimation of CRP levels of 6% by QuikRead. CRP estimates obtained from the QuikRead test correlate well with a central laboratory assay and the measurement displays low inter-assay variation. Therefore, the QuikRead test is a good candidate for CRP testing in primary care.


Similar content being viewed by others
References
Vashist SK, Venkatesh AG, Marion Schneider E, Beaudoin C, Luppa PB, Luong JH (2016) Bioanalytical advances in assays for C-reactive protein. Biotechnol Adv 34(3):272–290. https://doi.org/10.1016/j.biotechadv.2015.12.010
Verbakel JY, Lemiengre MB, De Burghgraeve T, De Sutter A, Aertgeerts B, Shinkins B, Perera R, Mant D, Van den Bruel A, Buntinx F (2016) Should all acutely ill children in primary care be tested with point-of-care CRP: a cluster randomised trial. BMC Med 14(1):016–0679. https://doi.org/10.1186/s12916-2
Cals JW, Butler CC, Hopstaken RM, Hood K, Dinant GJ (2009) Effect of point of care testing for C reactive protein and training in communication skills on antibiotic use in lower respiratory tract infections: cluster randomised trial. BMJ 5 (338). Doi: 10.1136/bmj.b1374
Little P, Stuart B, Francis N, Douglas E, Tonkin-Crine S, Anthierens S, Cals JW, Melbye H, Santer M, Moore M, Coenen S, Butler C, Hood K, Kelly M, Godycki-Cwirko M, Mierzecki A, Torres A, Llor C, Davies M, Mullee M, O'Reilly G, van der Velden A, Geraghty AW, Goossens H, Verheij T, Yardley L (2013) Effects of internet-based training on antibiotic prescribing rates for acute respiratory-tract infections: a multinational, cluster, randomised, factorial, controlled trial. Lancet 382(9899):1175–1182. https://doi.org/10.1016/S0140-6736(13)60994-0
van Vugt SF, Broekhuizen BD, Lammens C, Zuithoff NP, de Jong PA, Coenen S, Ieven M, Butler CC, Goossens H, Little P, Verheij TJ (2013) Use of serum C reactive protein and procalcitonin concentrations in addition to symptoms and signs to predict pneumonia in patients presenting to primary care with acute cough: diagnostic study. BMJ 30 (346). Doi: 10.1136/bmj.f2450
Ieven M, Coenen S, Loens K, Lammens C, Coenjaerts F, Vanderstraeten A, Henriques-Normark B, Crook D, Huygen K, Butler CC, Verheij TJ, Little P, Zlateva K, van Loon A, Claas EC, Goossens H (2018) Aetiology of lower respiratory tract infection in adults in primary care: a prospective study in 11 European countries. Clin Microbiol Infect 12(18):30152–30156. https://doi.org/10.1016/j.cmi.2018.02.004
Teepe J, Broekhuizen BD, Loens K, Lammens C, Ieven M, Goossens H, Little P, Butler CC, Coenen S, Godycki-Cwirko M, Verheij TJ (2016) Predicting the presence of bacterial pathogens in the airways of primary care patients with acute cough. CMAJ 24:151364. https://doi.org/10.1503/cmaj.151364
Cals JW, Schot MJ, de Jong SA, Dinant GJ, Hopstaken RM (2010) Point-of-care C-reactive protein testing and antibiotic prescribing for respiratory tract infections: a randomized controlled trial. Ann Fam Med 8(2):124–133. https://doi.org/10.1370/afm.1090
Monteny M, ten Brinke MH, van Brakel J, de Rijke YB, Berger MY (2006) Point-of-care C-reactive protein testing in febrile children in general practice. Clin Chem Lab Med 44(12):1428–1432. https://doi.org/10.1515/CCLM.2006.270
Brouwer N, van Pelt J (2015) Validation and evaluation of eight commercially available point of care CRP methods. Clin Chim Acta 439:195–201. https://doi.org/10.1016/j.cca.2014.10.028
Esposito S, Tremolati E, Begliatti E, Bosis S, Gualtieri L, Principi N (2005) Evaluation of a rapid bedside test for the quantitative determination of C-reactive protein. Clin Chem Lab Med 43(4):438–440. https://doi.org/10.1515/CCLM.2005.077
Hernandez-Bou S, Trenchs V, Vanegas MI, Valls AF, Luaces C (2017) Evaluation of the bedside Quikread go(R) CRP test in the management of febrile infants at the emergency department. Eur J Clin Microbiol Infect Dis 36(7):1205–1211. https://doi.org/10.1007/s10096-017-2910-2
Zecca E, Barone G, Corsello M, Romagnoli C, Tiberi E, Tirone C, Vento G (2009) Reliability of two different bedside assays for C-reactive protein in newborn infants. Clin Chem Lab Med 47(9):1081–1084. https://doi.org/10.1515/CCLM.2009.246
Minnaard MC, van de Pol AC, Broekhuizen BD, Verheij TJ, Hopstaken RM, van Delft S, Kooijman-Buiting AM, de Groot JA, De Wit NJ (2013) Analytical performance, agreement and user-friendliness of five C-reactive protein point-of-care tests. Scand J Clin Lab Invest 73(8):627–634. https://doi.org/10.3109/00365513.2013.841985
Papaevangelou V, Papassotiriou I, Sakou I, Ferentinos G, Liapi G, Kyrka A, Konstantopoulos A (2006) Evaluation of a quick test for C-reactive protein in a pediatric emergency department. Scand J Clin Lab Invest 66(8):717–721. https://doi.org/10.1080/00365510600977869
Minnaard MC, van de Pol AC, Hopstaken RM, van Delft S, Broekhuizen BD, Verheij TJ, de Wit NJ (2016) C-reactive protein point-of-care testing and associated antibiotic prescribing. Fam Pract 33(4):408–413. https://doi.org/10.1093/fampra/cmw039
Funding
The study was part of the European Union FP6 funded Network of Excellence GRACE. Orion Diagnostics provided the QuikRead instruments and kits for this study. The study sponsors played no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Matheeussen, V., Van Hoof, V., Loens, K. et al. Analytical performance of a platform for point-of-care CRP testing in adults consulting for lower respiratory tract infection in primary care. Eur J Clin Microbiol Infect Dis 37, 1319–1323 (2018). https://doi.org/10.1007/s10096-018-3253-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-018-3253-3