Abstract
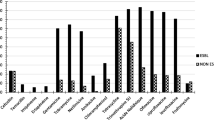
Nonfermenting Gram-negative bacilli (NF-GNB) are ubiquitous environmental opportunistic bacteria frequently misidentified by conventional phenotypic methods. The aim of this study was to determine the distribution of NF-GNB species by 16 S rRNA gene sequencing (used as reference method) and to compare performances of biochemical tests and matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF-MS). From nine French hospitals, 188 NF-GNB isolates (except P. aeruginosa and A. baumannii) were prospectively collected from 187 clinical samples between December 2008 and May 2009. By using the genotypic approach, 173 (92%) and 188 (100%) isolates were identified to the species and genus level, respectively. They covered 35 species and 20 genera, with a predominance of Stenotrophomonas maltophilia, Achromobacter xylosoxidans, and Pseudomonas putida group bacteria. Of the 173 species-level identified strains, concordant identification to the species-level was obtained for 75.1%, 83% and 88.9% of isolates with API 20 NE strip, the VITEK-2 (ID-GN card) system and MALDI-TOF-MS, respectively. By excluding S. maltophilia isolates accurately identified by the three methods, genus-level identification was much higher for MALDI-TOF-MS (92.9%), compared with API 20 NE and VITEK-2 (76.2% and 80.8%, respectively). In conclusion, MALDI-TOF-MS represents a rapid, inexpensive, and accurate tool for routine identification of NF-GNB in human clinical samples.
Similar content being viewed by others
References
Enoch DA, Birkett CI, Ludlam HA (2007) Non-fermentative Gram-negative bacteria. Int J Antimicrob Agents 29(Suppl 3):S33–41
McMenamin JD, Zaccone TM, Coenye T, Vandamme P, LiPuma JJ (2000) Misidentification of Burkholderia cepacia in US cystic fibrosis treatment centers: an analysis of 1,051 recent sputum isolates. Chest 117:1661–1665
Qin X, Emerson J, Stapp J, Stapp L, Abe P, Burns JL (2003) Use of real-time PCR with multiple targets to identify Pseudomonas aeruginosa and other nonfermenting Gram-negative bacilli from patients with cystic fibrosis. J Clin Microbiol 41:4312–4317
Ryan MP, Pembroke JT, Adley CC (2006) Ralstonia pickettii: a persistent gram-negative nosocomial infectious organism. J Hosp Infect 62:278–284
Bosshard PP, Zbinden R, Abels S, Boddinghaus B, Altwegg M, Bottger EC (2006) 16 S rRNA gene sequencing versus the API 20 NE system and the VITEK 2 ID-GNB card for identification of nonfermenting Gram-negative bacteria in the clinical laboratory. J Clin Microbiol 44:1359–1366
Wellinghausen N, Kothe J, Wirths B, Sigge A, Poppert S (2005) Superiority of molecular techniques for identification of gram-negative, oxidase-positive rods, including morphologically nontypical Pseudomonas aeruginosa, from patients with cystic fibrosis. J Clin Microbiol 43:4070–4075
Wallet F, Loiez C, Renaux E, Lemaitre N, Courcol RJ (2005) Performances of VITEK 2 colorimetric cards for identification of Gram-positive and Gram-negative bacteria. J Clin Microbiol 43:4402–4406
Zbinden A, Bottger EC, Bosshard PP, Zbinden R (2007) Evaluation of the colorimetric VITEK 2 card for identification of Gram-negative nonfermentative rods: comparison to 16 S rRNA gene sequencing. J Clin Microbiol 45:2270–2273
Ferroni A, Sermet-Gaudelus I, Abachin E, Quesne G, Lenoir G, Berche P, Gaillard JL (2002) Use of 16 S rRNA gene sequencing for identification of nonfermenting Gram-negative bacilli recovered from patients attending a single cystic fibrosis center. J Clin Microbiol 40:3793–3797
Cloud JL, Harmsen D, Iwen PC, Dunn JJ, Hall G, Lasala PR, Hoggan K, Wilson D, Woods GL, Mellmann A (2010) Comparison of traditional phenotypic identification methods with partial 5′ 16 S rRNA gene sequencing for species-level identification of nonfermenting Gram-negative bacilli. J Clin Microbiol 48:1442–1444
Seng P, Drancourt M, Gouriet F, La Scola B, Fournier PE, Rolain JM, Raoult D (2009) Ongoing revolution in bacteriology: routine identification of bacteria by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Clin Infect Dis 49:543–551
Bizzini A, Durussel C, Bille J, Greub G, Prod’hom G (2010) Performance of matrix-assisted laser desorption ionization-time of flight mass spectrometry for identification of bacterial strains routinely isolated in a clinical microbiology laboratory. J Clin Microbiol 48(5):1549–1554
Nagy E, Maier T, Urban E, Terhes G, Kostrzewa M (2009) Species identification of clinical isolates of Bacteroides by matrix-assisted laser-desorption/ionization time-of-flight mass spectrometry. Clin Microbiol Infect 15:796–802
Carbonnelle E, Beretti JL, Cottyn S, Quesne G, Berche P, Nassif X, Ferroni A (2007) Rapid identification of Staphylococci isolated in clinical microbiology laboratories by matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol 45:2156–2161
Pignone M, Greth KM, Cooper J, Emerson D, Tang J (2006) Identification of mycobacteria by matrix-assisted laser desorption ionization-time-of-flight mass spectrometry. J Clin Microbiol 44:1963–1970
Degand N, Carbonnelle E, Dauphin B, Beretti JL, Le Bourgeois M, Sermet-Gaudelus I, Segonds C, Berche P, Nassif X, Ferroni A (2008) Matrix-assisted laser desorption ionization-time of flight mass spectrometry for identification of nonfermenting Gram-negative bacilli isolated from cystic fibrosis patients. J Clin Microbiol 46:3361–3367
Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36:309–332
Lane D (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. John Wiley & Sons, Ltd, Chichester, United Kingdom, pp 115–175
Clarridge JE 3rd (2004) Impact of 16 S rRNA gene sequence analysis for identification of bacteria on clinical microbiology and infectious diseases. Clin Microbiol Rev 17:840–862
Martino R, Gomez L, Pericas R, Salazar R, Sola C, Sierra J, Garau J (2000) Bacteraemia caused by non-glucose-fermenting Gram-negative bacilli and Aeromonas species in patients with haematological malignancies and solid tumours. Eur J Clin Microbiol Infect Dis 19:320–323
Jugo J, Kennedy R, Crowe MJ, Lamrock G, McClurg RB, Rooney PJ, Morris TC, Johnston PG (2002) Trends in bacteraemia on the haematology and oncology units of a UK tertiary referral hospital. J Hosp Infect 50:48–55
Aumeran C, Paillard C, Robin F, Kanold J, Baud O, Bonnet R, Souweine B, Traore O (2007) Pseudomonas aeruginosa and Pseudomonas putida outbreak associated with contaminated water outlets in an oncohaematology paediatric unit. J Hosp Infect 65:47–53
Nseir S, Di Pompeo C, Brisson H, Dewavrin F, Tissier S, Diarra M, Boulo M, Durocher A (2006) Intensive care unit-acquired Stenotrophomonas maltophilia: incidence, risk factors, and outcome. Crit Care 10:R143
Saiman L, Siegel J (2004) Infection control in cystic fibrosis. Clin Microbiol Rev 17:57–71
Funke G, Funke-Kissling P (2004) Evaluation of the new VITEK 2 card for identification of clinically relevant gram-negative rods. J Clin Microbiol 42:4067–4071
Mellmann A, Cloud J, Maier T, Keckevoet U, Ramminger I, Iwen P, Dunn J, Hall G, Wilson D, Lasala P, Kostrzewa M, Harmsen D (2008) Evaluation of matrix-assisted laser desorption ionization-time-of-flight mass spectrometry in comparison to 16 S rRNA gene sequencing for species identification of nonfermenting bacteria. J Clin Microbiol 46:1946–1954
Mellmann A, Bimet F, Bizet C, Borovskaya AD, Drake RR, Eigner U, Fahr AM, He Y, Ilina EN, Kostrzewa M, Maier T, Mancinelli L, Moussaoui W, Prevost G, Putignani L, Seachord CL, Tang YW, Harmsen D (2009) High interlaboratory reproducibility of matrix-assisted laser desorption ionization-time of flight mass spectrometry-based species identification of nonfermenting bacteria. J Clin Microbiol 47:3732–3734
Minan A, Bosch A, Lasch P, Stammler M, Serra DO, Degrossi J, Gatti B, Vay C, D’Aquino M, Yantorno O, Naumann D (2009) Rapid identification of Burkholderia cepacia complex species including strains of the novel Taxon K, recovered from cystic fibrosis patients by intact cell MALDI-ToF mass spectrometry. Analyst 134:1138–1148
Acknowledgements
We thank Pr. X. Nassif for access to MALDI-TOF-MS and Andromas@Software. We are grateful to Michel Auzou (Caen), Brigitte Carzon (Versailles), and Brunhilde Dauphin (Andromas) for excellent technical assistance.
Transparency declaration
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors H. Jacquier and E. Carbonnelle are treated as first authors.
Rights and permissions
About this article
Cite this article
Jacquier, H., Carbonnelle, E., Corvec, S. et al. Revisited distribution of nonfermenting Gram-negative bacilli clinical isolates. Eur J Clin Microbiol Infect Dis 30, 1579–1586 (2011). https://doi.org/10.1007/s10096-011-1263-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-011-1263-5