Abstract
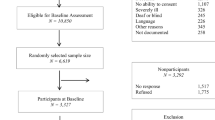
Cognitive Function Instrument (CFI) is a questionnaire aimed at detecting very early changes in cognitive and functional abilities and useful for monitoring cognitive decline in individuals without clinical impairment. The Italian version has been recently validated. The aim of the present study was to investigate the utility of the Italian version of CFI in tracking early cognitive changes in a cohort of healthy elderly subjects. A consecutive series of 257 cognitively healthy and functionally independent subjects, recruited either among relatives of patients attending our Memory Clinic or as volunteers after advertisement, underwent a baseline neuropsychological assessment. Of them, 157 subjects performed a 1-year follow-up assessment. All subjects completed the CFI, a short questionnaire composed of 14 items administered to both the subject and the referent (study-partner). Cognitive performance was assessed by Mini-Mental State Examination (MMSE) and the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS). At 1-year follow-up, Cronbach’s α was 0.79 (95% CI, 0.74–0.84) in self-report and 0.83 (95% CI, 0.79–0.87) for partner-report. CFI self-report correlated with MMSE (rS = − 0.22, p = 0.006) and RBANS (rS = − 0.23, p = 0.004). CFI partner-report showed negative correlation with MMSE (rS = − 0.17, p = 0.037) and RBANS (rS = − 0.20, p = 0.014). CFI 1-year follow-up score correlated with baseline both in self-report (rS = 0.56, p < 0.001) and partner-report (rS = 0.66, p < 0.001). Baseline CFI partner-report (p = 0.014) and CFI self+partner report (p = 0.023) were associated with RBANS total score less than 85 at 1-year follow-up, while only a trend was found considering baseline CFI self-report. Our results support the suitability of the Italian version of CFI for tracking cognitive changes along aging.

Similar content being viewed by others
References
Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH (2011) Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement 7(3):280–292
Dubois B, Hampel H, Feldman HH, Scheltens P, Aisen P, Andrieu S, Bakardjian H, Benali H, Bertram L, Blennow K, Broich K, Cavedo E, Crutch S, Dartigues JF, Duyckaerts C, Epelbaum S, Frisoni GB, Gauthier S, Genthon R, Gouw AA, Habert MO, Holtzman DM, Kivipelto M, Lista S, Molinuevo JL, O’Bryant SE, Rabinovici GD, Rowe C, Salloway S, Schneider LS, Sperling R, Teichmann M, Carrillo MC, Cummings J, Jack CR Jr (2016) Preclinical Alzheimer’s disease: definition, natural history, and diagnostic criteria. Alzheimer’s Dementia. 12(3):292–323
Jessen F, Amariglio RE, Van Boxtel M, Breteler M, Ceccaldi M, Chételat G et al (2014) A conceptual framework for research on subjective cognitive decline in preclinical Alzheimer’s disease. Alzheimer’s Dement 10(6):844–852
Amariglio RE, Becker JA, Carmasin J, Wadsworth LP, Lorius N, Sullivan C et al (2012) Subjective cognitive complaints and amyloid burden in cognitively normal older individuals. Neuropsychologia. Elsevier 50(12):2880–2886
Buckley RF, Maruff P, Ames D, Bourgeat P, Martins RN, Masters CL, Rainey-Smith S, Lautenschlager N, Rowe CC, Savage G, Villemagne VL, Ellis KA, AIBL study (2016) Subjective memory decline predicts greater rates of clinical progression in preclinical Alzheimer’s disease. Alzheimer’s Dement Elsevier Inc 12(7):796–804
Mitchell AJ, Beaumont H, Ferguson D, Yadegarfar M, Stubbs B (2014) Risk of dementia and mild cognitive impairment in older people with subjective memory complaints: meta-analysis. Acta Psychiatr Scand 130(6):439–451
Walsh SP, Raman R, Jones KB, Aisen PS (2006) ADCS prevention instrument project: the Mail-In Cognitive Function Screening Instrument (MCFSI). Alzheimer Dis Assoc Disord 20:S170–S178
Amariglio RE, Donohue MC, Marshall GA, Rentz DM, Salmon DP, Ferris SH, Karantzoulis S, Aisen PS, Sperling RA (2015) Tracking early decline in cognitive function in older individuals at risk for Alzheimer disease dementia. JAMA Neurol 72(4):446
Chipi E, Frattini G, Eusebi P, Mollica A, D’Andrea K, Russo M, Bernardelli A, Montanucci C, Luchetti E, Calabresi P, Parnetti L (2018) The Italian version of cognitive function instrument (CFI): reliability and validity in a cohort of healthy elderly. Neurol Sci 39(1):111–118
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12(3):189–198
Randolph C, Tierney MC, Mohr E, Chase TN (1998) The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): preliminary clinical validity. J Clin Exp Neuropsychol 20(3):310–319
Ponteri M, Pioli R, Padovani A, Tunesi SDGG (2007) RBANS repeatable battery for the assessment of neuropsychological status. Giunti
Petersen RC, Smith GE, Ivnik RJ, Kokmen E, Tangalos EG (1994) Memory function in very early Alzheimer’s disease. Neurology 44(5):867–867
Karantzoulis S, Novitski J, Gold M, Randolph C (2013) The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): utility in detection and characterization of mild cognitive impairment due to Alzheimer’s disease. Arch Clin Neuropsychol 28(8):837–844
England HB, Gillis MM, Hampstead BM (2014) RBANS memory indices are related to medial temporal lobe volumetrics in healthy older adults and those with mild cognitive impairment. Arch Clin Neuropsychol 29(4):322–328
Schmitt AL, Livingston RB, Smernoff EN, Reese EM, Hafer DG, Harris JB (2010) Factor analysis of the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) in a large sample of patients suspected of dementia. Appl Neuropsychol 17(1):8–17
Duff K, Humphreys Clark JD, O’Bryant SE, Mold JW, Schiffer RB, Sutker PB (2008) Utility of the RBANS in detecting cognitive impairment associated with Alzheimer’s disease: sensitivity, specificity, and positive and negative predictive powers. Arch Clin Neuropsychol 23(5):603–612
Solomon A, Kivipelto M, Molinuevo JL, Tom B, Ritchie CW (2018) European Prevention of Alzheimer’s Dementia Longitudinal Cohort Study (EPAD LCS): study protocol. BMJ Open 8(12):1–12
Jack CR, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB et al (2018) NIA-AA research framework: toward a biological definition of Alzheimer’s disease. Alzheimer’s Dement Elsevier Inc 14(4):535–62. 20
Faison WE, Schultz SK, Aerssens J, Alvidrez J, Anand R, Farrer LA, Jarvik L, Manly J, McRae T, Murphy GM, Olin JT, Regier D, Sano M, Mintzer JE (2007) Potential ethnic modifiers in the assessment and treatment of Alzheimer’s disease: challenges for the future. Int Psychogeriatr 19(3):539–558
Nuño MM, Gillen DL, Dosanjh KK, Brook J, Elashoff D, Ringman JM, Grill JD (2017) Attitudes toward clinical trials across the Alzheimer’s disease spectrum. Alzheimers Res Ther 9(1):81
Caselli RJ, Chen K, Locke DEC, Lee W, Roontiva A, Bandy D, Fleisher AS, Reiman EM (2014) Subjective cognitive decline: self and informant comparisons. Alzheimers Dement 10(1):93–98
Wolfsgruber S, Wagner M, Schmidtke K, Frölich L, Kurz A, Schulz S et al (2014) Memory concerns, memory performance and risk of dementia in patients with mild cognitive impairment. PLoS One 9(7):e100812
Gifford KA, Liu D, Lu Z, Tripodis Y, Cantwell NG, Palmisano J, Kowall N, Jefferson AL (2014) The source of cognitive complaints predicts diagnostic conversion differentially among nondemented older adults. Alzheimer’s Dement Elsevier Ltd 10(3):319–327
Glymour MM, Chêne G, Tzourio C, Dufouil C (2012) Brain MRI markers and dropout in a longitudinal study of cognitive aging. Neurology 79(13):1340–1348
Chatfield MD, Brayne CE, Matthews FE (2005) A systematic literature review of attrition between waves in longitudinal studies in the elderly shows a consistent pattern of dropout between differing studies. J Clin Epidemiol 58(1):13–19
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the local Ethics Committee (CEAS Umbria), and all participants signed the informed consent.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chipi, E., Montanucci, C., Eusebi, P. et al. The Italian version of Cognitive Function Instrument (CFI) for tracking changes in healthy elderly: results at 1-year follow-up. Neurol Sci 40, 2147–2153 (2019). https://doi.org/10.1007/s10072-019-03960-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-019-03960-x