Abstract
Background
Autologous hematopoietic stem-cell transplantation (AHSCT) has been utilized as a treatment option for multiple sclerosis (MS) since 1995. However, this procedure has not been widely implemented in clinical practice owing to its mortality risk. Here, we conduct a meta-analysis to evaluate the long-term efficacy and safety of AHSCT in MS treatment, aiming to optimize the benefit/risk ratio of this therapeutic strategy.
Methods
We searched the PubMed Web site and clinicaltrials.gov databases. The efficacy endpoints were progression-free survival (PFS) and disease activity-free survival. The safety outcomes were transplant-related mortality (TRM) and overall deaths.
Results
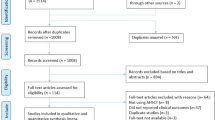
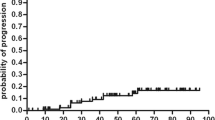
Eighteen eligible studies with a total of 732 participants were enrolled. The PFS was 75% (95% CI, 0.69–0.81), and the estimate of disease activity-free survival was 61% with 48-month follow-up. Subgroups analysis showed that low- and intermediate-intensity regimens were associated with higher PFS 80%. Relapsing remitting MS (RRMS) benefited more from AHSCT than other MS subtypes with PFS 85%. Patients with Gd+ lesions at baseline MRI responded better to AHSCT with PFS 77%. The estimate of TRM was 1.34% (95% CI, 0.39–2.30), and the overall mortality was 3.58%. TRM was significantly higher in high-intensity regimen studies (3.13%) and in older studies (1.93%) performed before 2006.
Conclusions
This meta-analysis provides evidences that AHSCT can induce long-term remissions for MS patients with a high degree of safety. We indicate low- and intermediate-intensity regimens and RRMS patients with the presence of Gd+ lesions at baseline MRI can obtain the optimal benefit/risk ratio from AHSCT.





Similar content being viewed by others
References
Wolinsky JS, Narayana PA, O'Connor P, Coyle PK, Ford C, Johnson K, Miller A, Pardo L, Kadosh S, Ladkani D, PROMiSe Trial Study Group (2007) Glatiramer acetate in primary progressive multiple sclerosis: results of a multinational, multicenter, double-blind, placebo-controlled trial. Ann Neurol 61(1):14–24. https://doi.org/10.1002/ana.21079
Wiendl H, Hohlfeld R (2009) Multiple sclerosis therapeutics: unexpected outcomes clouding undisputed successes. Neurology 72(11):1008–1015. https://doi.org/10.1212/01.wnl.0000344417.42972.54
Bittner S, Ruck T, Wiendl H, Grauer OM, Meuth SG (2017) Targeting B cells in relapsing-remitting multiple sclerosis: from pathophysiology to optimal clinical management. Ther Adv Neurol Disord 10(1):51–66. https://doi.org/10.1177/1756285616666741
von Budingen HC, Palanichamy A, Lehmann-Horn K et al (2015) Update on the autoimmune pathology of multiple sclerosis: B-cells as disease-drivers and therapeutic targets. Eur Neurol 73(3–4):238–246. https://doi.org/10.1159/000377675
Moreno Torres I, Garcia-Merino A (2017) Anti-CD20 monoclonal antibodies in multiple sclerosis. Expert Rev Neurother 17(4):359–371. https://doi.org/10.1080/14737175.2017.1245616
Hawker K, O'Connor P, Freedman MS, Calabresi PA, Antel J, Simon J, Hauser S, Waubant E, Vollmer T, Panitch H, Zhang J, Chin P, Smith CH, OLYMPUS trial group (2009) Rituximab in patients with primary progressive multiple sclerosis: results of a randomized double-blind placebo-controlled multicenter trial. Ann Neurol 66(4):460–471. https://doi.org/10.1002/ana.21867
Curro D, Mancardi G (2016) Autologous hematopoietic stem cell transplantation in multiple sclerosis: 20 years of experience. Neurol Sci 37(6):857–865. https://doi.org/10.1007/s10072-016-2564-3
Tyndall A, van Laar JM (2016) Stem cell transplantation and mesenchymal cells to treat autoimmune diseases. Presse Med 45(6 Pt 2):e159–e169. https://doi.org/10.1016/j.lpm.2016.05.002
Rebeiro P, Moore J (2016) The role of autologous haemopoietic stem cell transplantation in the treatment of autoimmune disorders. Intern Med J 46(1):17–28. https://doi.org/10.1111/imj.12944
Kelsey PJ, MC Oliveira MB et al (2016) Haematopoietic stem cell transplantation in autoimmune diseases: from basic science to clinical practice. Curr Res Transl Med 64(2):71–82. https://doi.org/10.1016/j.retram.2016.03.003
Mancardi G, Saccardi R (2008) Autologous haematopoietic stem-cell transplantation in multiple sclerosis. Lancet Neurol 7(7):626–636. https://doi.org/10.1016/S1474-4422(08)70138-8
Saccardi R, Kozak T, Bocelli-Tyndall C, Fassas A, Kazis A, Havrdova E, Carreras E, Saiz A, Löwenberg B, te Boekhorst PAW, Gualandi F, Openshaw H, Longo G, Pagliai F, Massacesi L, Deconink E, Ouyang J, Nagore FJZ, Besalduch J, Lisukov IA, Bonini A, Merelli E, Slavin S, Gratwohl A, Passweg J, Tyndall A, Steck AJ, Andolina M, Capobianco M, Martin JLD, Lugaresi A, Meucci G, Sáez RA, Clark RE, Fernandez MN, Fouillard L, Herstenstein B, Koza V, Cocco E, Baurmann H, Mancardi GL, Autoimmune Diseases Working Party of EBMT (2006) Autologous stem cell transplantation for progressive multiple sclerosis: update of the European Group for Blood and Marrow Transplantation autoimmune diseases working party database. Mult Scler 12(6):814–823. https://doi.org/10.1177/1352458506071301
Saiz A, Graus F (2002) Transplantation of haematopoietic stem cells in multiple sclerosis. Rev Neurol 35(12):1136–1141
Muraro PA, Martin R, Mancardi GL, Nicholas R, Sormani MP, Saccardi R (2017) Autologous haematopoietic stem cell transplantation for treatment of multiple sclerosis. Nat Rev Neurol 13(7):391–405. https://doi.org/10.1038/nrneurol.2017.81
Mancardi GL, MP Sormani FG et al (2015) Autologous hematopoietic stem cell transplantation in multiple sclerosis: a phase II trial. Neurology 84(10):981–988. https://doi.org/10.1212/WNL.0000000000001329
Casanova B, Jarque I, Gascón F, Hernández-Boluda JC, Pérez-Miralles F, de la Rubia J, Alcalá C, Sanz J, Mallada J, Cervelló A, Navarré A, Carcelén-Gadea M, Boscá I, Gil-Perotin S, Solano C, Sanz MA, Coret F (2017) Autologous hematopoietic stem cell transplantation in relapsing-remitting multiple sclerosis: comparison with secondary progressive multiple sclerosis. Neurol Sci 38(7):1213–1221. https://doi.org/10.1007/s10072-017-2933-6
Nash RA, Hutton GJ, Racke MK et al (2017) High-dose immunosuppressive therapy and autologous HCT for relapsing-remitting MS. Neurology 88(9):842–852. https://doi.org/10.1212/WNL.0000000000003660
Burt RK, Balabanov R, Han X, Sharrack B, Morgan A, Quigley K, Yaung K, Helenowski IB, Jovanovic B, Spahovic D, Arnautovic I, Lee DC, Benefield BC, Futterer S, Oliveira MC, Burman J (2015) Association of nonmyeloablative hematopoietic stem cell transplantation with neurological disability in patients with relapsing-remitting multiple sclerosis. JAMA 313(3):275–284. https://doi.org/10.1001/jama.2014.17986
Bowen JD, GH Kraft AW et al (2012) Autologous hematopoietic cell transplantation following high-dose immunosuppressive therapy for advanced multiple sclerosis: long-term results. Bone Marrow Transplant 47(7):946–951. https://doi.org/10.1038/bmt.2011.208
Atkins HL, Bowman M, Allan D, Anstee G, Arnold DL, Bar-Or A, Bence-Bruckler I, Birch P, Bredeson C, Chen J, Fergusson D, Halpenny M, Hamelin L, Huebsch L, Hutton B, Laneuville P, Lapierre Y, Lee H, Martin L, McDiarmid S, O'Connor P, Ramsay T, Sabloff M, Walker L, Freedman MS (2016) Immunoablation and autologous haemopoietic stem-cell transplantation for aggressive multiple sclerosis: a multicentre single-group phase 2 trial. Lancet 388(10044):576–585. https://doi.org/10.1016/S0140-6736(16)30169-6
Burman J, Iacobaeus E, Svenningsson A, Lycke J, Gunnarsson M, Nilsson P, Vrethem M, Fredrikson S, Martin C, Sandstedt A, Uggla B, Lenhoff S, Johansson JE, Isaksson C, Hägglund H, Carlson K, Fagius J (2014) Autologous haematopoietic stem cell transplantation for aggressive multiple sclerosis: the Swedish experience. J Neurol Neurosurg Psychiatry 85(10):1116–1121. https://doi.org/10.1136/jnnp-2013-307207
Fassas A, Anagnostopoulos A, Kazis A, Kapinas K, Sakellari I, Kimiskidis V, Smias C, Eleftheriadis N, Tsimourtou V (2000) Autologous stem cell transplantation in progressive multiple sclerosis—an interim analysis of efficacy. J Clin Immunol 20(1):24–30
Hamerschlak N, Rodrigues M, Moraes DA, Oliveira MC, Stracieri ABPL, Pieroni F, Barros GMN, Madeira MIA, Simões BP, Barreira AA, Brum DG, Ribeiro AAF, Kutner JM, Tylberi CP, Porto PP, Santana CL, Neto JZ, Barros JC, Paes AT, Burt RK, Oliveira EA, Mastropietro AP, Santos AC, Voltarelli JC (2010) Brazilian experience with two conditioning regimens in patients with multiple sclerosis: BEAM/horse ATG and CY/rabbit ATG. Bone Marrow Transplant 45(2):239–248. https://doi.org/10.1038/bmt.2009.127
Krasulova E, Trneny M, Kozak T et al (2010) High-dose immunoablation with autologous haematopoietic stem cell transplantation in aggressive multiple sclerosis: a single centre 10-year experience. Mult Scler 16(6):685–693. https://doi.org/10.1177/1352458510364538
Mancardi GL, MP Sormani MDG et al (2012) Autologous haematopoietic stem cell transplantation with an intermediate intensity conditioning regimen in multiple sclerosis: the Italian multi-centre experience. Mult Scler 18(6):835–842. https://doi.org/10.1177/1352458511429320
Nash RA, Bowen JD, McSweeney PA et al (2003) High-dose immunosuppressive therapy and autologous peripheral blood stem cell transplantation for severe multiple sclerosis. Blood 102(7):2364–2372. https://doi.org/10.1182/blood-2002-12-3908
Ni XS, Ouyang J, Zhu WH, Wang C, Chen B (2006) Autologous hematopoietic stem cell transplantation for progressive multiple sclerosis: report of efficacy and safety at three yr of follow up in 21 patients. Clin Transpl 20(4):485–489. https://doi.org/10.1111/j.1399-0012.2006.00510.x
Saccardi R, GL Mancardi AS et al (2005) Autologous HSCT for severe progressive multiple sclerosis in a multicenter trial: impact on disease activity and quality of life. Blood 105(6):2601–2607. https://doi.org/10.1182/blood-2004-08-3205
Saiz A, Blanco Y, Carreras E, Berenguer J, Rovira M, Pujol T, Marin P, Arbizu T, Graus F (2004) Clinical and MRI outcome after autologous hematopoietic stem cell transplantation in MS. Neurology 62(2):282–284
Shevchenko JL, Kuznetsov AN, Ionova TI, Melnichenko VY, Fedorenko DA, Kurbatova KA, Gorodokin GI, Novik AA (2015) Long-term outcomes of autologous hematopoietic stem cell transplantation with reduced-intensity conditioning in multiple sclerosis: physician's and patient's perspectives. Ann Hematol 94(7):1149–1157. https://doi.org/10.1007/s00277-015-2337-8
Xu J, BX Ji LS et al (2011) Clinical outcome of autologous peripheral blood stem cell transplantation in opticospinal and conventional forms of secondary progressive multiple sclerosis in a Chinese population. Ann Hematol 90(3):343–348. https://doi.org/10.1007/s00277-010-1071-5
Chen B, Zhou M, Ouyang J, Zhou R, Xu J, Zhang Q, Yang Y, Xu Y, Shao X, Meng L, Wang J, Xu Y, Ni X, Zhang X (2012) Long-term efficacy of autologous haematopoietic stem cell transplantation in multiple sclerosis at a single institution in China. Neurol Sci 33(4):881–886. https://doi.org/10.1007/s10072-011-0859-y
Burt RK, BA Cohen ER et al (2003) Hematopoietic stem cell transplantation for progressive multiple sclerosis: failure of a total body irradiation-based conditioning regimen to prevent disease progression in patients with high disability scores. Blood 102(7):2373–2378. https://doi.org/10.1182/blood-2003-03-0877
Sormani MP, PA Muraro IS et al (2017) Autologous hematopoietic stem cell transplantation in multiple sclerosis: a meta-analysis. Neurology 88(22):2115–2122. https://doi.org/10.1212/WNL.0000000000003987
Reston JT, Uhl S, Treadwell JR, Nash RA, Schoelles K (2011) Autologous hematopoietic cell transplantation for multiple sclerosis: a systematic review. Mult Scler 17(2):204–213. https://doi.org/10.1177/1352458510383609
Muraro PA, Pasquini M, Atkins HL, Bowen JD, Farge D, Fassas A, Freedman MS, Georges GE, Gualandi F, Hamerschlak N, Havrdova E, Kimiskidis VK, Kozak T, Mancardi GL, Massacesi L, Moraes DA, Nash RA, Pavletic S, Ouyang J, Rovira M, Saiz A, Simoes B, Trnený M, Zhu L, Badoglio M, Zhong X, Sormani MP, Saccardi R, for the Multiple Sclerosis–Autologous Hematopoietic Stem Cell Transplantation (MS-AHSCT) Long-term Outcomes Study Group (2017) Long-term outcomes after autologous hematopoietic stem cell transplantation for multiple sclerosis. JAMA Neurol 74(4):459–469. https://doi.org/10.1001/jamaneurol.2016.5867
Ontaneda D, Thompson AJ, Fox RJ et al (2017) Progressive multiple sclerosis: prospects for disease therapy, repair, and restoration of function. Lancet 389(10076):1357–1366. https://doi.org/10.1016/S0140-6736(16)31320-4
Shevchenko JL, Kuznetsov AN, Ionova TI, Melnichenko VY, Fedorenko DA, Kartashov AV, Kurbatova KA, Gorodokin GI, Novik AA (2012) Autologous hematopoietic stem cell transplantation with reduced-intensity conditioning in multiple sclerosis. Exp Hematol 40(11):892–898. https://doi.org/10.1016/j.exphem.2012.07.003
Funding
This work is financially supported by the National Natural Science Foundation of China (grant 81671233).
Author information
Authors and Affiliations
Contributions
C.T and G.F.F performed the study selection, data extraction, statistical analysis, and drafted the manuscript. All authors participated in study design, revised the protocol, contributed to interpretation of the results, critically revised the manuscript for important intellectual content, and read and approved the final version of this manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Figure A1
Forest plot for disease activity-free survival in each study and pooled estimates (PNG 146 kb)
High resolution image
(TIF 23638 kb)
Figure A2
Forest plot for overall mortality in each study and pooled estimates (PNG 283 kb)
High resolution image
(TIF 28961 kb)
Rights and permissions
About this article
Cite this article
Ge, F., Lin, H., Li, Z. et al. Efficacy and safety of autologous hematopoietic stem-cell transplantation in multiple sclerosis: a systematic review and meta-analysis. Neurol Sci 40, 479–487 (2019). https://doi.org/10.1007/s10072-018-3670-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-018-3670-1