Abstract
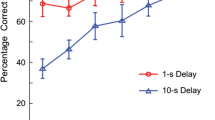
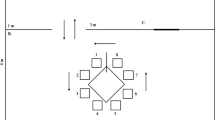
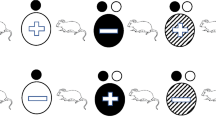
Delayed matching-to-sample (dMTS) is commonly used to study working memory (WM) processes in non-humans. Previous procedures for studying dog WM, including versions of the dMTS, did not separate the impact of delay and interference on memory performance. These studies were also limited to auditory and spatial stimuli, neglecting dogs' dominant sensory modality (i.e., olfaction). Therefore, we designed the first olfactory dMTS in dogs, with systematically varied delays and number of odors in a session, to dissociate the effects of delay and within-session proactive interference on dog WM. Dogs (n = 5) initially trained on matching-to-sample with 48 odors, with a zero-second delay, were tested on four delay lengths (0, 30, 60, and 90 s), counterbalanced across three, trial-unique, sessions. Although there was a slight decrease in accuracy across delays, dogs performed above chance on delays up to 60 s, suggesting a WM duration of at least 60 s. To explore the effect of within-session proactive interference on WM duration, the size of the stimulus set was reduced to six and two odors. There was no effect on the memory function with six odors compared to the trial-unique sessions. However, the interference caused by the two-odor set was enough to decrease accuracy at each delay length. These findings suggest that forgetting in dog working memory for odors can be simultaneously influenced by delay and within-session proactive interference.


Similar content being viewed by others
References
Adams B, Chan A, Callahan H, Milgram NW (2000a) The canine as a model of human cognitive aging: recent developments. Prog Neuro-Psychopharmacol Biol Psychiatry 24(5):675–692. https://doi.org/10.1016/s0278-5846(00)00101-9
Adams B, Chan A, Callahan H, Siwak C, Tapp D, Ikeda-Douglas C, Atkinson P, Head E, Cotman CW, Milgram NW (2000b) Use of a delayed non-matching to position task to model age-dependent cognitive decline in the dog. Behav Brain Res 108(1):47–56. https://doi.org/10.1016/S0166-4328(99)00132-1
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Chan ADF, Nippak PMD, Murphey H, Ikeda-Douglas CJ, Muggenburg B, Head E, Cotman CW, Milgram NW (2002) Visuospatial impairments in aged canines (Canis familiaris): the role of cognitive-behavioral flexibility. Behav Neurosci 116(3):443–454. https://doi.org/10.1037/0735-7044.116.3.443
Fiset S, Beaulieu C, Landry F (2003) Duration of dogs’ (Canis familiaris) working memory in search for disappearing objects. Anim Cogn 6(1):1–10. https://doi.org/10.1007/s10071-002-0157-4
Gadbois S, Reeve C (2014) Canine olfaction: scent, sign, and situation. Domestic dog cognition and behavior. Springer, Berlin, Heidelberg, pp 3–29
Gagnon S, Doré FY (1994) Cross-sectional study of object permanence in domestic puppies (Canis familiaris). J Comp Psychol 108(3):220–232. https://doi.org/10.1037/0735-7036.108.3.220
Hayes JE, McGreevy PD, Forbes SL, Laing G, Stuetz RM (2018) Critical review of dog detection and the influences of physiology, training, and analytical methodologies. Talanta 185:499–512
Head E, Mehta R, Hartley J, Kameka M, Cummings BJ, Cotman CW, Ruehl WW, Milgram NW (1995) Spatial learning and memory as a function of age in the dog. Behav Neurosci 109(5):851–858. https://doi.org/10.1037/0735-7044.109.5.851
Honig WK (1978) Studies of working memory in the pigeon. Cogn Process Anim Behav 211:248
Iversen IH (1993) Acquisition of matching-to-sample performance in rats using visual stimuli on nose keys. J Exp Anal Behav 59(3):471–482. https://doi.org/10.1901/jeab.1993.59-471
Iversen I (1997) Matching-to-sample performance in rats: a case of mistaken identity? J Exp Anal Behav 68(1):27–45. https://doi.org/10.1901/jeab.1997.68-27
Krichbaum S, Rogers B, Cox E, Waggoner LP, Katz JS (2020) Odor span task in dogs (Canis familiaris). Anim Cogn. https://doi.org/10.1007/s10071-020-01362-7
Kuœmierek P, Kowalska DM (2002) Effect of sound source position on learning and performance of auditory delayed matching-to-sample task in dogs. Acta Neurobiol Exp 62(4):251–262
Lazarowski L, Davila A, Krichbaum S, Cox E, Smith GJ, Waggoner LP, Katz J (2021) Olfactory matching-to-sample abstract-concept learning by dogs (Canis familiaris). J Exp Psychol Anim Learn Cogn. https://doi.org/10.1037/xan0000281
Lind J, Enquist M, Ghirlanda S (2015) Animal memory: a review of delayed matching-to-sample data. Behav Proc 117:52–58. https://doi.org/10.1016/j.beproc.2014.11.019
Martucci K, Swindell S (2014) How smart is your dog? An experimental analysis of canine learning abilities (Unpublished master’s thesis). Washington State University, Pullman, WA. https://research.libraries.wsu.edu/xmlui/bitstream/handle/2376/5333/Martucci thesis.pdf?sequence=1
Miklósi Á (2015) Dog behaviour, evolution, and cognition. Oxford University Press, Oxford
Milgram NW, Head E, Weiner E, Thomas E (1994) Cognitive functions and aging in the dog: acquisition of nonspatial visual tasks. Behav Neurosci 108(1):57–68. https://doi.org/10.1037//0735-7044.108.1.57
Olton DS, Samuelson RJ (1976) Remembrance of places passed: spatial memory in rats. J Exp Psychol Anim Behav Process 2(2):97–116. https://doi.org/10.1037/0097-7403.2.2.97
Overman WH, Doty RW (1980) Prolonged visual memory in macaques and man. Neuroscience 5(11):1825–1831. https://doi.org/10.1016/0306-4522(80)90032-9
Peña T, Pitts RC, Galizio M (2006) Identity matching-to-sample with olfactory stimuli in rats. J Exp Anal Behav 85(2):203–221. https://doi.org/10.1901/jeab.2006.111-04
Shettleworth SJ (2009) Cognition, evolution, and behavior. Oxford University Press, Oxford
Tapp PD, Siwak CT, Estrada J, Holowachuk D, Milgram NW (2003) Effects of age on measures of complex working memory span in the beagle dog (Canis familiaris) using two versions of a spatial list learning paradigm. Learn Mem 10(2):148–160. https://doi.org/10.1101/lm.56503
Wright AA (2012) Memory processing. In: Zentall TR, Wasserman EA (eds) The Oxford handbook of comparative cognition. Oxford University Press, Oxford, pp 239–260
Wright AA, Kelly DM, Katz JS (2018) Comparing cognition by integrating concept learning, proactive interference, and list memory. Learn Behav 46(2):107–123
Zanghi BM, Araujo J, Milgram NW (2015) Cognitive domains in the dog: independence of working memory from object learning, selective attention, and motor learning. Anim Cogn 18(3):789–800. https://doi.org/10.1007/s10071-015-0847-3
Acknowledgements
We thank the staff at Auburn University’s Canine Performance Sciences and the Auburn University College of Veterinary Medicine Department of Lab Animal Health for providing logistical support and care of the dogs in the study. We also thank Brook Caudill, Jordan Farrell, Anna Beth Freeman, Kierra Kuhlman and Lindsey Rogers for assistance with data collection and Auburn University Department of Psychological Sciences for funding the study.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest..
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The IACUC number was 2018–3334.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Krichbaum, S., Lazarowski, L., Davila, A. et al. Dissociating the effects of delay and interference on dog (Canis familiaris) working memory. Anim Cogn 24, 1259–1265 (2021). https://doi.org/10.1007/s10071-021-01509-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-021-01509-0