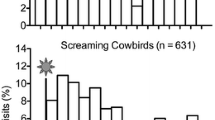
Abstract
Shiny and screaming cowbirds are avian interspecific brood parasites that locate and prospect host nests in daylight and return from one to several days later to lay an egg during the pre-dawn twilight. Thus, during nest location and prospecting, both location information and visual features are available, but the latter become less salient in the low-light conditions when the nests are visited for laying. This raises the question of how these different sources of information interact, and whether this reflects different behavioural specializations across sexes. Differences are expected, because in shiny cowbirds, females act alone, but in screaming cowbirds, both sexes make exploratory and laying nest visits together. We trained females and males of shiny and screaming cowbird to locate a food source signalled by both colour and position (cues associated), and evaluated performance after displacing the colour cue to make it misleading (cues dissociated). There were no sex or species differences in acquisition performance while the cues were associated. When the colour cue was relocated, individuals of both sexes and species located the food source making fewer visits to non-baited wells than expected by chance, indicating that they all retained the position as an informative cue. In this phase, however, shiny cowbird females, but not screaming, outperformed conspecific males, visiting fewer non-baited wells before finding the food location and making straighter paths in the search. These results are consistent with a greater reliance on spatial memory, as expected from the shiny cowbird female’s specialization on nest location behaviour.



Similar content being viewed by others
Data availability
Data are available from Figshare Digital Repository, https://doi.org/10.6084/m9.figshare.12152316.
Code availability
The R scripts generated for analysis presented in this study are available from the corresponding author on request.
References
Astié AA, Kacelnik A, Reboreda JC (1998) Sexual differences in memory in shiny cowbirds. Anim Cogn 1:77–82. https://doi.org/10.1007/s100710050011
Astié AA, Scardamaglia RC, Muzio RN, Reboreda JC (2015) Sex differences in retention after a visual or a spatial discrimination learning task in brood parasitic shiny cowbirds. Behav Process 119:99–104. https://doi.org/10.1016/j.beproc.2015.07.016
Benhamou S (2004) How to reliably estimate the tortuosity of an animal’s path: straightness, sinuosity, or fractal dimension? J Theor Biol 229:209–220. https://doi.org/10.1016/j.jtbi.2004.03.016
Bettis TJ, Jacobs LF (2013) Sex differences in memory for landmark arrays in C57BL/J6 mice. Anim Cogn 16:873–882. https://doi.org/10.1007/s10071-013-0619-x
Brodbeck DR (1994) Memory for spatial and local cues: a comparison of a storing and a nonstoring species. Anim Learn Behav 22:119–133. https://doi.org/10.3758/BF03199912
Brodbeck DR, Shettleworth SJ (1995) Matching location and color of a compound stimulus: comparison of a food-storing and a nonstoring bird species. J Exp Psychol Anim Behav Process 21:64–77. https://doi.org/10.1037/0097-7403.21.1.64
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Maechler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://journal.r-project.org/archive/2017/RJ-2017-066/index.html
Clayton NS, Krebs JR (1994) Memory for spatial and object-specific cues in food-storing and non-storing birds. J Comp Physiol A 174:371–379. https://doi.org/10.1007/BF00240218
Clayton NS, Kacelnik A, Reboreda JC (1997) Seasonal changes in hippocampus size in parasitic cowbirds. Behav Process 41:237–243. https://doi.org/10.1016/S0376-6357(97)00050-8
Coluccia E, Louse G (2004) Gender differences in spatial orientation: a review. J Environ Psychol 24:329–340. https://doi.org/10.1016/j.jenvp.2004.08.006
Davies HB, White DJ (2018) Specializations in cognition generalize across contexts: cowbirds are consistent in nest prospecting and foraging tasks. Anim Behav 144:1–7. https://doi.org/10.1016/j.anbehav.2018.07.019
De Mársico MC, Reboreda JC (2008) Egg-laying behaviour in screaming cowbirds. Why does a specialist brood parasite waste so many eggs? Condor 110:143–153. https://doi.org/10.1525/cond.2008.110.1.143
Feenders G, Smulders TV (2011) Magpies can use local cues to retrieve their food caches. Anim Cogn 14:235–243. https://doi.org/10.1007/s10071-010-0357-2
Galea LAM, Kavaliers M, Ossenkopp KP (1996) Sexually dimorphic spatial learning in meadow voles Microtus pennsylvanicus and deer mice Peromyscus maniculatus. J Exp Biol 199:195–200
Gaulin SJC, FitzGerald RW (1986) Sex differences in spatial ability: an evolutionary hypothesis and test. Am Nat 127:74–88. https://doi.org/10.1086/284468
Gaulin SJC, FitzGerald RW (1989) Sexual selection for spatial-learning ability. Anim Behav 37:322–331. https://doi.org/10.1016/0003-3472(89)90121-8
Gaulin SJC, FitzGerald RW, Wartell MS (1990) Sex differences in spatial ability and activity in two vole species (Microtus ochrogaster and M. pennsylvanicus). J Comp Psychol 104:88–93. https://doi.org/10.1037/0735-7036.104.1.88
Gloag R, Fiorini VD, Reboreda JC, Kacelnik A (2013) The wages of violence: mobbing by mockingbirds as a frontline defence against brood-parasitic cowbirds. Anim Behav 86:1023–1029. https://doi.org/10.1016/j.anbehav.2013.09.007
Gould KL, Kelly DM, Kamil AC (2010) What scatter-hoarding animals have taught us about small-scale navigation. Phil Trans R Soc B 365:901–914. https://doi.org/10.1098/rstb.2009.0214
Gray ER, Bloomfield LL, Ferrey A, Spetch ML, Sturdy CB (2005) Spatial encoding in mountain chickadees: features overshadow geometry. Biol Lett 1:314–317. https://doi.org/10.1098/rsbl.2005.0347
Guigueno MF, Sherry DF (2017) Hippocampus and spatial memory in brood parasitic cowbirds. In: Soler M (ed) Avian brood parasitism. Springer, Cham, pp 203–218. https://doi.org/10.1007/978-3-319-73138-4_11
Guigueno MF, Snow DA, MacDougall-Shackleton SA, Sherry DF (2014) Female cowbirds have more accurate spatial memory than males. Biol Lett 10:20140026. https://doi.org/10.1098/rsbl.2014.0026
Guigueno MF, MacDougall-Shackleton SA, Sherry DF (2015) Sex differences in spatial memory in brown-headed cowbirds: males outperform females on a touchscreen task. PLoS ONE 10(6):e0128302. https://doi.org/10.1371/journal.pone.0128302
Guigueno MF, MacDougall-Shackleton SA, Sherry DF (2016) Sex and seasonal differences in hippocampal volume and neurogenesis in brood-parasitic brown-headed cowbirds (Molothrus ater). Dev Neurobiol 76:1275–1290. https://doi.org/10.1002/dneu.22421
Hahn D, Sedgwick J, Painter I, Casna N (1999) A spatial and genetic analysis of cowbird host selection. Stud Avian Biol 18:204–217
Hartig F (2020) DHARMa: residual diagnostics for hierarchical (multi-level/mixed) regression models. v. 0.3.1. https://florianhartig.github.io/DHARMa
Healy SE (1995) Memory for objects and positions: delayed non-matching-to-sample in storing and non-storing tits. Q J Exp Psychol B 48:179–191. https://doi.org/10.1080/14640749508401446
Healy SD, Krebs JR (1992a) Comparing spatial memory in two species of tit: recalling a single positive location. Anim Learn Behav 20:121–126. https://doi.org/10.3758/BF03200409
Healy SD, Krebs JR (1992b) Delayed-Matching-to-Sample by marsh tits and great tits. Q J Exp Psychol B 45:33–47. https://doi.org/10.1080/14640749208401023
Hodgson ZG, Healy SD (2005) Preference for spatial cues in a non-storing songbird species. Anim Cogn 8:211–214. https://doi.org/10.1007/s10071-004-0249-4
Hornsby MAW, Hurly TA, Hamilton CE, Pritchard DJ, Healy DE (2014) Wild, free-living rufous hummingbirds do not use geometric cues in a spatial task. Behav Process 108:138–141. https://doi.org/10.1016/j.beproc.2014.10.003
Hurly TA, Healy SD (1996) Memory for flowers in rufous hummingbirds: location or local visual cues? Anim Behav 51:1149–1157. https://doi.org/10.1006/anbe.1996.0116
Hurly TA, Healy SD (2002) Cue learning by rufous hummingbirds (Selasphorus rufus). J Exp Psychol Anim Behav Process 28:209–223. https://doi.org/10.1037//0097-7403.28.2.209
Hurly TA, Fox TAO, Zwueste DM, Healy SD (2014) Wild hummingbirds rely on landmarks not geometry when learning an array of flowers. Anim Cogn 17:1157–1165. https://doi.org/10.1007/s10071-014-0748-x
Kelly DM (2010) Features enhance the encoding of geometry. Anim Cogn 13:453–462. https://doi.org/10.1007/s10071-009-0296-y
LaDage LD, Roth TC II, Fox RA, Pravosudov VV (2009) Flexible cue use in food-caching birds. Anim Cogn 12:419–426. https://doi.org/10.1007/s10071-008-0201-0
Mayer U, Bischof HJ (2012) Brain activation pattern depends on the strategy chosen by zebra finches to solve an orientation task. J Exp Biol 215:426–434. https://doi.org/10.1242/jeb.063941
Morandi-Raikova A, Vallortigara G, Mayer U (2020) The use of spatial and local cues for orientation in domestic chicks (Gallus gallus). Anim Cogn 23:367–387. https://doi.org/10.1007/s10071-019-01342-6
Nair-Roberts RG, Erichsen JT, Reboreda JC, Kacelnik A (2006) Distribution of substance P reveals a novel subdivision in the hippocampus of parasitic South American cowbirds. J Comp Neurol 496:610–626. https://doi.org/10.1002/cne.20915
O'Keefe J, Nadel L (1978) The hippocampus as a cognitive map. Oxford University Press, Oxford
Pearce JM, Graham M, Good MA, Jones PM, McGregor A (2006) Potentiation, overshadowing, and blocking of spatial learning based on the shape of the environment. J Exp Psychol Anim Behav Process 32:201–214. https://doi.org/10.1037/0097-7403.32.3.201
Peer BD, Sealy SG (1999) Laying time of the bronzed cowbird. Wilson Bull 111:137–139
Pritchard DJ, Hurly TA, Healy SD (2015) Effects of landmark distance and stability on accuracy of reward relocation. Anim Cogn 18:1285–1297. https://doi.org/10.1007/s10071-015-0896-7
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org
Reboreda JC, Clayton N, Kacelnik A (1996) Species and sex differences in hippocampus size in parasitic and non-parasitic cowbirds. NeuroReport 7:505–508. https://doi.org/10.1097/00001756-199601310-00031
Rothstein SI, Yokel DA, Fleischer RC (1986) Social dominance, mating and spacing systems, female fecundity, and vocal dialects in captive and free-ranging brown-headed cowbirds. In: Johnston RF (ed) Current ornithology. Plenum, New York, pp 127–185. https://doi.org/10.1007/978-1-4615-6784-4_3
Scardamaglia RC, Reboreda JC (2014) Ranging behavior of female and male shiny and screaming cowbirds while searching for host nests. Auk 131:610–618. https://doi.org/10.1642/AUK-14-54.1
Scardamaglia RC, Fiorini VD, Kacelnik A, Reboreda JC (2017) Planning host exploitation through prospecting visits by parasitic cowbirds. Behav Ecol Sociobiol 71:23. https://doi.org/10.1007/s00265-016-2250-8
Scardamaglia RC, Kacelnik A, Reboreda JC (2018) Temporal and spatial patterns of roosting behavior by brood parasitic cowbirds. Ibis 160:779–789. https://doi.org/10.1111/ibi.12587
Sherry DF (2006) Neuroecology. Annu Rev Psychol 57:167–197. https://doi.org/10.1146/annurev.psych.56.091103.070324
Sherry DF, Guigueno MF (2019) Cognition and the brain of brood parasitic cowbirds. Integ Zool 14:145–157. https://doi.org/10.1111/1749-4877.12312
Sherry DF, MacDougall-Shackleton SA (2015) Seasonal change in the avian hippocampus. Front Neuroendocrinol 37:158–167. https://doi.org/10.1016/j.yfrne.2014.11.008
Sherry DF, Forbes MR, Khurgel M, Ivy GO (1993) Females have a larger hippocampus than males in the brood-parasitic brown-headed cowbird. Proc Natl Acad Sci USA 90:7839–7843. https://doi.org/10.1073/pnas.90.16.7839
Spottiswoode CN, Kilner RM, Davies NB (2012) Brood parasitism. In: Royle NJ, Smiseth PT, Kölliker M (eds) The evolution of parental care. Oxford University Press, Oxford, pp 226–243
Vallortigara G (1996) Learning of colour and position cues in domestic chicks: males are better at position, females at colour. Behav Process 36:289–296. https://doi.org/10.1016/0376-6357(95)00063-1
White DJ (2019) Cowbird cognition: examinations of the mental skills of a brood parasite. Adv Stud Behav 51:143–175. https://doi.org/10.1016/bs.asb.2018.12.002
White DJ, Davies HB, Agyapong S, Seegmiller N (2017) Nest prospecting brown-headed cowbirds ‘parasitize’ social information when the value of personal information is lacking. Proc R Soc B 284:20171083. https://doi.org/10.1098/rspb.2017.1083
Acknowledgements
JLM was supported by a fellowship from the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). JCR is Research Fellow of CONICET. This manuscript has been greatly improved thanks to the suggestions of Associate Editor, N Miller, and two anonymous reviewers. We thank M Corral for the assistance in the experiments with shiny cowbirds, RO Gómez for helping with Fig. 1 and D Rodríguez and G Cueto for statistical advice. We are also grateful to C Barbato, LA Ciancio and ELPE SRL for donating different supplies.
Funding
This work was supported by grants from Universidad de Buenos Aires (UBACyT 20020170100521BA) and Agencia Nacional de Promoción Científica y Tecnológica (PICT 2015-1628).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing or financial interests.
Ethical approval
All work complied with Argentinean law and was undertaken with permission from Dirección Nacional de Fauna Silvestre from Argentina.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lois-Milevicich, J., Kacelnik, A. & Reboreda, J.C. Sex differences in the use of spatial cues in two avian brood parasites. Anim Cogn 24, 205–212 (2021). https://doi.org/10.1007/s10071-020-01434-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-020-01434-8