Abstract
Objective
The present study aimed to examine the effects of cDMARD and bDMARD therapy on both gene expressions and protein levels of TNF-α, IL-6, IL-10 and fatty acid levels in patients with RA.
Method
Plasma TNF-α, IL-6, and IL-10 levels were examined by the ELISA method, while TNF-α, IL-6, and IL-10 gene expression levels were examined by RT-qPCR, and fatty acid levels were examined by GC/MS.
Results
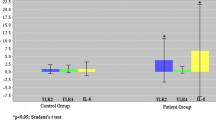
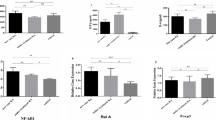
IL-10 gene expression levels significantly increased in RA patients receiving cDMARD treatment compared to those of the control group. Also, eicosadienoic acid, myristoleic acid and capric acid levels were significantly lower in the patient groups compared to those in the control group.
Conclusion
The drugs used in the treatment of RA had no effect on the fatty acid levels whereas had effects on the mRNA and protein levels of the target cytokines.
Key Points • There are some studies investigating inflammatory cytokines in rheumatoid arthritis but no study includes fatty acid levels besides the cytokines. Therefore, this study has an important contribution for original articles because of including fatty acid levels besides the cytokines on rheumatoid arthritis. |


Similar content being viewed by others
References
Chauhan K, Jandu JS, Goyal A, Bansal P, Al-Dhahir MA (2020) Rheumatoid arthritis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK441999/
Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M, Bridgett L, Williams S, Guillemin F, Hill CL, Laslett LL, Jones G, Cicuttini F, Osborne R, Vos T, Buchbinder R, Woolf A, March L (2014) The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis 73(7):1323–1330. https://doi.org/10.1136/annrheumdis-2013-204763
Smolen JS, Aletaha D, Barton A, Burmester GR, Emery P, Firestein GS, Kavanaugh A, McInnes IB, Solomon DH, Strand V, Yamamoto K (2018) Rheumatoid arthritis Nat Rev Dis Primers 8(4):18001. https://doi.org/10.1038/nrdp.2018.1
Ferreira HB, Melo T, Paiva A, Domingues MDR (2021) Insights in the role of lipids, oxidative stress and inflammation in rheumatoid arthritis unveiled by new trends in lipidomic investigations. Antioxidants (Basel) 2 10(1):45. https://doi.org/10.3390/antiox10010045
Quiñonez-Flores CM, González-Chávez SA, Del Río ND, Pacheco-Tena C (2016) Oxidative stress relevance in the pathogenesis of the rheumatoid arthritis: a systematic review. Biomed Res Int 2016:6097417. https://doi.org/10.1155/2016/6097417
Phull AR, Nasir B, Haq IU, Kim SJ (2018) Oxidative stress, consequences and ROS mediated cellular signaling in rheumatoid arthritis. Chem Biol Interact 1(281):121–136. https://doi.org/10.1016/j.cbi.2017.12.024
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd, Birnbaum NS, Burmester GR, Bykerk VP, Cohen MD, Combe B, Costenbader KH, Dougados M, Emery P, Ferraccioli G, Hazes JM, Hobbs K, Huizinga TW, Kavanaugh A, Kay J, Kvien TK, Laing T, Mease P, Ménard HA, Moreland LW, Naden RL, Pincus T, Smolen JS, Stanislawska-Biernat E, Symmons D, Tak PP, Upchurch KS, Vencovský J, Wolfe F, Hawker G (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62(9):2569–2581. https://doi.org/10.1002/art.27584
Lim JH, Goh LPW, Chong ETJ, Lee PC (2016) High integrity total RNA isolation from human peripheral blood that is as competitive to commercialize kits. Res J Pharm Biol Chem Sci 7(4):1937–1942
Rio DC, Ares M Jr, Hannon GJ, Nilsen TW (2010) Purification of RNA using TRIzol (TRI reagent). Cold Spring Harb Protoc 2010(6):pdb.prot5439. https://doi.org/10.1101/pdb.prot5439
Hruz T, Wyss M, Docquier M, Pfaffl MW, Masanetz S, Borghi L, Descombes P (2011) RefGenes: identification of reliable and condition specific reference genes for RT-qPCR data normalization. BMC Genomics 12(1):156. https://doi.org/10.1186/1471-2164-12-156
Keller C, Keller P, Marshal S, Pedersen BK (2003) IL-6 gene expression in human adipose tissue in response to exercise–effect of carbohydrate ingestion. J Physiol 1 550(3):927–931. https://doi.org/10.1113/jphysiol.2003.044883
Vors C, Allaire J, Marin J, Lépine MC, Charest A, Tchernof A, Lamarche B (2017) Inflammatory gene expression in whole blood cells after EPA vs. DHA supplementation: Results from the ComparED study. Atherosclerosis 257:116–122. https://doi.org/10.1016/j.atherosclerosis.2017.01.025
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Noack M, Miossec P (2017) Selected cytokine pathways in rheumatoid arthritis. Semin Immunopathol 39(4):365–383. https://doi.org/10.1007/s00281-017-0619-z
Radner H, Aletaha D (2015) Anti-TNF in rheumatoid arthritis: an overview. Wien Med Wochenschr 165(1–2):3–9. https://doi.org/10.1007/s10354-015-0344-y
Zhao PW, Jiang WG, Wang L, Jiang ZY, Shan YX, Jiang YF (2014) Plasma levels of IL-37 and correlation with TNF-α, IL-17A, and disease activity during DMARD treatment of rheumatoid arthritis. PLoS One 1 9(5):e95346. https://doi.org/10.1371/journal.pone.0095346
Musacchio E, Valvason C, Botsios C, Ostuni F, Furlan A, Ramonda R, Modesti V, Sartori L, Punzi L (2009) The tumor necrosis factor-{alpha}-blocking agent infliximab inhibits interleukin 1beta (IL-1beta) and IL-6 gene expression in human osteoblastic cells. J Rheumatol 36(8):1575–1579. https://doi.org/10.3899/jrheum.081321
Wielińska J, Dratwa M, Świerkot J, Korman L, Iwaszko M, Wysoczańska B, Bogunia-Kubik K (2018) Interleukin 6 gene polymorphism is associated with protein serum level and disease activity in Polish patients with rheumatoid arthritis. HLA 92(2):38–41. https://doi.org/10.1111/tan.13355
Smolen JS, Emery P, Fleischmann R, van Vollenhoven RF, Pavelka K, Durez P, Guérette B, Kupper H, Redden L, Arora V, Kavanaugh A (2014) Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet 383(9914):321–332. https://doi.org/10.1016/S0140-6736(13)61751-1
Detert J, Bastian H, Listing J, Weiß A, Wassenberg S, Liebhaber A, Rockwitz K, Alten R, Krüger K, Rau R, Simon C, Gremmelsbacher E, Braun T, Marsmann B, Höhne-Zimmer V, Egerer K, Buttgereit F, Burmester GR (2013) Induction therapy with adalimumab plus methotrexate for 24 weeks followed by methotrexate monotherapy up to week 48 versus methotrexate therapy alone for DMARD-naive patients with early rheumatoid arthritis: HIT HARD, an investigator-initiated study. Ann Rheum Dis 72(6):844–850. https://doi.org/10.1136/annrheumdis-2012-201612
Chen Z, Bozec A, Ramming A et al (2019) Anti-inflammatory and immune-regulatory cytokines in rheumatoid arthritis. Nat Rev Rheumatol 15:9–17. https://doi.org/10.1038/s41584-018-0109-2
Qu CH, Hou Y, Bi YF, Han QR, Jiao CH, Zou QF (2019) Diagnostic values of serum IL-10 and IL-17 in rheumatoid arthritis and their correlation with serum 14–3–3η protein. Eur Rev Med Pharmacol Sci 23(5):1899–1906. https://doi.org/10.26355/eurrev_201903_17227
Osiri M, Wongpiyabovorn J, Sattayasomboon Y, Thammacharoenrach N (2016) Inflammatory cytokine levels, disease activity, and function of patients with rheumatoid arthritis treated with combined conventional disease-modifying antirheumatic drugs or biologics. Clin Rheumatol 35(7):1673–1681. https://doi.org/10.1007/s10067-016-3306-x
Hernández-Bello J, Oregón-Romero E, Vázquez-Villamar M, García-Arellano S, Valle Y, Padilla-Gutiérrez JR, Román-Fernández IV, Palafox-Sánchez CA, Martínez-Bonilla GE, Muñoz-Valle JF (2017) Aberrant expression of interleukin-10 in rheumatoid arthritis: relationship with IL10 haplotypes and autoantibodies. Cytokine 95:88–96. https://doi.org/10.1016/j.cyto.2017.02.022
Farazi TA, Spitzer JI, Morozov P, Tuschl T (2011) miRNAs in human cancer. J Pathol 223(2):102–115. https://doi.org/10.1002/path.2806
Friedman JM, Jones PA (2009) MicroRNAs: critical mediators of differentiation, development and disease. Swiss Med Wkly 139(33–34):466–472
Liu Y, Chen Q, Song Y, Lai L, Wang J, Yu H, Cao X, Wang Q (2011) MicroRNA-98 negatively regulates IL-10 production and endotoxin tolerance in macrophages after LPS stimulation. FEBS Lett 585(12):1963–8. https://doi.org/10.1016/j.febslet.2011.05.029
Billeter AT, Hellmann J, Roberts H, Druen D, Gardner SA, Sarojini H, Galandiuk S, Chien S, Bhatnagar A, Spite M, Polk HC Jr (2014) MicroRNA-155 potentiates the inflammatory response in hypothermia by suppressing IL-10 production. FASEB J 28(12):5322–5336. https://doi.org/10.1096/fj.14-258335
Brouwers H, von Hegedus J, Toes R, Kloppenburg M, Ioan-Facsinay A (2015) Lipid mediators of inflammation in rheumatoid arthritis and osteoarthritis. Best Pract Res Clin Rheumatol 29(6):741–755. https://doi.org/10.1016/j.berh.2016.02.003
Frommer KW, Schäffler A, Rehart S, Lehr A, Müller-Ladner U, Neumann E (2015) Free fatty acids: potential proinflammatory mediators in rheumatic diseases. Ann Rheum Dis 74(1):303–310. https://doi.org/10.1136/annrheumdis-2013-203755
Frommer KW, Hasseli R, Schäffler A, Lange U, Rehart S, Steinmeyer J, Rickert M, Sarter K, Zaiss MM, Culmsee C, Ganjam G, Michels S, Müller-Ladner U, Neumann E (2019) Free fatty acids in bone pathophysiology of rheumatic diseases. Front Immunol 3(10):2757. https://doi.org/10.3389/fimmu.2019.02757
Rodríguez-Carrio J, Alperi-López M, López P, Ballina-García FJ, Suárez A (2016) Non-esterified fatty acids profiling in rheumatoid arthritis: associations with clinical features and Th1 response. PLoS One 11(8):e0159573. https://doi.org/10.1371/journal.pone.0159573
Kwon J-O, Jin WJ, Kim B, Kim H-H, Lee ZH (2015) Myristoleic acid inhibits osteoclast formation and bone resorption by suppressing the RANKL activation of Src and Pyk2. Eur J Pharmacol 768:189–198. https://doi.org/10.1016/j.ejphar.2015.10.053
Choi YK, Kang JI, Hyun JW, Koh YS, Kang JH, Hyun CG, Yoon KS, Lee KS, Lee CM, Kim TY, Yoo ES, Kang HK (2021) Myristoleic acid promotes anagen signaling by autophagy through activating Wnt/β-catenin and ERK pathways in dermal papilla cells. Biomol Ther (Seoul) 29(2):211–219. https://doi.org/10.4062/biomolther.2020.169
Romanowicz L, Bańkowski E (2009) Lipid compounds of the umbilical cord vein and their alterations in preeclampsia. Biochimie 91(2):288–294. https://doi.org/10.1016/j.biochi.2008.10.004
Kimura I, Ichimura A, Ohue-Kitano R, Igarashi M (2020) Free fatty acid receptors in health and disease. Physiol Rev 100(1):171–210. https://doi.org/10.1152/physrev.00041.2018
Shu J, Zhang F, Zhang L, Wei W (2017) G protein coupled receptors signaling pathways implicate in inflammatory and immune response of rheumatoid arthritis. Inflamm Res 66(5):379–387. https://doi.org/10.1007/s00011-016-1011-5
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The approval (2020/115) was obtained from the Ethics Committee of Hatay Mustafa Kemal University Faculty of Medicine. Each patient signed a consent form and the present study was performed in accordance with the Helsinki Declaration.
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dogan, S., Kimyon, G., Ozkan, H. et al. TNF-alpha, IL-6, IL-10 and fatty acids in rheumatoid arthritis patients receiving cDMARD and bDMARD therapy. Clin Rheumatol 41, 2341–2349 (2022). https://doi.org/10.1007/s10067-022-06180-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06180-5