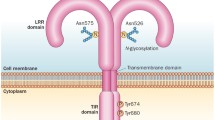
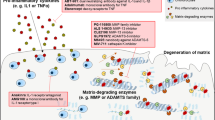
Abstract
Osteoarthritis (OA) is the most common joint disorder worldwide, and it has an enormous socioeconomic impact both in the United States and throughout the world. The degree of articular inflammation is usually associated with the disease’s progression, indicating that this process could contribute to articular damage. IL-1 beta and anti-TNF alpha are the two major cytokines players in the physiopathology of OA. Hence, we aimed to review the current literature on the effects of IL-1 and TNF-alpha neutralization as a new OA therapy. In vitro and experimental models showed a reduction in cartilage destruction with IL-1 inhibition therapy by IL-1 receptor antagonists (IL-1Ra). Despite this favorable evidence in animal models, studies on the inhibition of IL-1R in humans are still scarce. Although there is clear evidence that TNF-alpha plays a role in the pathophysiology of OA, only a few experimental trials have investigated the efficacy of blocking this pro-inflammatory cytokine in the treatment of OA. So far, the few studies available in humans using anti-TNF-alpha and IL-1 receptor antagonist are not remarkable, suggesting that further investigation and new therapeutic approaches are needed.

Similar content being viewed by others
References
Kramer JS, Yelin EH, Epstein WV (1983) Social and economic impacts of four musculoskeletal conditions: a study using national community-based data. J Rheumatol 26:901–907
Levy E, Ferme A, Perocheau D, Bono I (1993) Socioeconomic costs of osteoarthritis in France. Rev Rheum 60:63S–67S
Pelletier JP, Martel-Pelletier J, Abramson SB (2001) Osteoarthritis, an inflammatory disease: potential implication for the selection of new therapeutic targets. Arthritis Rheum 44(6):1237–1247
Mankin HJ, Dorfman H, Lippiello L, Zarins A (1971) Biochemical and metabolic abnormalities in articular cartilage from osteo-arthritic hips. II. Correlation of morphology with biochemical and metabolic data. J Bone Joint Surg Am 53:523–537
Meachim G, Osborne GV (1970) Repair at the femoral articular surface in osteo-arthritis of the hip. J Pathol 102:1–8
Shibakawa A, Aoki H, Masuko-Hongo K, Kato T et al (2003) Presence of pannus-like tissue on osteoarthritic cartilage and its histological character. Osteoarthritis Cartilage 11:133–140
Sakkas LI, Scanzello C, Johanson N, Burkholder J et al (1998) T cells and T-cell cytokine transcripts in the synovial membrane in patients with osteoarthritis. Clin Diagn Lab Immunol 5(4):430–437
Sharif M, Shepstone L, Elson CJ, Dieppe PA, Kirwan JR (2000) Increased serum C reactive protein may reflect events that precede radiographic progression in osteoarthritis of the knee. Ann Rheum Dis 59:71–74
Loeuille D, Chary-Valckenaere I, Champigneulle J, Rat AC, Toussaint F et al (2005) Macroscopic and microscopic features of synovial membrane inflammation in the osteoarthritic knee. Correlating magnetic resonance imaging findings with disease severity. Arthritis Rheum 52(11):3492–3501
Bondeson J, Wainwright SD, Lauder S, Amos N et al (2006) The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis. Arthritis Research & Therapy 8(6):1–12
Pelletier JP, Faure MP, DiBattista JA, Wilhelm S, Visco D, Martel-Pelletier J (1993) Coordinate synthesis of stromelysin, interleukin-1, and oncogene proteins in experimental osteoarthritis. An immunohistochemical study. Am J Pathol 142:95–105
Goldring MB (1999) The role of cytokines as inflammatory mediators in osteoarthritis: lessons from animal models. Connect Tissue Res 40:1–11
Schlaak JF, Schwarting A, Knolle P, Meyer zum Buschenfelde KH et al (1995) Effects of Th1 and Th2 cytokines on cytokine production and ICAM-1 expression on synovial fibroblasts. Ann Rheum Dis 54:560–565
Goldring MB, Sandel LJ, Stephenson ML, Krane SM (1996) Immune interferon suppresses levels of procollagen in mRNA and type II collagen synthesis in cultured human articular and costal chondrocytes. J Biol Chem 261:9049–9056
Jacques C, Bereziat G, Humbert L, Corvol M, Olivier JL, Masliah J et al (1997) Post-transcriptional effect of IGF-I on IL-1ß-induced type II secreted phospholipase A2 gene expression in rabbit articular chondrocytes. J Clin Invest 99:1864–1872
Clancy RM, Amin AR, Abramsom SB (1998) The role of nitric oxide in inflammation and immunity. Arthritis Rheum 41:1141–1151
Lotz M (1999) The role of nitric oxide in articular cartilage damage. Rheum Dis Clin North Am 25:269–282
Goldring MB, Berembaum F (1999) Human chondrocyte culture models for studying cyclooxygenase expression and prostaglandin regulation of collagen gene expression. Osteoarthritis Cartilage 7:386–388
Martel-Pelletier J, McCollum R, DiBattista J, Faure M-P, Chin JA, Fournier S et al (1992) The interleukin-1 receptor in normal and osteoarthritic human articular chondrocytes: identification as the type I receptor and analysis of binding kinetics and biologic function. Arthritis Rheum 35:530–540
Hedbom E, Hauselmann HJ (2002) Molecular aspects of pathogenesis in osteoarthritis: the role of inflammation. Cell Mol Life Sci 59:45–53
Martel-Pelletier J, Alaaeddine N, Pelletier JP (1999) Cytokines and their role in pathophysiology of osteoarthritis. Front Biosci 4:694–703
Shalom-Barak T, Quach J, Lotz M (1998) Interleukin-17-induced gene expression in articular chondrocytes is associated with activation of mitogen-activation protein kinases and NF-kB. J Biol Chem 273:27467–27473
Olee T, Hashimoto S, Quach J, Lotz M (1999) IL-18 is produced by articular chondrocytes and induces proinflammatory and catabolic responses. J Immunol 162:1096–1100
Arend WP, Dayer JM (1995) Inhibition of the production and effects of interleukin-1 and tumor necrosis alpha in rheumatoid arthritis. Arthritis Rheum 38:151–160
Arner EC, Harris RR, DiMeo TM, Collins RC, Galbraith W (1995) Interleukin-1 receptor antagonist inhibits proteoglycan breakdown in antigen induced but not polycation induced arthritis in the rabbit. J Rheumatol 22:1338–1346
Arend WP (1993) Interleukin-1 receptor antagonist. Adv Immunol 54:167–227
Caron JP, Fernandes JC, Martel-Pelletier J et al (1996) Chondroprotective effect of intra-articular injections of interleukin-1 receptor antagonist in experimental osteoarthritis. Suppression of collagenase-1 expression. Arthritis Rheum 39:1535–1544
Fernandes JC, Tardif G, Martel-Pelletier J et al (1999) In vivo transfer of interleukin-1 receptor antagonist gene in osteoarthritic rabbit knee joints: prevention of osteoarthitic progression. Am J Pathol 154:1535–1544
Pelletier JP, Caron JP, Evans C et al (1997) In vivo suppression of early experimental osteoarthritis by interleukin-1 receptor antagonist using gene therapy. Arthritis Rheum 40:1012–1019
Frisbie DD, Ghivizzani SC, Robbins PD, Evans CH, McIlwraith CW (2002) Treatment of experimental equine osteoarthritis by in vivo delivery of the equine interleukin-1 receptor antagonist gene. Gene Ther 9:12–20
Martel-Pelletier J, Mineau F, Jolicoeur FC et al (1998) In vitro effects of diacerhein and rhein on interleukin 1 and tumor necrosis factor-alpha systems in human osteoarthritic synovium and chondrocytes. J Rheumatol 25:753–762
Pelletier JP, Yaron N, Haraqui B et al (2000) Efficacy and safety of diacerein in osteoarthritis of the knee: a double blind, placebo-control trial. The diacerein study group. Arthritis Rheum 43:2339–2348
Dougados M, Nguyen M, Berdah L et al (2003) Evaluation of the structure-modifying effects of diacerein in hip osteoarthritis: ECHODIAH, a three-year, placebo-controlled trial. Evaluation of the chondromodulating effect of diacerein in OA of the hip. Clin Exp Rheumatol 21:152–154
Chevalier X, Girardeau B, Conrozier T, Marliere J, Kiefer P, Goupille P (2005) Safety study of intraarticular injection of interleukin 1 receptor antagonist in patients with painful knee osteoarthritis: a multicenter study. J Rheumatol 32:1317–1323
Chevalier X, Goupille P, Beaulieu AD, Burch FX, Bensen WG, Conrozier T, Loeuille D, Kivits AJ, Silver D, Appleton BE (2009) Intraarticular injection of anakinra in osteoarthritis of the knee: a multicenter, randomized, double-blind, placebo-controlled study. Artthritis Rheum 61:344–352
Grunke M, Schulze-Koops H (2006) Successful treatment of inflammatory knee osteoarthritis with tumor necrosis factor blockade. Ann Rheum Dis 65:555–556
Magnano MD, Chakravarty EF, Broudy C, Chung L, Kelman A, Hillygus J, Genovese MC (2007) A pilot study of tumor necrosis factor inhibition in erosive/inflammatory osteoarthritis of the hands. J Rheumatol 34:1323–1327
Abdiche YN, Malashock DS, Pons J (2008) Probing the binding mechanism and affinity of tanezumab, a recombinant humanized anti-NGF monoclonal antibody, using a repertoire of biosensors. Protein Sci 17:1326–1335
Lane NE, Schnitzer TJ, Smith MD et al (2008) Tanezumab relieves moderate to severe pain due to osteoarthritis of the knee: a phase 2 trial. In: American College of Rheumatology. http://acr.confex.com/acr/2008/webprogram/Paper3546.html
Pfizer (2008) A phase 3 randomized, double blind, placebo-controlled multicenter study of the analgesic efficacy and safety of tanezumab in patients with osteoarthritis of the knee. In: ClinicalTrials.gov. http://www.clinicaltrials.gov/show/NCT00733902. Accessed 4 Oct 2009
Pfizer (2008) A phase 3 randomized, double blind, placebo-controlled multicenter study of the analgesic efficacy and safety of tanezumab in patients with osteoarthritis of the hip. In: ClinicalTrials.gov. http://www.clinicaltrials.gov/show/NCT00744471. Acessed 4 Oct 2009
Disclosures
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Calich, A.L.G., Domiciano, D.S. & Fuller, R. Osteoarthritis: can anti-cytokine therapy play a role in treatment?. Clin Rheumatol 29, 451–455 (2010). https://doi.org/10.1007/s10067-009-1352-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-009-1352-3