Abstract
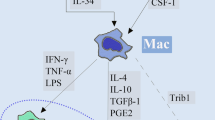
Tumor-associated macrophages (TAMs) accumulate in various cancers and promote tumor angiogenesis and metastasis, and thus may be ideal targets for the clinical diagnosis of tumor metastasis with high specificity. However, there are few specific markers to distinguish between TAMs and normal or inflammatory macrophages. Here, we show that TAMs localize in green fluorescent protein-labeled tumors of metastatic lymph nodes (MLNs) from B16F1 melanoma cells but not in necrotic tumor regions, suggesting that TAMs may promote the growth of tumor cells and the progression of tumor metastasis. Furthermore, we isolated pure populations of TAMs from MLNs and characterized their gene expression signatures compared to peritoneal macrophages (PMs), and found that TAMs significantly overexpress immunosuppressive cytokines such as IL-4, IL-10, and TGF-β as well as proangiogenic factors such as VEGF, TIE2, and CD31. Notably, immunological analysis revealed that TIE2+/CD31+ macrophages constitute the predominant population of TAMs that infiltrate MLNs, distinct from tissue or inflammatory macrophages. Importantly, these TIE2+/CD31+ macrophages also heavily infiltrated MLNs from human breast cancer biopsies but not reactive hyperplastic LNs. Thus, TIE2+/ CD31+ macrophages may be a unique histopathological biomarker for detecting metastasis in clinical diagnosis, and a novel and promising target for TAM-specific cancer therapy.
Similar content being viewed by others
References
Ahn, G.O., Tseng, D., Liao, C.H., Dorie, M.J., Czechowicz, A., and Brown, J.M. (2010). Inhibition of Mac-1 (CD11b/CD18) enhances tumor response to radiation by reducing myeloid cell recruitment. Proc. Natl. Acad. Sci. USA 107, 8363–8368.
Fei, Y.Y., Landry, J.P., Sun, Y.S., Zhu, X.D., Luo, J.T., Wang, X.B., and Lam, K.S. (2008). A novel high-throughput scanning microscope for label-free detection of protein and small-molecule chemical microarrays. Rev. Sci. Instrum. 79, 013708.
Garcia, J., Sandi, M.J., Cordelier, P., Binetruy, B., Pouyssegur, J., Iovanna, J.L., and Tournaire, R. (2012). Tie1 deficiency induces endothelial-mesenchymal transition. EMBO Rep. 13, 431–439.
Giatromanolaki, A., Koukourakis, M.I., Kakolyris, S., Kaklamanis, L., Barbatis, K., O’Byrne, K.J., Theodosssiou, D., Harris, A.L., and Gatter, K.C. (1998). Focal expression of thymidine phosphorylase associates with CD31 positive lymphocytic aggregation and local neo-angiogenesis in non-small cell lung cancer. Anticancer Res. 18, 71–76.
Groblewska, M., Mroczko, B., Wereszczynska-Siemiatkowska, U., Mysliwiec, P., Kedra, B., and Szmitkowski, M. (2007). Serum levels of granulocyte colony-stimulating factor (G-CSF) and macrophage colony-stimulating factor (M-CSF) in pancreatic cancer patients. Clin. Chem. Lab. Med. 45, 30–34.
Heusinkveld, M., and van der Burg, S.H. (2011). Identification and manipulation of tumor associated macrophages in human cancers. J. Transl. Med. 9, 216.
Hill, R.P., Chambers, A.F., Ling, V., and Harris, J.F. (1984). Dynamic heterogeneity: rapid generation of metastatic variants in mouse B16 melanoma cells. Science 224, 998–1001.
Huang, S., Xie, K., Bucana, C.D., Ullrich, S.E., and Bar-Eli, M. (1996). Interleukin 10 suppresses tumor growth and metastasis of human melanoma cells: potential inhibition of angiogenesis. Clin. Cancer Res. 2, 1969–1979.
Jenkins, S.J., Ruckerl, D., Cook, P.C., Jones, L.H., Finkelman, F.D., van Rooijen, N., MacDonald, A.S., and Allen, J.E. (2011). Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science 332, 1284–1288.
Kim, D.W., Min, H.S., Lee, K.H., Kim, Y.J., Oh, D.Y., Jeon, Y.K., Lee, S.H., Im, S.A., Chung, D.H., Kim, Y.T., et al. (2008). High tumour islet macrophage infiltration correlates with improved patient survival but not with EGFR mutations, gene copy number or protein expression in resected non-small cell lung cancer. Br. J. Cancer 98, 1118–1124.
Kim, S.J., Kim, J.S., Papadopoulos, J., Wook Kim, S., Maya, M., Zhang, F., He, J., Fan, D., Langley, R., and Fidler, I.J. (2009). Circulating monocytes expressing CD31: implications for acute and chronic angiogenesis. Am. J. Pathol. 174, 1972–1980.
Lan, C., Huang, X., Lin, S., Huang, H., Cai, Q., Wan, T., Lu, J., and Liu, J. (2013) Expression of M2-polarized macrophages is associated with poor prognosis for advanced epithelial ovarian cancer. Technol. Cancer Res. Treat 12, 259–267.
Mantovani, A., and Sica, A. (2010). Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr. Opin. Immunol. 22, 231–237.
Mantovani, A., Sozzani, S., Locati, M., Allavena, P., and Sica, A. (2002). Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 23, 549–555.
Mantovani, A., Allavena, P., Sica, A., and Balkwill, F. (2008). Cancer-related inflammation. Nature 454, 436–444.
Mazzieri, R., Pucci, F., Moi, D., Zonari, E., Ranghetti, A., Berti, A., Politi, L.S., Gentner, B., Brown, J.L., Naldini, L., et al. (2011). Targeting the ANG2/TIE2 axis inhibits tumor growth and metastasis by impairing angiogenesis and disabling rebounds of proangiogenic myeloid cells. Cancer Cell 19, 512–526.
McKenney, J.K., Weiss, S.W., and Folpe, A.L. (2001). CD31 expression in intratumoral macrophages: a potential diagnostic pitfall. Am. J. Surg. Pathol. 25, 1167–1173.
Pollard, J.W. (2004). Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer 4, 71–78.
Pollard, J.W. (2008). Macrophages define the invasive microenvironment in breast cancer. J. Leukoc. Biol. 84, 623–630.
Pollard, J.W. (2009). Trophic macrophages in development and disease. Nat. Rev. Immunol. 9, 259–270.
Pucci, F., Venneri, M.A., Biziato, D., Nonis, A., Moi, D., Sica, A., Di Serio, C., Naldini, L., and De Palma, M. (2009) A distinguishing gene signature shared by tumor-infiltrating Tie2-expressing monocytes, blood “resident” monocytes, and embryonic macrophages suggests common functions and developmental relationships. Blood 114, 901–914.
Qian, B.Z., and Pollard, J.W. (2010). Macrophage diversity enhances tumor progression and metastasis. Cell 141, 39–51.
Quatromoni, J.G., and Eruslanov, E. (2012). Tumor-associated macrophages: function, phenotype, and link to prognosis in human lung cancer. Am. J. Transl. Res. 4, 376–389.
Ryder, M., Ghossein, R.A., Ricarte-Filho, J.C., Knauf, J.A., and Fagin, J.A. (2008). Increased density of tumor-associated macro-phages is associated with decreased survival in advanced thyroid cancer. Endocr. Relat. Cancer 15, 1069–1074.
Schoppmann, S.F., Fenzl, A., Nagy, K., Unger, S., Bayer, G., Geleff, S., Gnant, M., Horvat, R., Jakesz, R., and Birner, P. (2006). VEGF-C expressing tumor-associated macrophages in lymph node positive breast cancer: impact on lymphangiogenesis and survival. Surgery 139, 839–846.
Steidl, C., Lee, T., Shah, S.P., Farinha, P., Han, G., Nayar, T., Delaney, A., Jones, S.J., Iqbal, J., Weisenburger, D.D., et al. (2010). Tumor-associated macrophages and survival in classic Hodgkin’s lymphoma. N. Engl. J. Med. 362, 875–885.
Welford, A.F., Biziato, D., Coffelt, S.B., Nucera, S., Fisher, M., Pucci, F., Di Serio, C., Naldini, L., De Palma, M., Tozer, G.M., et al. (2011). TIE2-expressing macrophages limit the therapeutic efficacy of the vascular-disrupting agent combretastatin A4 phosphate in mice. J. Clin. Invest. 121, 1969–1973.
White, E.S., Flaherty, K.R., Carskadon, S., Brant, A., Iannettoni, M.D., Yee, J., Orringer, M.B., and Arenberg, D.A. (2003). Macrophage migration inhibitory factor and CXC chemokine expression in non-small cell lung cancer: role in angiogenesis and prognosis. Clin. Cancer Res. 9, 853–860.
Xu, J., Escamilla, J., Mok, S., David, J., Priceman, S., West, B., Bollag, G., McBride, W., and Wu, L. (2013). CSF1R signaling blockade stanches tumor-infiltrating myeloid cells and improves the efficacy of radiotherapy in prostate cancer. Cancer Res. 73, 2782–2794.
Yang, H., Kim, C., Kim, M.J., Schwendener, R.A., Alitalo, K., Heston, W., Kim, I., Kim, W.J., and Koh, G.Y. (2011). Soluble vascular endothelial growth factor receptor-3 suppresses lymphangiogenesis and lymphatic metastasis in bladder cancer. Mol. Cancer 10, 36.
Zeisberger, S.M., Odermatt, B., Marty, C., Zehnder-Fjallman, A.H., Ballmer-Hofer, K., and Schwendener, R.A. (2006). Clodronateliposome-mediated depletion of tumour-associated macrophages: a new and highly effective antiangiogenic therapy approach. Br. J. Cancer 95, 272–281.
Zijlmans, H.J., Fleuren, G.J., Baelde, H.J., Eilers, P.H., Kenter, G.G., and Gorter, A. (2006). The absence of CCL2 expression in cervical carcinoma is associated with increased survival and loss of heterozygosity at 17q11.2. J. Pathol. 208, 507–517.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kim, OH., Kang, GH., Noh, H. et al. Proangiogenic TIE2+/CD31+ macrophages are the predominant population of tumor-associated macrophages infiltrating metastatic lymph nodes. Mol Cells 36, 432–438 (2013). https://doi.org/10.1007/s10059-013-0194-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-0194-7