Abstract
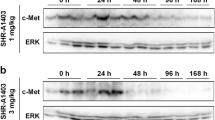
c-Met, the high affinity receptor for hepatocyte growth factor (HGF), is one of the most frequently activated tyrosine kinases in many human cancers and a target for cancer therapy. However, inhibitory targeting of c-Met with antibodies has proven difficult, because most antibodies have intrinsic agonist activity. Therefore, the strategy for reducing the agonism is critical for successful development of cancer therapies based on anti-c-Met antibodies. Here we developed a mechanism-based assay method for rapid screening of anti-c-Met antibodies, involving the determination of Akt phosphorylation and c-Met degradation for agonism and efficacy, respectively. Using the method, we identified an antibody, F46, that binds to human c-Met with high affinity (Kd = 2.56 nM) and specificity, and induces the degradation of c-Met in multiple cancer cells (including MKN45, a gastric cancer cell line) with minimal activation of c-Met signaling. F46 induced c-Met internalization in both HGF-dependent and HGF-independent cells, suggesting that the degradation of c-Met results from antibody-mediated receptor internalization. Furthermore, F46 competed with HGF for binding to c-Met, resulting in the inhibition of both HGF-mediated invasion and angiogenesis. Consistently, F46 inhibited the proliferation of MKN45 cells, in which c-Met is constitutively activated in an HGF-independent manner. Xenograft analysis revealed that F46 markedly inhibits the growth of subcutaneously implanted gastric and lung tumors. These results indicate that F46, identified by a novel mechanism-based assay, induces c-Met degradation with minimal agonism, implicating a potential role of F46 in therapy of human cancers.
Similar content being viewed by others
References
Benvenuti, S., and Comoglio, P.M. (2007). The MET receptor tyrosine kinase in invasion and metastasis. J. Cell. Physiol. 213, 316–325.
Birchmeier, C., and Gherardi, E. (1998). Developmental roles of HGF/SF and its receptor, the c-Met tyrosine kinase. Trends Cell Biol. 8, 404–410.
Birchmeier, C., Birchmeier, W., Gherardi, E., and Vande Woude, G.F. (2003). Met, metastasis, motility and more. Nat. Rev. Mol. Cell Biol. 4, 915–925.
Boccaccio, C., and Comoglio, P.M. (2006). Invasive growth, a METdriven genetic programme for cancer and stem cells. Nat. Rev. Cancer 6, 637–645.
Borowiak, M., Garratt, A.N., Wustefeld, T., Strehle, M., Trautwein, C., and Birchmeier, C. (2004). Met provides essential signals for liver regeneration. Proc. Natl. Acad. Sci. USA 101, 10608–10613.
Burgess, T., Coxon, A., Meyer, S., Sun, J., Rex, K., Tsuruda, T., Chen, Q., Ho, S.Y., Li, L., Kaufman, S., et al. (2006). Fully human monoclonal antibodies to hepatocyte growth factor with therapeutic potential against hepatocyte growth factor/c-Metdependent human tumors. Cancer Res. 66, 1721–1729.
Burgess, T.L., Sun, J., Meyer, S., Tsuruda, T.S., Elliott, G., Chen, Q., Haniu, M., Barron, W.F., Juan, T., Zhang, K., et al. (2010). Biochemical characterization of AMG 102, a neutralizing, fully human monoclonal antibody to human and nonhuman primate hepatocyte growth factor. Mol. Cancer Ther. 9, 400–409.
Bussolino, F., Di Renzo, M.F., Ziche, M., Bocchietto, E., Olivero, M., Naldini, L., Gaudino, G., Tamagnone, L., Coffer, A., and Comoglio, P.M. (1992). Hepatocyte growth factor is a potent angiogenic factor which stimulates endothelial cell motility and growth. J. Cell Biol. 119, 629–641.
Cao, B., Su, Y., Oskarsson, M., Zhao, P., Kort, E.J., Fisher, R.J., Wang, L.M., and Vande Woude, G.F. (2001). Neutralizing monoclonal antibodies to hepatocyte growth factor/scatter factor (HGF/SF) display antitumor activity in animal models. Proc. Natl. Acad. Sci. USA 98, 7443–7448.
Chmielowiec, J., Borowiak, M., Morkel, M., Stradal, T., Munz, B., Werner, S., Wehland, J., Birchmeier, C., and Birchmeier, W. (2007). c-Met is essential for wound healing in the skin. J. Cell Biol. 177, 151–162.
Comoglio, P.M., and Trusolino, L. (2002). Invasive growth, from development to metastasis. J. Clin. Invest. 109, 857–862.
Cragg, M.S., French, R.R., and Glennie, M.J. (1999). Signaling antibodies in cancer therapy. Curr. Opin. Immun. 11, 541–547.
Ferrara, N., Hillan, K.J., Gerber, H.P., and Novotny, W. (2004). Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat. Rev. Drug Dis. 3, 391–400.
Gschwind, A., Fischer, O.M., and Ullrich, A. (2004). The discovery of receptor tyrosine kinases, targets for cancer therapy. Nat. Rev. Cancer 4, 361–370.
Hanahan, D., and Weinberg, R.A. (2011). Hallmarks of cancer, the next generation. Cell 144, 646–674.
Huh, C.G., Factor, V.M., Sanchez, A., Uchida, K., Conner, E.A., and Thorgeirsson, S.S. (2004). Hepatocyte growth factor/c-met signaling pathway is required for efficient liver regeneration and repair. Proc. Natl. Acad. Sci. USA 101, 4477–4482.
Jin, H., Yang, R., Zheng, Z., Romero, M., Ross, J., Bou-Reslan, H., Carano, R.A., Kasman, I., Mai, E., Young, J., et al. (2008). Met-MAb, the one-armed 5D5 anti-c-Met antibody, inhibits orthotopic pancreatic tumor growth and improves survival. Cancer Res. 68, 4360–4368.
Jo, S., Hong, S.W., Yoo, J.W., Lee, C.H., Kim, S., Kim, S., and Lee, D. (2011). Selection and optimization of asymmetric siRNA targeting the human c-MET gene. Mol. Cells 32, 543–548.
Kim, K.J., Wang, L., Su, Y.C., Gillespie, G.Y., Salhotra, A., Lal, B., and Laterra, J. (2006). Systemic anti-hepatocyte growth factor monoclonal antibody therapy induces the regression of intracranial glioma xenografts. Clin. Cancer Res.12, 1292–1298.
Lamszus, K., Laterra, J., Westphal, M., and Rosen, E.M. (1999). Scatter factor/hepatocyte growth factor (SF/HGF) content and function in human gliomas. Int. J. Dev. Neurosci. 17, 517–530.
Li, S., Schmitz, K.R., Jeffrey, P.D., Wiltzius, J.J., Kussie, P., and Ferguson, K.M. (2005). Structural basis for inhibition of the epidermal growth factor receptor by cetuximab. Cancer Cell 7, 301–311.
Ma, P.C., Jagadeeswaran, R., Jagadeesh, S., Tretiakova, M.S., Nallasura, V., Fox, E.A., Hansen, M., Schaefer, E., Naoki, K., Lader, A., et al. (2005). Functional expression and mutations of c-Met and its therapeutic inhibition with SU11274 and small interfering RNA in non-small cell lung cancer. Cancer Res. 65, 1479–1488.
Martens, T., Schmidt, N.O., Eckerich, C., Fillbrandt, R., Merchant, M., Schwall, R., Westphal, M., and Lamszus, K. (2006). A novel one-armed anti-c-Met antibody inhibits glioblastoma growth in vivo. Clin. Cancer Res. 12, 6144–6152.
Nakagawa, T., Tohyama, O., Yamaguchi, A., Matsushima, T., Takahashi, K., Funasaka, S., Shirotori, S., Asada, M., and Obaishi, H. (2010). E7050, a dual c-Met and VEGFR-2 tyrosine kinase inhibitor promotes tumor regression and prolongs survival in mouse xenograft models. Cancer Sci. 101, 210–215.
Ohashi, K., Marion, P.L., Nakai, H., Meuse, L., Cullen, J.M., Bordier, B.B., Schwall, R., Greenberg, H.B., Glenn, J.S., and Kay, M.A. (2000). Sustained survival of human hepatocytes in mice, A model for in vivo infection with human hepatitis B and hepatitis delta viruses. Nat. Med. 6, 327–331.
Prat, M., Crepaldi, T., Pennacchietti, S., Bussolino, F., and Comoglio, P.M. (1998). Agonistic monoclonal antibodies against the Met receptor dissect the biological responses to HGF. J. Cell Sci. 111 (Pt 2), 237–247.
Trusolino, L., and Comoglio, P.M. (2002). Scatter-factor and semaphorin receptors, cell signalling for invasive growth. Nat. Rev. Cancer 2, 289–300.
Trusolino, L., Bertotti, A., and Comoglio, P.M. (2010). MET signalling, principles and functions in development, organ regeneration and cancer. Nat. Rev. Mol. Cell Biol. 11, 834–848.
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Oh, Y.M., Song, YJ., Lee, S.B. et al. A new anti-c-met antibody selected by a mechanism-based dual-screening method: Therapeutic potential in cancer. Mol Cells 34, 523–529 (2012). https://doi.org/10.1007/s10059-012-0194-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-012-0194-z