Abstract
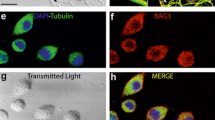
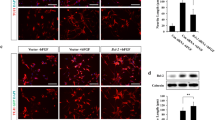
The BNIPs (BCL2 and adenovirus E1B 19 kDa interacting proteins) are a subfamily of BCL2 family proteins typically containing a single BCL2 homology 3 (BH3) domain. BNIPs exert important roles in two major degradation processes in cells — apoptosis and autophagy. Although it is known that the function of BNIPs is transcriptionally regulated under hypoxic conditions in tumors, their regulation in the developing brain and neurons following the induction of apoptosis/autophagy is largely unknown. In this study, we demonstrate that three members of the BNIP family, BNIP1, BNIP3 and BNIP3L, are expressed in the developing brain with distinct brain region specificity. BNIP3 mRNA was especially enriched in the entorhinal cortex, raising a possibility that it may have additional biological functions in addition to its apoptotic and autophagic functions. Following starvation-induced autophagy induction, BNIP1 mRNA was selectively increased in cultured neurons. However, the apoptogenic chemical staurosporine failed to modulate the expression of BNIPs, which is in contrast to the marked induction of all BNIPs by glucose-oxygen deprivation. Finally, neonatal nerve axotomy, which triggers apoptosis in motoneurons, selectively enhanced BNIP3 mRNA expression. Collectively, these results suggest that the expression of BNIPs is differentially regulated depending on the stimuli, and BNIPs may exert unique biological functions.
Similar content being viewed by others
References
Birse-Archbold, J.L., Kerr, L.E., Jones, P.A., McCulloch, J., and Sharkey, J. (2005). Differential profile of Nix upregulation and translocation during hypoxia/ischaemia in vivo versus in vitro. J. Cereb. Blood Flow Metab. 25, 1356–1365.
Boyd, J.M., Malstrom, S., Subramanian, T., Venkatesh, L.K., Schaeper, U., Elangovan, B., D’sa-Eipper, C., and Chinnadurai, G. (1994). Adenovirus E1B 19 kDa and Bcl-2 proteins interact with a common set of cellular proteins. Cell 79, 341–351.
Buss, R.R., Sun, W., and Oppenheim, R.W. (2006). Adaptive roles of programmed cell death during nervous system development. Annu. Rev. Neurosci. 29, 1–35.
Chinnadurai, G., Vijayalingam, S., and Gibson, S.B. (2008). BNIP3 subfamily BH3-only proteins: mitochondrial stress sensors in normal and pathological functions. Oncogene 27(Suppl 1), S114–127.
Cleland, M.M., Norris, K.L., Karbowski, M., Wang, C., Suen, D.F., Jiao, S., George, N.M., Luo, X., Li, Z., and Youle, R.J. (2011). Bcl-2 family interaction with the mitochondrial morphogenesis machinery. Cell Death Differ. 18, 235–247.
Daido, S., Kanzawa, T., Yamamoto, A., Takeuchi, H., Kondo, Y., and Kondo, S. (2004). Pivotal role of the cell death factor BNIP3 in ceramide-induced autophagic cell death in malignant glioma cells. Cancer Res. 64, 4286–4293.
Galvez, A.S., Brunskill, E.W., Marreez, Y., Benner, B.J., Regula, K.M., Kirschenbaum, L.A., and Dorn, G.W., 2nd (2006). Distinct pathways regulate proapoptotic Nix and BNip3 in cardiac stress. J. Biol. Chem. 281, 1442–1448.
Gilley, J., Coffer, P.J., and Ham, J. (2003). FOXO transcription factors directly activate bim gene expression and promote apoptosis in sympathetic neurons. J. Cell Biol. 162, 613–622.
Halterman, M.W., Miller, C.C., and Federoff, H.J. (1999). Hypoxiainducible factor-1alpha mediates hypoxia-induced delayed neuronal death that involves p53. J. Neurosci. 19, 6818–6824.
Hamacher-Brady, A., Brady, N.R., Logue, S.E., Sayen, M.R., Jinno, M., Kirshenbaum, L.A., Gottlieb, R.A., and Gustafsson, A.B. (2007). Response to myocardial ischemia/reperfusion injury involves Bnip3 and autophagy. Cell Death Differ. 14, 146–157.
Imazu, T., Shimizu, S., Tagami, S., Matsushima, M., Nakamura, Y., Miki, T., Okuyama, A., and Tsujimoto, Y. (1999). Bcl-2/E1B 19 kDa-interacting protein 3-like protein (Bnip3L) interacts with bcl-2/Bcl-xL and induces apoptosis by altering mitochondrial membrane permeability. Oncogene 18, 4523–4529.
Kanzawa, T., Zhang, L., Xiao, L., Germano, I.M., Kondo, Y., and Kondo, S. (2005). Arsenic trioxide induces autophagic cell death in malignant glioma cells by upregulation of mitochondrial cell death protein BNIP3. Oncogene 24, 980–991.
Karbowski, M., Norris, K.L., Cleland, M.M., Jeong, S.Y., and Youle, R.J. (2006). Role of Bax and Bak in mitochondrial morphogenesis. Nature 443, 658–662.
Kiryu-Seo, S., Hirayama, T., Kato, R., and Kiyama, H. (2005). Noxa is a critical mediator of p53-dependent motor neuron death after nerve injury in adult mouse. J. Neurosci. 25, 1442–1447.
Landes, T., Emorine, L.J., Courilleau, D., Rojo, M., Belenguer, P., and Arnaune-Pelloquin, L. (2010). The BH3-only Bnip3 binds to the dynamin Opa1 to promote mitochondrial fragmentation and apoptosis by distinct mechanisms. EMBO Rep. 11, 459–465.
Lee, H., and Paik, S.G. (2006). Regulation of BNIP3 in normal and cancer cells. Mol. Cells 21, 1–6.
Lee, Y., Lee, H.Y., Hanna, R.A., and Gustafsson, A.B. (2011). Mitochondrial autophagy by Bnip3 involves Drp1-mediated mitochondrial fission and recruitment of Parkin in cardiac myocytes. Am. J. Physiol. Heart Circ. Physiol. 301, H1924–1931.
Leist, M., and Jaattela, M. (2001). Triggering of apoptosis by cathepsins. Cell Death Differ. 8, 324–326.
Levine, B., Sinha, S., and Kroemer, G. (2008). Bcl-2 family members: dual regulators of apoptosis and autophagy. Autophagy 4, 600–606.
Liang, X.H., Kleeman, L.K., Jiang, H.H., Gordon, G., Goldman, J.E., Berry, G., Herman, B., and Levine, B. (1998). Protection against fatal Sindbis virus encephalitis by beclin, a novel Bcl-2-interacting protein. J. Virol. 72, 8586–8596.
Lomonosova, E., and Chinnadurai, G. (2008). BH3-only proteins in apoptosis and beyond: an overview. Oncogene 27(Suppl 1), S2–19.
Matsushima, M., Fujiwara, T., Takahashi, E., Minaguchi, T., Eguchi, Y., Tsujimoto, Y., Suzumori, K., and Nakamura, Y. (1998). Isolation, mapping, and functional analysis of a novel human cDNA (BNIP3L) encoding a protein homologous to human NIP3. Genes Chromosomes Cancer 21, 230–235.
Mizushima, N., and Komatsu, M. (2011). Autophagy: renovation of cells and tissues. Cell 147, 728–741.
Noh, M.R., Kim, S.K., Sun, W., Park, S.K., Choi, H.C., Lim, J.H., Kim, I.H., Kim, H.J., Kim, H., and Eun, B.L. (2006). Neuroprotective effect of topiramate on hypoxic ischemic brain injury in neonatal rats. Exp. Neurol. 201, 470–478.
Oakes, S.A., Scorrano, L., Opferman, J.T., Bassik, M.C., Nishino, M., Pozzan, T., and Korsmeyer, S.J. (2005). Proapoptotic BAX and BAK regulate the type 1 inositol trisphosphate receptor and calcium leak from the endoplasmic reticulum. Proc. Natl. Acad. Sci. USA 102, 105–110.
Oppenheim, R.W., Houenou, L.J., Johnson, J.E., Lin, L.F., Li, L., Lo, A.C., Newsome, A.L., Prevette, D.M., and Wang, S. (1995). Developing motor neurons rescued from programmed and axotomy-induced cell death by GDNF. Nature 373, 344–346.
Park, O.H., Lee, K.J., Rhyu, I.J., Geum, D., Kim, H., Buss, R., Oppenheim, R.W., and Sun, W. (2007). Bax-dependent and — independent death of motoneurons after facial nerve injury in adult mice. Eur. J. Neurosci. 26, 1421–1432.
Ryu, S.W., Choi, K., Kim, S., and Choi, C. (2012). Endoplasmic reticulum-specific BH3-only protein BNIP1 induces mitochondrial fragmentation in a Bcl-2- and Drp1-dependent manner. J. Cell Physiol. 227, 3027–3035.
Sandoval, H., Thiagarajan, P., Dasgupta, S.K., Schumacher, A., Prchal, J.T., Chen, M., and Wang, J. (2008). Essential role for Nix in autophagic maturation of erythroid cells. Nature 454, 232–235.
Scorrano, L., Oakes, S.A., Opferman, J.T., Cheng, E.H., Sorcinelli, M.D., Pozzan, T., and Korsmeyer, S.J. (2003). BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science 300, 135–139.
Sendtner, M., Holtmann, B., Kolbeck, R., Thoenen, H., and Barde, Y.A. (1992). Brain-derived neurotrophic factor prevents the death of motoneurons in newborn rats after nerve section. Nature 360, 757–759.
Sowter, H.M., Ratcliffe, P.J., Watson, P., Greenberg, A.H., and Harris, A.L. (2001). HIF-1-dependent regulation of hypoxic induction of the cell death factors BNIP3 and NIX in human tumors. Cancer Res. 61, 6669–6673.
Sun, W., and Oppenheim, R.W. (2003). Response of motoneurons to neonatal sciatic nerve axotomy in Bax-knockout mice. Mol. Cell. Neurosci. 24, 875–886.
Tracy, K., Dibling, B.C., Spike, B.T., Knabb, J.R., Schumacker, P., and Macleod, K.F. (2007). BNIP3 is an RB/E2F target gene required for hypoxia-induced autophagy. Mol. Cell. Biol. 27, 6229–6242.
Villunger, A., Michalak, E.M., Coultas, L., Mullauer, F., Bock, G., Ausserlechner, M.J., Adams, J.M., and Strasser, A. (2003). p53- and drug-induced apoptotic responses mediated by BH3-only proteins puma and noxa. Science 302, 1036–1038.
Wakabayashi, T., Kosaka, J., and Hommura, S. (2002). Up-regulation of Hrk, a regulator of cell death, in retinal ganglion cells of axotomized rat retina. Neurosci. Lett. 318, 77–80.
Wakabayashi, T., Kosaka, J., and Oshika, T. (2005). JNK inhibitory kinase is up-regulated in retinal ganglion cells after axotomy and enhances BimEL expression level in neuronal cells. J. Neurochem. 95, 526–536.
Yan, Q., Matheson, C., and Lopez, O.T. (1995). In vivo neurotrophic effects of GDNF on neonatal and adult facial motor neurons. Nature 373, 341–344.
Youle, R.J., and Strasser, A. (2008). The BCL-2 protein family: opposing activities that mediate cell death. Nat. Rev. Mol. Cell. Biol. 9, 47–59.
Zhang, J., and Ney, P.A. (2009). Role of BNIP3 and NIX in cell death, autophagy, and mitophagy. Cell Death Differ. 16, 939–946.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Cho, B., Choi, S.Y., Park, Oh. et al. Differential expression of BNIP family members of BH3-only proteins during the development and after axotomy in the rat. Mol Cells 33, 605–610 (2012). https://doi.org/10.1007/s10059-012-0051-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-012-0051-0