Abstract
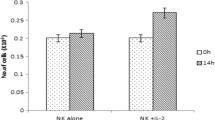
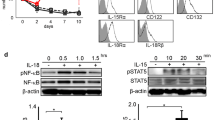
Interleukin 10 (IL-10) is a multifunctional cytokine that regulates diverse functions of immune cells. Natural killer (NK) cells express the IL-10 and IL-10 receptor, but little is known about the function of IL-10 on NK cell activation. In this study, we show the expression and role of IL-10 in human NK cells. Among the cytokines tested, IL-15 was the most potent inducer of IL-10, with a maximal peak expression at 5 h after treatment. Furthermore, IL-10 receptor was shown to be expressed in NK cells. IL-10 alone had a significant effect on NK cytotoxicity which additively increased NK cell cytotoxicity in the presence of IL-15. Neutralizing IL-10 with anti-IL-10 antibody suppressed the inductive effect of IL-10 on NK cell cytotoxicity; however, IL-10 had no effect on IFN-γ or TNF-α production or NK cell activatory receptor expression. STAT signals are implicated as a key mediator of IL-10/IL-15 cytotoxicity response. Thus, the effect of IL-10 on NK cells is particularly interesting with regard to the STAT3 signal that was enhanced by IL-10 or IL-15.
Similar content being viewed by others
References
Alter, G., Kavanagh, D., Rihn, S., Luteijn, R., Brooks, D., Oldstone, M., van Lunzen, J., and Altfeld, M. (2010). IL-10 induces aberrant deletion of dendritic cells by natural killer cells in the context of HIV infection. J. Clin. Invest. 120, 1905–1913.
Asseman, C., Mauze, S., Leach, M.W., Coffman, R.L., and Powrie, F. (1999). An essential role for interleukin 10 in the function of regulatory T cells that inhibit intestinal inflammation. J. Exp. Med. 190, 995–1004.
Becknell, B., and Caligiuri, M.A. (2005). Interleukin-2, interleukin-15, and their roles in human natural killer cells. Adv. Immunol. 86, 209–239.
D’Andrea, A., Aste-Amezaga, M., Valiante, N.M., Ma, X., Kubin, M., and Trinchieri, G. (1993). Interleukin 10 (IL-10) inhibits human lymphocyte interferon gamma-production by suppressing natural killer cell stimulatory factor/IL-12 synthesis in accessory cells. J. Exp. Med. 178, 1041–1048.
de Waal Malefyt, R., Haanen, J., Spits, H., Roncarolo, M.G., te Velde, A., Figdor, C., Johnson, K., Kastelein, R., Yssel, H., and de Vries, J.E. (1991). Interleukin 10 (IL-10) and viral IL-10 strongly reduce antigen-specific human T cell proliferation by diminishing the antigen-presenting capacity of monocytes via downregulation of class II major histocompatibility complex expression. J. Exp. Med. 174, 915–924.
Dubois, S., Patel, H.J., Zhang, M., Waldmann, T.A., and Muller, J.R. (2008). Preassociation of IL-15 with IL-15R alpha-IgG1-Fc enhances its activity on proliferation of NK and CD8+/CD44high T cells and its antitumor action. J. Immunol. 180, 2099–2106.
Fiorentino, D.F., Bond, M.W., and Mosmann, T.R. (1989). Two types of mouse T helper cell. IV. Th2 clones secrete a factor that inhibits cytokine production by Th1 clones. J. Exp. Med. 170, 2081–2095.
Hunter, C.A., Ellis-Neyer, L., Gabriel, K.E., Kennedy, M.K., Grabstein, K.H., Linsley, P.S., and Remington, J.S. (1997a). The role of the CD28/B7 interaction in the regulation of NK cell responses during infection with Toxoplasma gondii. J. Immunol. 158, 2285–2293.
Hunter, C.A., Gabriel, K.E., Radzanowski, T., Neyer, L.E., and Remington, J.S. (1997b). Type I interferons enhance production of IFN-gamma by NK cells. Immunol. Lett. 59, 1–5.
Hunter, C.A., Timans, J., Pisacane, P., Menon, S., Cai, G., Walker, W., Aste-Amezaga, M., Chizzonite, R., Bazan, J.F., and Kastelein, R.A. (1997c). Comparison of the effects of interleukin-1 alpha, interleukin-1 beta and interferon-gamma-inducing factor on the production of interferon-gamma by natural killer. Eur. J. Immunol. 27, 2787–2792.
Kennedy, M.K., Glaccum, M., Brown, S.N., Butz, E.A., Viney, J.L., Embers, M., Matsuki, N., Charrier, K., Sedger, L., Willis, C.R., et al. (2000). Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J. Exp. Med. 191, 771–780.
Lanier, L.L., and Phillips, J.H. (1992). Natural killer cells. Curr. Opin. Immunol. 4, 38–42.
Ma, A., Boone, D.L., and Lodolce, J.P. (2000). The pleiotropic functions of interleukin 15: not so interleukin 2-like after all. J. Exp. Med. 191, 753–756.
Ma, A., Koka, R., and Burkett, P. (2006). Diverse functions of IL-2, IL-15, and IL-7 in lymphoid homeostasis. Annu. Rev. Immunol. 24, 657–679.
Mocellin, S., Panelli, M., Wang, E., Rossi, C.R., Pilati, P., Nitti, D., Lise, M., and Marincola, F.M. (2004). IL-10 stimulatory effects on human NK cells explored by gene profile analysis. Genes Immun. 5, 621–630.
Moore, K.W., O’Garra, A., de Waal Malefyt, R., Vieira, P., and Mosmann, T.R. (1993). Interleukin-10. Annu. Rev. Immunol. 11, 165–190.
Murray, P.J., Wang, L., Onufryk, C., Tepper, R.I., and Young, R.A. (1997). T cell-derived IL-10 antagonizes macrophage function in mycobacterial infection. J. Immunol. 158, 315–321.
Nagalakshmi, M.L., Murphy, E., McClanahan, T., and de Waal Malefyt, R. (2004). Expression patterns of IL-10 ligand and receptor gene families provide leads for biological characterization. Int. Immunopharmacol. 4, 577–592.
Nandi, D., Gross, J.A., and Allison, J.P. (1994). CD28-mediated costimulation is necessary for optimal proliferation of murine NK cells. J. Immunol. 152, 3361–3369.
Neyer, L.E., Grunig, G., Fort, M., Remington, J.S., Rennick, D., and Hunter, C.A. (1997). Role of interleukin-10 in regulation of T-cell-dependent and T-cell-independent mechanisms of resistance to Toxoplasma gondii. Infect. Immun. 65, 1675–1682.
Ortaldo, J.R., and Longo, D.L. (1988). Human natural lymphocyte effector cells: definition, analysis of activity, and clinical effectiveness. J. Natl. Cancer Inst. 80, 999–1010.
Pillet, A.H., Bugault, F., Theze, J., Chakrabarti, L.A., and Rose, T. (2009). A programmed switch from IL-15- to IL-2-dependent activation in human NK cells. J. Immunol. 182, 6267–6277.
Scharton, T.M., and Scott, P. (1993). Natural killer cells are a source of interferon gamma that drives differentiation of CD4+ T cell subsets and induces early resistance to Leishmania major in mice. J. Exp. Med. 178, 567–577.
Scharton-Kersten, T.M., and Sher, A. (1997). Role of natural killer cells in innate resistance to protozoan infections. Curr. Opin. Immunol. 9, 44–51.
Schulz, U., Kreutz, M., Multhoff, G., Stoelcker, B., Kohler, M., Andreesen, R., and Holler, E. (2010). Interleukin-10 promotes NK cell killing of autologous macrophages by stimulating expression of NKG2D ligands. Scand. J. Immunol. 72, 319–331.
Spits, H., Lanier, L.L., and Phillips, J.H. (1995) Development of human T and natural killer cells. Blood 85, 2654–2670.
Sun, R., Fan, J., Wei, H., Zhang, C., and Tian, Z. (2003) Use of interleukin-15 for preparation of adherent NK cells from human peripheral blood: comparison with interleukin-2. J. Immunol. Methods 279, 79–90.
Tripp, C.S., Wolf, S.F., and Unanue, E.R. (1993) Interleukin 12 and tumor necrosis factor alpha are costimulators of interferon gamma production by natural killer cells in severe combined immunode ficiency mice with listeriosis, and interleukin 10 is a physiologic antagonist. Proc. Natl. Acad. Sci. USA 90, 3725–3729.
Waldmann, T.A. (2006). The biology of interleukin-2 and interleukin-15: implications for cancer therapy and vaccine design. Nat. Rev. Immunol. 6, 595–601.
Waldmann, T.A., Dubois, S., and Tagaya, Y. (2001). Contrasting roles of IL-2 and IL-15 in the life and death of lymphocytes: implications for immunotherapy. Immunity 14, 105–110.
Weber-Nordt, R.M., Riley, J.K., Greenlund, A.C., Moore, K.W., Darnell, J.E., and Schreiber, R.D. (1996). Stat3 recruitment by two distinct ligand-induced, tyrosine-phosphorylated docking sites in the interleukin-10 receptor intracellular domain. J. Biol. Chem. 271, 27954–27961.
Yang, A.S., and Lattime, E.C. (2003). Tumor-induced interleukin 10 suppresses the ability of splenic dendritic cells to stimulate CD4 and CD8 T-cell responses. Cancer Res. 63, 2150–2157.
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
Park, J.Y., Lee, S.H., Yoon, SR. et al. IL-15-induced IL-10 increases the cytolytic activity of human natural killer cells. Mol Cells 32, 265–272 (2011). https://doi.org/10.1007/s10059-011-1057-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-011-1057-8