Abstract
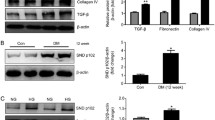
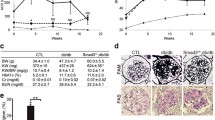
Smad1 has previously been shown to play a key role in the development of diabetic nephropathy (DN), by increasing synthesis of extracellular matrix. However, the regulatory mechanism of Smad1 in DN is still unclear. This study aims to elucidate molecular interactions between activin receptor-like kinase 1 (ALK1)/Smad1 signaling pathway and transcription factor 7-like 2 (TCF7L2) in the progression of DN in vitro and in vivo. The expressions of TCF7L2 and ALK1 were induced by advanced glycation end products (AGEs) in parallel with Smad1, phosphorylated Smad1 (pSmad1), and alpha-smooth muscle actin (α-SMA) through TGF-β1 in cultured mesangial cells. Constitutively active ALK1 increased pSmad1 and α-SMA expressions. The binding of TCF7L2 to ALK1 promoter was confirmed by chromatin immunoprecipitation assay. Furthermore, TCF7L2 induced promoter activity of ALK1. AGEs and TGF-β1 induced a marked increase in TCF7L2 expression in parallel with ALK1. Overexpression of TCF7L2 increased the expressions of ALK1 and Smad1. Inversely, TCF7L2 knockdown by siRNA suppressed α-SMA expression as well as ALK1 and Smad1. The iNOS transgenic mice (iNOS-Tgm), which developed diabetic glomerulosclerosis resembling human diabetic nephropathy, exhibited markedly increased expressions of ALK1, TCF7L2, Smad1, pSmad1, and α-SMA in glomeruli in association with mesangial matrix expansion. These results provide a new evidence that the TCF7L2/ALK1/Smad1 pathway plays a key role in the development of DN.
Similar content being viewed by others
References
Abe, H., Matsubara, T., Iehara, N., Nagai, K., Takahashi, T., Arai, H., Kita, T., and Doi, T. (2004). Type IV collagen is transcriptionally regulated by Smad1 under advanced glycation end product (AGE) stimulation. J. Biol. Chem. 279, 14201–14206.
Bojestig, M., Arnqvist, H.J., Hermansson, G., Karlberg, B.E., and Ludvigsson, J. (1994). Declining incidence of nephropathy in insulin-dependent diabetes mellitus. N. Engl. J. Med. 330, 15–18.
Davies, M. (1994). The mesangial cell: a tissue culture view. Kidney Int. 45, 320–327.
Doi, T., Vlassara, H., Kirstein, M., Yamada, Y., Striker, G.E., and Striker, L.J. (1992). Receptor-specific increase in extracellular matrix production in mouse mesangial cells by advanced glycosylation end products is mediated via platelet-derived growth factor. Proc. Natl. Acad. Sci. USA 89, 2873–2877.
Elbein, S.C., Das, S.K., Hallman, D.M., Hanis, C.L., and Hasstedt, S.J. (2009). Genome-wide linkage and admixture mapping of type 2 diabetes in African American families from the American Diabetes Association GENNID (Genetics of NIDDM) Study Cohort. Diabetes 58, 268–274.
Essawy, M., Soylemezoglu, O., Muchaneta-Kubara, E.C., Shortland, J., Brown, C.B., and el Nahas, A.M. (1997). Myofibroblasts and the progression of diabetic nephropathy. Nephrol. Dial. Transplant 12, 43–50.
Fink, L., Seeger, W., Ermert, L., Hanze, J., Stahl, U., Grimminger, F., Kummer, W., and Bohle, R.M. (1998). Real-time quantitative RT-PCR after laser-assisted cell picking. Nat. Med. 4, 1329–1333.
Finnson, K.W., Parker, W.L., ten Dijke, P., Thorikay, M., and Philip, A. (2008). ALK1 opposes ALK5/Smad3 signaling and expression of extracellular matrix components in human chondrocytes. J. Bone Miner. Res. 23, 896–906.
Florez, J.C., Jablonski, K.A., Bayley, N., Pollin, T.I., de Bakker, P.I., Shuldiner, A.R., Knowler, W.C., Nathan, D.M., and Altshuler, D. (2006). TCF7L2 polymorphisms and progression to diabetes in the Diabetes Prevention Program. N. Engl. J. Med. 355, 241–250.
Gerstein, H.C., Mann, J.F., Yi, Q., Zinman, B., Dinneen, S.F., Hoogwerf, B., Halle, J.P., Young, J., Rashkow, A., Joyce, C., et al. (2001). Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 286, 421–426.
Go, A.S., Chertow, G.M., Fan, D., McCulloch, C.E., and Hsu, C.Y. (2004). Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 351, 1296–1305.
Goumans, M.J., Valdimarsdottir, G., Itoh, S., Rosendahl, A., Sideras, P., and ten Dijke, P. (2002). Balancing the activation state of the endothelium via two distinct TGF-beta type I receptors. EMBO J. 21, 1743–1753.
Grant, S.F., Thorleifsson, G., Reynisdottir, I., Benediktsson, R., Manolescu, A., Sainz, J., Helgason, A., Stefansson, H., Emilsson, V., Helgadottir, A., et al. (2006). Variant of transcription factor 7-like 2 (TCF7L2) gene confers risk of type 2 diabetes. Nat. Genet. 38, 320–323.
Hautmann, M.B., Madsen, C.S., and Owens, G.K. (1997). A transforming growth factor beta (TGFbeta) control element drives TGFbeta-induced stimulation of smooth muscle alpha-actin gene expression in concert with two CArG elements. J. Biol. Chem. 272, 10948–10956.
Inman, G.J., Nicolas, F.J., Callahan, J.F., Harling, J.D., Gaster, L.M., Reith, A.D., Laping, N.J., and Hill, C.S. (2002). SB-431542 is a potent and specific inhibitor of transforming growth factor-beta superfamily type I activin receptor-like kinase (ALK) receptors ALK4, ALK5, and ALK7. Mol. Pharmacol. 62, 65–74.
Isomaa, B., Almgren, P., Tuomi, T., Forsen, B., Lahti, K., Nissen, M., Taskinen, M.R., and Groop, L. (2001). Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 24, 683–689.
Kanematsu, A., Ramachandran, A., and Adam, R.M. (2007). GATA-6 mediates human bladder smooth muscle differentiation: involvement of a novel enhancer element in regulating alphasmooth muscle actin gene expression. Am. J. Physiol. Cell Physiol. 293, C1093–1102.
Kanwar, Y.S., Wada, J., Sun, L., Xie, P., Wallner, E.I., Chen, S., Chugh, S., and Danesh, F.R. (2008). Diabetic nephropathy: mechanisms of renal disease progression. Exp. Biol. Med. 233, 4–11.
Keelan, P.C., Bielak, L.F., Ashai, K., Jamjoum, L.S., Denktas, A.E., Rumberger, J.A., Sheedy, I.P., Peyser, P.A., and Schwartz, R.S. (2001). Long-term prognostic value of coronary calcification detected by electron-beam computed tomography in patients undergoing coronary angiography. Circulation 104, 412–417.
Konig, H.G., Kogel, D., Rami, A., and Prehn, J.H. (2005). TGF-ta1 activates two distinct type I receptors in neurons: implications for neuronal NF-{kappa}B signaling. J. Cell Biol. 168, 1077–1086.
Korinek, V., Barker, N., Morin, P.J., van Wichen, D., de Weger, R., Kinzler, K.W., Vogelstein, B., and Clevers, H. (1997). Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC-/- colon carcinoma. Science 275, 1784–1787.
Kottgen, A., Hwang, S.J., Rampersaud, E., Coresh, J., North, K.E., Pankow, J.S., Meigs, J.B., Florez, J.C., Parsa, A., Levy, D., et al. (2008). TCF7L2 variants associate with CKD progression and renal function in population-based cohorts. J. Am. Soc. Nephrol. 19, 1989–1999.
Kou, I., Nakajima, M., and Ikegawa, S. (2007). Expression and regulation of the osteoarthritis-associated protein asporin. J. Biol. Chem. 282, 32193–32199.
Liu, Z., and Habener, J.F. (2008). Glucagon-like peptide-1 activation of TCF7L2-dependent Wnt signaling enhances pancreatic beta cell proliferation. J. Biol. Chem. 283, 8723–8735.
Luo, R.X., Postigo, A.A., and Dean, D.C. (1998). Rb interacts with histone deacetylase to repress transcription. Cell 92, 463–473.
Lux, A., Attisano, L., and Marchuk, D.A. (1999). Assignment of transforming growth factor beta1 and beta3 and a third new ligand to the type I receptor ALK-1. J. Biol. Chem. 274, 9984–9992.
Lyssenko, V., Lupi, R., Marchetti, P., Del Guerra, S., Orho-Melander, M., Almgren, P., Sjogren, M., Ling, C., Eriksson, K.F., Lethagen, A.L., et al. (2007). Mechanisms by which common variants in the TCF7L2 gene increase risk of type 2 diabetes. J. Clin. Invest. 117, 2155–2163.
Matsubara, T., Abe, H., Arai, H., Nagai, K., Mima, A., Kanamori, H., Sumi, E., Takahashi, T., Matsuura, M., Iehara, N., et al. (2006). Expression of Smad1 is directly associated with mesangial matrix expansion in rat diabetic nephropathy. Lab. Invest. 86, 357–368.
Mima, A., Arai, H., Matsubara, T., Abe, H., Nagai, K., Tamura, Y., Torikoshi, K., Araki, M., Kanamori, H., Takahashi, T., et al. (2008). Urinary Smad1 is a novel marker to predict later onset of mesangial matrix expansion in diabetic nephropathy. Diabetes 57, 1712–1722.
Nakamura, T., Fukui, M., Ebihara, I., Osada, S., Nagaoka, I., Tomino, Y., and Koide, H. (1993). mRNA expression of growth factors in glomeruli from diabetic rats. Diabetes 42, 450–456.
Oh, S.P., Seki, T., Goss, K.A., Imamura, T., Yi, Y., Donahoe, P.K., Li, L., Miyazono, K., ten Dijke, P., Kim, S., et al. (2000). Activin receptor-like kinase 1 modulates transforming growth factor-beta 1 signaling in the regulation of angiogenesis. Proc. Natl. Acad. Sci. USA 97, 2626–2631.
Park, I.S., Kiyomoto, H., Abboud, S.L., and Abboud, H.E. (1997). Expression of transforming growth factor-beta and type IV collagen in early streptozotocin-induced diabetes. Diabetes 46, 473–480.
Parton, L.E., McMillen, P.J., Shen, Y., Docherty, E., Sharpe, E., Diraison, F., Briscoe, C.P., and Rutter, G.A. (2006). Limited role for SREBP-1c in defective glucose-induced insulin secretion from Zucker diabetic fatty rat islets: a functional and gene profiling analysis. Am. J. Physiol. Endocrinol. Metab. 291, E982–994.
Sale, M.M., Smith, S.G., Mychaleckyj, J.C., Keene, K.L., Langefeld, C.D., Leak, T.S., Hicks, P.J., Bowden, D.W., Rich, S.S., and Freedman, B.I. (2007). Variants of the transcription factor 7-like 2 (TCF7L2) gene are associated with type 2 diabetes in an African-American population enriched for nephropathy. Diabetes 56, 2638–2642.
Sanai, T., Sobka, T., Johnson, T., el-Essawy, M., Muchaneta-Kubara, E.C., Ben Gharbia, O., el Oldroyd, S., and Nahas, A.M. (2000). Expression of cytoskeletal proteins during the course of experimental diabetic nephropathy. Diabetologia 43, 91–100.
Schrijvers, B.F., De Vriese, A.S., and Flyvbjerg, A. (2004). From hyperglycemia to diabetic kidney disease: the role of metabolic, hemodynamic, intracellular factors and growth factors/cytokines. Endocr. Rev. 25, 971–1010.
Shimokawa, T., Furukawa, Y., Sakai, M., Li, M., Miwa, N., Lin, Y.M., and Nakamura, Y. (2003). Involvement of the FGF18 gene in colorectal carcinogenesis, as a novel downstream target of the beta-catenin/T-cell factor complex. Cancer Res. 63, 6116–6120.
Shu, L., Sauter, N.S., Schulthess, F.T., Matveyenko, A.V., Oberholzer, J., and Maedler, K. (2008). Transcription factor 7-like 2 regulates beta-cell survival and function in human pancreatic islets. Diabetes 57, 645–653.
Sjogren, M., Lyssenko, V., Jonsson, A., Berglund, G., Nilsson, P., Groop, L., and Orho-Melander, M. (2008). The search for putative unifying genetic factors for components of the metabolic syndrome. Diabetologia 51, 2242–2251.
Takamura, T., Kato, I., Kimura, N., Nakazawa, T., Yonekura, H., Takasawa, S., and Okamoto, H. (1998). Transgenic mice overexpressing type 2 nitric-oxide synthase in pancreatic beta cells develop insulin-dependent diabetes without insulitis. J. Biol. Chem. 273, 2493–2496.
Tang, Y., Urs, S., and Liaw, L. (2008). Hairy-related transcription factors inhibit Notch-induced smooth muscle alpha-actin expression by interfering with Notch intracellular domain/CBF-1 complex interaction with the CBF-1-binding site. Circ. Res. 102, 661–668.
Trembath, R.C., Thomson, J.R., Machado, R.D., Morgan, N.V., Atkinson, C., Winship, I., Simonneau, G., Galie, N., Loyd, J.E., Humbert, M., et al. (2001). Clinical and molecular genetic features of pulmonary hypertension in patients with hereditary hemorrhagic telangiectasia. N. Engl. J. Med. 345, 325–334.
Urness, L.D., Sorensen, L.K., and Li, D.Y. (2000). Arteriovenous malformations in mice lacking activin receptor-like kinase-1. Nat. Genet. 26, 328–331.
Wang, S., and Hirschberg, R. (2009). Diabetes-relevant regulation of cultured blood outgrowth endothelial cells. Microvasc. Res. 78, 174–179.
Warodomwichit, D., Arnett, D.K., Kabagambe, E.K., Tsai, M.Y., Hixson, J.E., Straka, R.J., Province, M., An, P., Lai, C.Q., Borecki, I., et al. (2009). Polyunsaturated fatty acids modulate the effect of TCF7L2 gene variants on postprandial lipemia. J. Nutr. 139, 439–446.
Waterman, M.L. (2004). Lymphoid enhancer factor/T cell factor expression in colorectal cancer. Cancer Metastasis Rev. 23, 41–52.
Weedon, M.N., Owen, K.R., Shields, B., Hitman, G., Walker, M., McCarthy, M.I., Hattersley, A.T., and Frayling, T.M. (2005). A large-scale association analysis of common variation of the HNF1alpha gene with type 2 diabetes in the U.K. Caucasian population. Diabetes 54, 2487–2491.
Wiercinska, E., Wickert, L., Denecke, B., Said, H.M., Hamzavi, J., Gressner, A.M., Thorikay, M., ten Dijke, P., Mertens, P.R., Breitkopf, K., et al. (2006). Id1 is a critical mediator in TGF-betainduced transdifferentiation of rat hepatic stellate cells. Hepatology 43, 1032–1041.
Yamagishi, S., Inagaki, Y., Okamoto, T., Amano, S., Koga, K., Takeuchi, M., and Makita, Z. (2002). Advanced glycation end product-induced apoptosis and overexpression of vascular endothelial growth factor and monocyte chemoattractant protein-1 in human-cultured mesangial cells. J. Biol. Chem. 277, 20309–20315.
Yamamoto, Y., Kato, I., Doi, T., Yonekura, H., Ohashi, S., Takeuchi, M., Watanabe, T., Yamagishi, S., Sakurai, S., Takasawa, S., et al. (2001). Development and prevention of advanced diabetic nephropathy in RAGE-overexpressing mice. J. Clin. Invest. 108, 261–268.
Yao, Y., Zebboudj, A.F., Torres, A., Shao, E., and Bostrom, K. (2007). Activin-like kinase receptor 1 (ALK1) in atherosclerotic lesions and vascular mesenchymal cells. Cardiovasc. Res. 74, 279–289.
Ziyadeh, F.N. (2004). Mediators of diabetic renal disease: the case for tgf-Beta as the major mediator. J. Am. Soc. Nephrol. 15, S55–S57.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Araoka, T., Abe, H., Tominaga, T. et al. Transcription factor 7-like 2 (TCF7L2) regulates activin receptor-like kinase 1 (ALK1)/Smad1 pathway for development of diabetic nephropathy. Mol Cells 30, 209–218 (2010). https://doi.org/10.1007/s10059-010-0109-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-010-0109-9