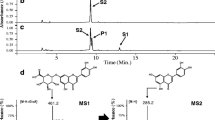
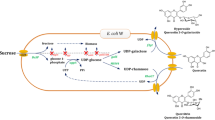
Abstract
Flavonoids are a group of polyphenolic compounds that have been recognized as important due to their physiological and pharmacological roles and their health benefits. Glycosylation of flavonoids has a wide range of effects on flavonoid solubility, stability, and bioavailability. We previously generated the E. coli BL21 (DE3) Δpgi host by deleting the glucose-phosphate isomerase (Pgi) gene in E. coli BL21 (DE3). This host was further engineered for whole-cell biotransformation by integration of galU from E. coli K12, and expression of calS8 (UDP-glucose dehydrogenase) and calS9 (UDP-glucuronic acid decarboxylase) from Micromonospora echinospora spp. calichensis and arGt-4 (7-O-glycosyltransferase) from Arabidopsis thaliana to form E. coli (US89Gt-4), which is expected to produce glycosylated flavonoids. To test the designed system, the engineered host was fed with naringenin as a substrate, and naringenin 7-O-xyloside, a glycosylated naringenin product, was detected. Product was verified by HPLCLC/MS and ESI-MS/MS analyses. The reconstructed host can be applied for the production of various classes of glycosylated flavonoids.
Similar content being viewed by others
References
Asres, K., Seyoum, A., Veeresham, C., Bucar, F., and Gibbons, S. (2005). Naturally derived anti-HIV agents. Phytother. Res. 19, 557–581.
Benavente-García, O., Castillo, J., Marín, F.R., Ortuño, A., and Del Río, J.A. (1997). Uses and properties of Citrus flavonoids. J. Agric. Food Chem. 45, 4505–4515.
Bililign, T., Shepard, E.M., Ahlert, J., and Thorson, J.S. (2002). On the origin of deoxypentoses: Evidence to support a glucose progenitor in the biosynthesis of calicheamicin. ChemBioChem 11, 1143–1146.
Borrelli, F., and Izzo, A.A. (2000). The plant kingdom as a source of anti-ulcer remedies. Phytother. Res. 14, 581–591.
Boue, M.S., Carter-Wientjes, H.C., Shih, Y.B., and Cleveland, E.T. (2003). Identification of flavone aglycones and glycosides in soybean pods by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 991, 61–68.
Bowles, D., Lim, E-K., Poppenberger B., and Vaistij F.E. (2006). Glycosyltransferase of lipophilic small molecules. Annu. Rev. Plant Biol. 57, 567–597.
Brugliera, F., Holton, T.A., Stevenson, T.W., Farcy, E., Lu, C., and Cornish, E.C. (1992). Isolation and characterization of a cDNA clone corresponding to the Rt locus of Petunia hybrida. Plant J. 5, 81–92.
Buslig, B.S., and Manthey, J.A. (2002). Eds; Flavonoids in cell function. (New York, NY: Kluwer Academic/Plenum Publishers).
Cao, G., Sofic, E., and Prior, R.L. (1997). Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships. Free Radic. Biol. Med. 22, 749–760.
Chen, L.J., Games, D.E., Jones, J., and Kidwell, H. (2003). Separation and identification of flavonoids in an extract from the seeds of Oroxylum indicum by CCC. J. Liq. Chromatogr. Relat. Technol. 26, 1623–1636.
Cushnie, T.P.T., and Lamb, A.J. (2005). Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agent. 26, 343–356.
Ferreres, F., Llorach, R., and Gil-Izquierdo, A. (2004). Characterization of the interglycosidic linkage in di-, tri-, tetra- and pentaglycosylated flavonoids and differentiation of positional isomers by liquid chromatography/electrospray ionization tandem mass spectrometry. J. Mass. Spectrum. 39, 1–15.
Fraenkel, D.G. (1968). The accumulation of glucose 6-phosphate from glucose and its effect in an Escherichia coli mutant lacking glucose phosphate isomeraseand glucose 6-phosphate dehydrogenase. J. Biol. Chem. 243, 6451–6457.
He, X.M., and Liu, H-W. (2002). Formation of unusual sugars: mechanistic studies and biosynthetic studies and biosynthetic applications. Annu. Rev. Biochem. 71, 701–754.
Hu, Y., and Walker, S. (2002). Remarkable structural similarities between diverse glycosyltransferases. Chem. Biol. 9, 1287–1296.
Kim, H.P., Son, K.H., Chang, H.W., and Kang, S.S. (2004). Antiinflammatory plant flavonoids and cellular action mechanisms. J. Pharmacol. Sci. 96, 229–245.
Kim, J.H., Kim, B.G., Park, Y., Ko, J.H., Lim, C.E., Lim, J., Lim, Y., and Ahn, J-H. (2006). Characterization of flavonoid 7-O-glucosyltrans-ferase from Arabidopsis thaliana. Biosci. Biotechnol. Biochem. 70, 1471–1477.
Ko, J.H., Kim, B.G., and Ahn, J.-H. (2006). Glycosylation of flavonoids with a glycosyltransferase from Bacillus cereus. FEMS Microbiol. Lett. 258, 263–268.
Kris-Etherton, P.M., Hecker, K.D., Bonanome, A., Coval, S.M., Binkoski, A.E., Hilpert, K.F., and Etherton, T.D. (2002). Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 113, 71–88.
Kris-Etherton, P.M., Lefevre, M., Beecher, G.R., Gross, M.D., Keen, C.L., and Etherton, T.D. (2004). Bioactive compounds in nutrition and health-research methodologies for stablishing biological function: the antioxidant and anti-inflammatory effects of flavonoids on atherosclerosis. Ann. Rev. Nutr. 24, 511–538.
Lee, Y.U., Kawasaki, I., Lim, Y., Oh, W.S., Paik, Y.K., and Shim, Y.H. (2008). Inhibition of developmental processes by Flavone in Caenorhabditis elegans and its application to the pinewood nematode, Bursaphelenchus xylophilus. Mol. Cells 26, 171–174.
Ma, Y.L., Li, Q.M., Van den Heuvel, H., and Claeys, M. (1997). Characterization of flavone and flavonol aglycones by collisioninduced dissociation tandem mass spectrometry. Rapid Commun. Mass Spectrum 11, 1357–1364.
Manach, C., and Donovan, J.L. (2004). Pharmacokinetics and metabolism of dietary flavonoids in humans. Free Radic. Res. 38, 771–785.
Middleton, E., and Kandaswami, C. (1992). Effects of flavonoids on immune and inflammatory cell functions. Biochem. Pharmacol. 43, 1167–1179.
Simkhada, D., Oh, T.J., Pageni, B.B., Lee, H.C., Liou, K.K., and Sohng, J.K. (2009). Characterization of CalS9 in the biosynthesis of UDP-xylose and production of xylosyl-attached hybrid compound. Appl. Microbiol. Biotechnol. 83, 885–895.
So, F.V., Guthrie, A.F., Chambers, A.F., Moussa, M., and Carroll, K.K. (1996). Inhibition of human breast cancer cell proliferation and delay of mammary tumorogenesis by flavonoids and citrus juices. Nutr. Cancer 26, 167–181.
Stobiecki, M. (2000). Application of mass spectrometry for identification and structural studies of flavonoid glycosides. Photochemistry 54, 237–256.
Tahara, S. (2007). A journey of twenty-five years through the ecological biochemistry of flavonoids. Biosci. Biotechnol. Biochem. 71, 1387–1404.
Tanaka, T. (1997). Chemoprevention of human cancer: biology and therapy. Crit. Rev. Oncol. Hematol. 25, 139–174.
Tijburg, L.B.M., Mattern, T., Folts, J.D., Weisgerber, U.M., and Katan, M.B. (1997). Tea flavonoids and cardiovascular diseases: a review. Crit. Rev. Food Sci. 37, 771–785.
Valenzuela, A., Sanhueza, J., and Nieto, S. (2003). Natural antioxidants in functional foods: from food safety to health benefits. Grasas Aceites 54, 295–303.
Yochum, L., Kushi, L.H., Meyer, K., and Folsom, A.R. (1999). Dietary flavonoid intake and risk of cardiovascular disease in postmenopausal women. Am. J. Epidemiol. 149, 943–949.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Simkhada, D., Kim, E., Lee, H.C. et al. Metabolic engineering of Escherichia coli for the biological synthesis of 7-O-xylosyl naringenin. Mol Cells 28, 397–401 (2009). https://doi.org/10.1007/s10059-009-0135-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0135-7