Abstract
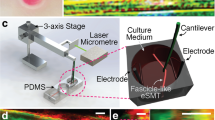
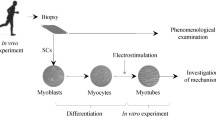
The effects of heat stress on tissue like skeletal muscle have been widely studied. However, the mechanism responsible for the effect of heat stress is still unclear. A useful experimental tissue model is necessary because muscle function in cell culture may differ from native muscle and measuring its contractility is difficult. We previously reported three-dimensional tissue-engineered artificial skeletal muscle (TEM) that can be easily set in a measurement apparatus for quantitative evaluation of contractility. We have now applied TEM to the investigation of heat stress. We analyzed contractility immediately after thermal exposure at 39 °C for 24 or 48 h to evaluate the acute effects and after thermal exposure followed by normal culture to evaluate the aftereffects. Peak twitch contractile force and time-to-peak twitch were used as contractile parameters. Heat stress increased the TCF in the early stage (1 week) after normal culture; the TCF decreased temporarily in the middle to late stages (2–3 weeks). These results suggest that heat stress may affect both myoblast fusion and myotube differentiation in the early stage of TEM culture, but not myotube maturation in the late stage. The TCF increase rate with thermal exposure was significantly higher than that without thermal exposure. Although detailed analysis at the molecular level is necessary for further investigation, our artificial skeletal muscle may be a promising tool for heat stress investigation.







Similar content being viewed by others
References
Giombini A, et al. Hyperthermia induced by microwave diathermy in the management of muscle and tendon injuries. Br Med Bull. 2007;83:379–96.
Moyer HR, Delman KA. The role of hyperthermia in optimizing tumor response to regional therapy. Int J Hyperthermia. 2008;24:251–61.
Giombini A, et al. Short-term effectiveness of hyperthermia for supraspinatus tendinopathy in athletes: a short-term randomized controlled study. Am J Sports Med. 2006;34:1247–53.
Yamaguchi T, et al. Continuous mild heat stress induces differentiation of mammalian myoblasts, shifting fiber type from fast to slow. Am J Physiol Cell Physiol. 2010;298:C140–8.
Lehmann JF, de Lateur BJ. Diathermy and superficial heat, laser and cold therapy. In: Kottke FJ, Lehmann JF, editors. Krusen’s handbook of physical medicine and rehabilitation. Philadelphia: Saunders; 1990. pp. 283–435.
Smith W. The application of cold and heat in the treatment of athletic injuries. In: Michlovitz SL, editor. Thermal agents in rehabilitation. Philadelphia: Davis; 1990. pp. 245–56.
Naito H, et al. Heat stress attenuates skeletal muscle atrophy in hindlimb-unweighted rats. J Appl Physiol. 2000;88:359–63.
Goto K, et al. Responses of muscle mass, strength and gene transcripts to long-term heat stress in healthy human subjects. Eur J Appl Physiol. 2011;111:17–27.
Uehara K, et al. Heat-stress enhances proliferative potential in rat soleus muscle. Jpn J Physiol. 2004;54:263–71.
Nakamura T, et al. Development and evaluation of a removable tissue-engineered muscle with artificial tendons. J Biosci Bioeng. 2017;123:265–71.
Pampaloni F, Reynaud EG, Stelzer EH. The third dimension bridges the gap between cell culture and live tissue. Nat Rev. 2007;8:839–45.
Smith AS, et al. Characterization and optimization of a simple, repeatable system for the long term in vitro culture of aligned myotubes in 3D. J Cell Biochem. 2012;113:1044–53.
Yamasaki K, et al. Control of myotube contraction using electrical pulse stimulation for bio-actuator. J Artif Organs. 2009;12:131–7.
Burke RE, et al. Physiological types and histochemical profiles in motor units of the cat gastrocnemius. J Physiol. 1973;234:723–48.
Desplanches D, et al. Skeletal muscle HSP72 response to mechanical unloading: influence of endurance training. Acta Physiol Scand. 2004;180:387–94.
Sato M, et al. Enhancement of contractile force generation of artificial skeletal muscle tissues by mild and transient heat treatment. Curr Pharm Biotechnol. 2014;14:1083–7.
Zhang M, et al. Autophagy and apoptosis act as partners to induce germ cell death after heat stress in mice. PLoS One. 2012;7:e41412. https://doi.org/10.1371/journal.pone.0041412.
Ohno Y, et. al. Possible role of NF-ĸB signals in heat stress-associated increase in protein content of cultured C2C12 cells. Cells Tissues Organs. 2011;194(5):363–70.
De Maio A. The heat-shock response. New Horiz. 1995;3:198–207.
Kojima A, et al. Heat stress facilitates the regeneration of injured skeletal muscle in rats. J Orthop Sci. 2007;12:74–82.
Rosenblatt JD, Parry DJ, Partridge TA. Phenotype of adult mouse muscle myoblasts reflects their fiber type of origin. Differentiation. 1996;60:39–45.
Acknowledgements
This work was supported by JSPS KAKENHI Grant Numbers JP24500521 and JP16K01383.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takagi, S., Nakamura, T. & Fujisato, T. Effect of heat stress on contractility of tissue-engineered artificial skeletal muscle. J Artif Organs 21, 207–214 (2018). https://doi.org/10.1007/s10047-018-1020-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-018-1020-y