Abstract
Groundwater samples from 22 wells completed in a regional fractured dolostone aquifer in the Guelph region of southern Ontario, Canada, were collected over an 8-month period and analyzed for viruses and Campylobacter jejuni. Only 8% of the 118 samples exhibited viruses at extremely low concentrations, but of the 22 wells sampled, 10 (45%) were positive for human enteric viruses (polyomavirus, adenovirus A, and GII norovirus) including 5 of the 8 public supply wells (62.5%) and 5 of the 11 private wells (45%). Each virus-positive well had only one virus occurrence with six sampling events during the 8-month sampling campaign and only one virus type was detected in each well. The probability of virus detection was positively associated with well open-interval length. Virus concentration (in the wells that were virus-positive) was negatively associated with well depth and open-interval length and positively associated with overburden thickness (i.e., the thickness of unconsolidated materials overlying bedrock facies) and the amount of precipitation 8–14 and 15–21 days prior to the sampling date. The ephemeral nature of the virus detections and the low detection rate on a per sample basis were consistent with previous studies. The percentage of virus-positive wells, however, was much higher than previous studies, but consistent with the fact that the hydrogeologic conditions of fractured bedrock aquifers create wide capture zones and short groundwater travel times to wells making them more vulnerable to contamination occurrence but at very low concentrations.
Résumé
Des échantillons d’eau souterraine de 22 puits réalisés dans un aquifère régional dolomitique et fracturé de la région de Guelph dans le Sud Ontario au Canada, ont été collectés sur une période de 8 mois et analysés pour détecter les virus et la bactérie Campylobacter jejuni. Seuls 8% des 118 échantillons comprenaient des virus à d’extrêmement faibles concentrations. Mais, parmi 22 des puits échantillonnés, 10 (45%) étaient positifs vis-à-vis des virus entériques humains (polyomavirus, adenovirus A et norovirus GII), dont 5 des 8 puits d’approvisionnement public (62.5%) et 5 des 11 puits privés (45%). Chaque puits positif vis-à-vis des virus montrait seulement la présence d’un virus sur les six prélèvements de la campagne d’échantillonnage de 8 mois et seul un type de virus a été détecté dans chaque puits. La probabilité de détection du virus est associée positivement à la longueur de l’intervalle crépiné (ou en trou nu) du puits. La concentration en virus (dans les puits qui étaient positifs vis-à-vis des virus) a été associée négativement avec l’épaisseur de la couverture (i.e., l’épaisseur des matériaux non consolidés recouvrant les faciès de type « substratum ») et la quantité des précipitations sur les périodes de 8 à14 jours et de 15 à 21 jours avant la date de prélèvement. La nature éphémère des détections de virus et le faible taux de détection sur une base « par échantillon » sont cohérents avec les études antérieures. Le pourcentage de puits positifs vis-à-vis des virus est cependant beaucoup plus élevé que lors des études antérieures, mais cohérent avec le fait que les conditions hydrogéologiques des aquifères de substratum fracturé créent de grandes zones de capture et de faibles temps de transit de l’eau souterraine vers les puits, ce qui les rend plus vulnérables à la contamination, mais à de très faibles concentrations.
Resumen
Se recolectaron 22 muestras de agua subterránea en pozos completos en un acuífero fracturado de dolomía en la región de Guelph, en el sur de Ontario, Canadá, durante un período de 8 meses y se analizaron los virus y Campylobacter jejuni. Sólo el 8% de las 118 muestras presentaron virus en concentraciones extremadamente bajas, pero de los 22 pozos muestreados, 10 (45%) fueron positivos para virus entéricos humanos (polyomavirus, adenovirus A y GII norovirus) incluyendo 5 de los 8 pozos de suministro público (62.5%) y 5 de los 11 pozos privados (45%). Cada pozo con virus positivo tuvo sólo una ocurrencia de virus en seis eventos de muestreo durante la campaña de 8 meses de muestreo y sólo se detectó un tipo de virus en cada pozo. La probabilidad de detección de virus se asoció positivamente con la longitud de los intervalos abiertos del pozo. La concentración de virus (en los pozos que fueron positivos para el virus) se asoció negativamente con la profundidad del pozo y la longitud del intervalo abierto y se asoció positivamente con el espesor de la cubierta (es decir, el espesor de los materiales no consolidados sobre las facies del basamento) y la cantidad de precipitación antes de la fecha de muestreo, 8–14 días y 15–21 días. La naturaleza efímera de las detecciones de virus y la baja tasa de detección por muestra fueron consistentes con estudios previos. Sin embargo, el porcentaje de pozos positivos a los virus fue mucho mayor que en los estudios previos, pero consistente con el hecho de que las condiciones hidrogeológicas de los acuíferos rocosos fracturados crean amplias zonas de captura y cortos tiempos de transporte del agua subterránea hacia los pozos, haciéndolo más vulnerables a los procesos de contaminación, pero en muy bajas concentraciones.
摘要
在8个月的期间内,从加拿大安大略省南部Guelph地区一个区域断裂白云岩含水层的22口井中采取了地下水样并对病毒和弯曲杆菌进行了分析。118个样品中,只有8%显示有含量很低的病毒,但在22口采样的井中,10口井(45%)对人类肠道病毒(多瘤病毒、腺病毒A及GII诺瓦克病毒)呈阳性反应,包括8口公共供水井(62.5%)中的5口及11口私人井(45%)中的5口。每一口呈病毒阳性反应的井在8个月采样期间6次采样事件中只有依次出现病毒,每口井中只有一种病毒检出。病毒检出的概率肯定和井的开区间长度有关。(病毒呈阳性的井中)病毒含量和井的深度及开区间程度无关,与覆盖层厚度(也就是覆盖基岩相的松散物质厚度)以及采样日期前8–14天及15–21天的降水量相关。病毒检测的转瞬既逝的特性及基于每个样品的低检出率与先前的研究结果一致。然而,对病毒呈阳性的井百分比比先前研究的结果高许多,但与下列事实一致,即断裂基岩含水层水文地质条件产生的捕获带很宽及地下水到井的行进时间很短致使井更容易受到污染,但污染程度很轻。
Resumo
Amostras de águas subterrâneas a partir de 22 poços concluídos em um aquífero dolomítico fraturado na região de Guelph do sul de Ontario, Canadá, foram coletadas durante um período de 8 meses e analisadas quanto aos vírus e Campylobacter jejuni. Apenas 8% das 118 amostras apresentaram vírus com concentrações extremamente baixas, porém de 22 poços amostrados, 10 (45%) foram positivos quanto aos entéricos humanos (polyomavirus, adenovírus A, e GII norovírus), incluindo 5 de 8 poços de abastecimento público (62.5%) e 5 de 11 poços privados (45%). Cada poço positivo para vírus tinha apenas uma ocorrência de vírus com seis eventos de amostragem durante os 8 meses de campanha de amostragem e apenas um tipo de vírus foi detectado em cada poço. A probabilidade de detecção de vírus foi positivamente associada com o comprimento do intervalo do poço aberto. A concentração de vírus (nos poços que foram positivos para vírus) foi negativamente associada com a profundidade e com o comprimento de intervalo do poço aberto, e positivamente associada com a espessura da sobrecarga (isto é, a espessura dos materiais não consolidados sobrepostos às faces da base rochosa) e a quantidade de precipitação de 8–14 dias e de 15–21 dias antes da data de amostragem. A natureza efêmera das detecções de vírus e a baixa taxa de detecção por amostra foram consistentes com estudos anteriores. A porcentagem de poços positivos para vírus, no entanto, foi muito maior do que estudos anteriores, mas consistente com o fato de que as condições hidrogeológicas dos aquíferos rochosos fraturados criaram amplas zonas de captura e pequenos tempos de deslocamento das águas subterrâneas até os poços, tornando-os mais vulneráveis à ocorrência de contaminação, mas, em concentrações muito baixas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Of the 818 drinking-water outbreaks recorded by the Centers for Disease Control and Prevention in the United States between 1971 and 2008, approximately a third (248) were linked with the consumption of untreated groundwater, and of these, 45 (26.2%) occurred in vulnerable hydrogeologic settings such as karst (Wallender et al. 2014). In Canada, the tragedy in Walkerton, Ontario, where seven people died and more than 2,300 people became ill when Escherichia coli (E. coli) and Campylobacter jejuni contaminated the municipal water supply (O’Connor 2002) drew the attention of Canada to the first issues concerning pathogens in groundwater, particularly in bedrock aquifers. The case study of a norovirus outbreak in Wisconsin (Borchardt et al. 2011) highlights the uniqueness of virus contamination of groundwater in a fractured bedrock aquifer as the virus exhibited rapid transport from a large septic system to a restaurant’s supply well in less than 1 week leading to over 200 individuals becoming infected with norovirus. In these cases and many others, the pathogenic outbreaks resulted from sewage-derived or manure-derived contamination of drinking-water supplies. When combining the results from 15 large-scale studies investigating the occurrence of sewage-derived pathogens and fecal indicators in public water systems, the United States Environmental Protection Agency (US EPA) projected that approximately 23.6% of all public supply wells across the United States will be contaminated with human enteric viruses at some point in time (US EPA 2006).
Lab-scale studies conducted since the late 1970s have determined the various influences on virus inactivation and mobility in natural groundwater systems. To date, it has been shown that virus longevity in groundwater is significantly increased by decreased microbial antagonism (Hurst 1988; Davies et al. 2006), low groundwater temperatures (Yates et al. 1985; Charles et al. 2009), and increased soil moisture content (Yeager and O’Brien 1979). As summarized by Zhang et al. (2012), virus mobility in the subsurface has been shown to occur as colloidal transport which makes the virus particles in fractured media less prone to typical sources of attenuation in porous media such as diffusion, adsorption to grain surfaces, or size and charge exclusion effects, and therefore promotes virus particles to exhibit transport speeds on the order of calculated average linear groundwater velocities assuming fracture porosities for rock. As effective porosities on the order of 10−3 to 10−5 in fractured rock promote large groundwater velocities (Freeze and Cherry 1979) and colloids have shown transverse dispersion coefficients up to 4 orders of magnitude greater than those of the solutes (Weisbrod et al. 2013), virus transport in fractured bedrock aquifers can be both rapid and widely distributed in a matter of days. While sediments overlying fractured bedrock aquifers are expected to offer some attenuation of viruses before they enter the aquifer, the predominant sources of enteric viruses are leaking sewers and septic systems that are commonly buried several meters below ground surface and in close proximity to the top of rock or down in trenches dug into the rock to maintain slopes for gravity flow. With shallow water tables and cool temperatures promoting virus longevity up to and potentially longer than 1 year (Charles et al. 2009) and groundwater velocities on the order of 10 m/day typical of fractured dolostone aquifers (Belan 2010), active viruses can potentially be transported several kilometers from a single source while maintaining their ability to infect a host. Despite the potential for rapid and widespread distribution of viruses in groundwater, to date, the number of studies investigating the presence of actual viruses in fractured bedrock aquifers is extremely limited (Table 1).
In the first studies investigating the transport of viruses through fractured bedrock, Borchardt et al. (2007) and Bradbury et al. (2013) demonstrated the vulnerability of the Mount Simon sandstone aquifer to virus contamination through the regionally extensive Eau Claire shale aquitard. In the 2007 paper, Borchardt et al. conducted monthly sampling of three public water supply wells in Madison WI to reveal the presence of human enteric viruses in two wells that were cased through the Eau Claire aquitard and open to the underlying confined sandstone aquifer. Similarly, Bradbury et al. (2013) sampled six wells all of which exhibited virus detections. The studies ultimately revealed that, while failed well casings could act as cross-connections from the upper unconfined aquifer to the lower confined aquifer, simultaneous detection of viruses in wells several kilometers apart would require several well casings to fail at the same time. As this scenario was unlikely, it was confirmed that preferential flow paths from the upper Wonewoc aquifer through the Eau Claire aquitard were likely present in the form of naturally occurring fractures and acted as the means for virus transport to the lower confined aquifer, therefore highlighting the fact that not even confined bedrock aquifers were impervious to virus contamination. While these studies confirmed the vulnerability of fractured bedrock aquifers to virus contamination, the number of wells and therefore type of wells investigated were relatively low.
In two studies conducted by Davis and Witt (2000) and Femmer (2000), over 200 Missouri municipal supply wells completed in the carbonate and karstic carbonate aquifers of the Ozark Plateaus were sampled for human enteric viruses. While the large array of wells exhibited varying constructions, ages, and surrounding land use and therefore allowed for the evaluation of the various factors contributing to virus contamination, each of the wells was only sampled 1 or 2 times. In Bradbury et al.’s 2013 study, the importance of temporal sampling is highlighted as even wells highly prone to virus contamination did not always exhibit viruses throughout over a year of monthly sampling events (the well with the highest number of positive virus detections had detections during only 61% of sampling events). Other studies sampling a wide array of wells such as Lindsey et al. (2002) and Johnson et al. (2011) also completed a limited number of sampling events at each of their wells and so detection rates are not representative of the actual vulnerability of the wells or aquifers. Studies including temporal sampling such as those conducted by Lieberman et al. (2002), Borchardt et al. (2003), and Locas et al. (2007), therefore, provide better insight with regard to the true vulnerability of a well completed in a fractured bedrock aquifer. In the study by Lieberman et al. (2002), temporal sampling of 13 wells completed in karstic bedrock was conducted over an entire year and found that 43% of the wells exhibited culturable viruses. In the study by Locas et al. (2007), the only known field-scale studies of viruses in Canadian groundwater, both of the bedrock wells that were sampled also exhibited viruses. While both of these studies produced temporal data, Lieberman et al. only sampled wells that had previously recorded contamination with microbial water quality indicators such as E. coli and Locas et al. (2007) sampled only a limited number of bedrock wells. In comparison, the investigation by Borchardt et al. (2003) conducted temporal sampling of a wide array of wells in a large variety of settings. Although Borchardt et al. (2003) temporally sampled 30 wells completed in bedrock and revealed that 10% of them were susceptible to virus contamination, all the wells were private wells and so no estimate for larger municipal supply wells was made. Despite the fact that each of the studies summarized in Table 1 sampled wells completed in fractured rock, a single, large-scale investigation focusing on the temporal susceptibility of various types of wells completed solely in fractured bedrock aquifers and the numerous factors contributing to their vulnerability has not been reported in the literature.
The goal of the current study is to assess the vulnerability of the regional fractured bedrock aquifer supplying drinking water to the residents and communities in southern Wellington County, Ontario to contamination with enteric viruses and determine the factors that promote this susceptibility. Over an 8-month period, 22 private, municipal supply, and monitoring wells situated in the Silurian dolostone aquifer were sampled monthly for enteric viruses including five human viruses (enterovirus, hepatitis A, norovirus genogroups I and II, polyomavirus, and adenovirus), bovine polyomavirus, and influenza A virus. To provide the study with several types of scenarios that are typical to both urban and rural settings, each of the wells chosen to be sampled exhibited different characteristics with regards to well completion depths and pumping rates, with variable sediment types and thicknesses, and proximity to different potential sources such as agriculture, surface-water bodies, sanitary sewers, and septic systems. As this study area is covered with glacially derived sediments with varying degrees of thickness and permeability in addition to the cool groundwater temperatures and rapid groundwater velocities on the order of 1–10 m/day (Belan 2010), it is expected that all well types completed in this fractured Silurian dolostone aquifer will be vulnerable to contamination with animal and human sewage-derived enteric viruses.
Materials and methods
Description of study area
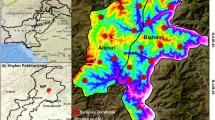
The array of 22 sampling wells (Table 2) is located approximately 100 km west of Toronto, Ontario, Canada in various urban and rural communities of southern Wellington County (Fig. 1). The area surrounding the City of Guelph (population: 130,000) is dominated by agriculture and small towns. Of the 2,511 farms located in Wellington County in 2011, there were 363 dairy cattle and milk production farms, 423 beef cattle ranching and farming farms, 120 hog and pig farms, and 166 poultry and egg farms, making Wellington County one of the largest agricultural hubs in Ontario. These farms host a total of 142,197 cattle and calves, 236,144 pigs, 5,706,394 hens and chickens, and 248,811 turkeys. To the south of Guelph are two agricultural and residential areas, Arkell and Aberfoyle (combined population: 7,000) and to the northeast is Eden Mills (population: 350) built along the banks of the Eramosa River. Northwest of Guelph are Fergus (population 19,000) and Elora (population: 4,000), the two largest communities within Centre Wellington Township. As Guelph, Fergus, and Elora are larger communities, they have both public water distribution systems and sanitary sewers serving the majority of their populations with septic systems and private wells on the outskirts of the villages and rural areas. Despite the availability of municipal drinking water works in the older sections of town, many residents of Fergus and Elora still rely on private wells and septic systems even within highly populated centres of the communities. The populations of Arkell, Aberfoyle, and Eden Mills are entirely reliant upon private wells and septic systems.
a–b Sampling locations in the City of Guelph, Ontario, Canada, and several surrounding communities within Wellington County. c Bedrock geology of Wellington County, which is dominated by a Silurian dolostone belt extending from Lake Huron to the Niagara River. All sampling wells are constructed into the Lower Silurian carbonate formations including the Guelph, Eramosa, Goat Island, and Gasport formations. d Locations of sampled wells
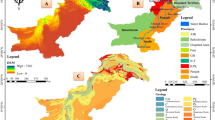
Although all of the communities chosen for this investigation are entirely encompassed by the 6,800 km2 of the Grand River watershed, the belt of Silurian dolostone that subcrops beneath the glacial sediments along the Niagara Escarpment and extends from Lake Huron/Georgian Bay to Niagara Falls, Ontario serves as a regionally important aquifer for nearly 800,000 people in southern Ontario including over 200,000 residents of Wellington County, and therefore, groundwater is the primary source of drinking water in the study area. All of the wells chosen for this investigation were completed in the Lower Silurian sequences as described by Brunton (2009) and consist of the Guelph, Eramosa, Goat Island, Gasport, Irondeqouit, Rockway and Merritton formations and regionally bounded by the underlying Cabot Head shale Formation. Both the Guelph and Gasport formations act as the predominant fractured bedrock aquifers in the region. In the study area, the Guelph Formation is characterized by crinoidal grainstones and wackestones, is generally 15–22 m thick, and exhibits a sharp basal contact with the shaley facies and crinoidal grainstones of the underlying Eramosa and Goat Island formations, respectively. Although the Eramosa and Goat Island are water-bearing formations, their relatively lower transmissivities cause them to be considered regional aquitards (Golder Associates 2006). When vertical fractures and preferential pathways in the form of cross-connecting boreholes are not present, the Eramosa and Goat Island formations can limit the transport of some surface-derived contaminants present in the unconfined Guelph aquifer to the underlying Gasport aquifer. The thickness and presence of the Eramosa and Goat Island formations, however, are highly variable in the study area and these formations are sometimes entirely absent (Belan 2010). The 25–70 m of the blue-grey crinoidal grainstones of the Gasport Formation can therefore be in direct contact with the overlying Guelph Formation, which in turn is overlain by various types of glacial deposits that potentially offer a source of attenuation of surface contaminants before they enter the rapid flow regime of the underlying fractured bedrock aquifers. The presence and thickness of both of the Port Stanley and Wentworth Tills is highly variable in the study area throughout southern Wellington County (Fig. 3b,c) and studies investigating the vulnerability of the underlying fractured bedrock aquifers have established that the varying thicknesses and degrees of permeability of these sediments result in the aquifers being highly susceptible to surface contamination (Lake Erie Region Source Protection Committee 2012).
Selection of wells for sampling
The 22 wells sampled during this investigation (Table 2; Fig. 2) consisted of existing private (PW), municipal supply (MSW), and monitoring (MW) wells completed in the local fractured dolostone aquifers within a 20-km radius of the City of Guelph. Each of the wells was chosen in order to represent the range of well construction and hydrological conditions that might affect the vulnerability of typical bedrock wells to virus and bacterial contamination and the variety of well constructions and pumping volumes common to the study area. Surficial geology data were obtained from Ontario Ministry of Northern Development and Mines (2013). Final selections were based on seven criteria: (1) well type, i.e., private, municipal supply, or monitoring well; (2) well completion, including total depth, length of open interval, and bedrock aquifer segment; (3) the well’s proximity to potential sources such as sanitary sewers, septic systems, animal agriculture operations, or surface water bodies; (4) the spatial distribution across the study area and flow system; (5) surrounding land use, i.e., urban versus rural;( 6) overburden thickness (i.e., the thickness of unconsolidated materials overlying bedrock facies); and (7) accessibility/permission.
Cross-section of sampling wells located across southern Wellington County illustrating well completions and bedrock geology. Although several of the wells are open in multiple formations, some are cased into a single hydrogeologic unit. MW1, PW1, PW3, and PW5 are solely open to the Guelph Formation, with casing being set into the top of rock. Wells MW2 and PW6 are open to the Eramosa Formation, while MW3, PW11, and PW10 are open only to the Gasport Formation. a Centre Wellington sampling wells. b Aberfoyle sampling wells. c City of Guelph sampling wells. d Arkell sampling wells. e Eden Mills sampling wells
Using these criteria alongside the Ontario Ministry of the Environment’s Well Record Database, 17 wells in the towns of Fergus, Elora, Arkell, Eden Mills, and Aberfoyle were chosen to be sampled. In addition to the aforementioned criteria and the Well Record Database, sewer invert elevations in Guelph were plotted relative to the top of bedrock (Fig. 3) and used to select five wells in areas where sewers were constructed into or near the top of bedrock. In the end, a total of 22 wells including 11 private wells, 8 municipal supply wells, and 3 monitoring wells were chosen to be sampled. The characteristics of each well are summarized in Table 2 and so only a brief summary of the wells will be presented here.
In Centre Wellington, six municipal supply wells were sampled, including three wells in Elora and three wells in Fergus. Each well was cased through the overburden sediments into the upper meter or so of the Guelph Formation. The wells were therefore open from the top of the Guelph Formation to varying depths within the Gasport Formation. In contrast to the wells sampled in Centre Wellington, those sampled within the City of Guelph did not consist of any actual municipal supply wells but two large capacity water supply wells operated by the University of Guelph and three monitoring wells. The three 2-inch (5 cm) diameter monitoring wells with varying completion depths are located along the Speed River at a site of known industry-derived contamination with various volatile organic compounds and within 10 m of the main trunk line of Guelph’s municipal sanitary sewer system that was constructed below the local water table and up to 4 m below the top of rock. The two University of Guelph water supply wells located within the centre of the City of Guelph are both 10-inch (25 cm) diameter wells and are pumped at an approximate rate of 280 L min–1. For the purpose of this study, these wells are considered comparable to municipal supply wells and are labeled as such.
Each of the 11 private wells was located in a small residential or agricultural community outside the City of Guelph and had a unique well completion. In the small Puslinch communities of Arkell and Aberfoyle, four and five private wells of varying completion depths were sampled, respectively. The remaining wells added to the study were two private wells of different total depths and completion years located along the Eramosa River in Eden Mills.
Sampling methods
Sampling began in the second week of June 2012 and extended until January 2013. Each of the wells was sampled once for general chemistry and several times for enteric pathogens within the 8-month sampling period. Most wells were sampled monthly for 6 months, while other wells were sampled less frequently due to limited accessibility. Temporal sampling for viruses is required as previous studies have shown virus concentrations in groundwater to be extremely variable (Bradbury et al. 2013). During virus sampling events in July and November 2012, samples for bacterial fecal indicators, E. coli and total coliforms, were also collected.
For the current study, all virus samples were collected by following a standard operating procedure (SOP) developed in accordance with the procedures outlined in Millen et al. (2012). Prior to sampling, all equipment set to come in contact with raw well water was soaked in a 0.52% NaClO solution for 30 min prior to being rinsed with 0.05 M sodium thiosulfate for 5 min. All hoses and tubes were then flushed with three volumes of DI water and covered with parafilm for transport to the sampling site. Samples from municipal supply wells were taken directly from raw water taps within each of the well houses, whereas private wells were sampled from external faucets bypassing water treatment such as filtration units or water softening systems. Monitoring wells were sampled using a Grundfos Redi-Flo2 electrical submersible pump. For all three well types, sampling hoses were connected to a flow-through cell with a YSI 556 multiprobe system to monitor field parameters including pH, temperature, and conductivity during purging. Once these parameters were constant, glass wool filters (Lambertini et al. 2008) were attached to well taps and faucets by sterile half-inch tubing. At one site known to have VOC contamination, filtered water was collected into 55-gal drums and treated at an onsite treatment centre. To maximize virus adsorption to glass wool well water pH levels ≥ 7.5 were adjusted to between 6.5 and 7.0 by injecting 0.5 M HCl before the glass wool filter using a Masterflex precision peristaltic pump. Four of the 22 study wells required pH adjustment during sampling. Mean sample volume was 877 L ± 210 l (±1 SD), range 483 –9,16 L, n = 118.
Laboratory methods
Glass wool filters were packed on ice and shipped overnight to the USDA Agriculture Research Service in Marshfield, WI for analysis using the procedures outlined by Borchardt et al. (2012). Immediately upon arrival glass wool filters were eluted using 3% beef extract containing 0.05 M glycine (pH 9.5) in the direction opposite of sample flow to maximize collection of bacterial and viral targets (Abd-Elmaksoud et al. 2014). Eluate pH was adjusted to 7.0–7.5 using 1 M HCl and flocculated with polyethylene glycol 8000 (8% wt/vol) and NaCl (final concentration, 0.2 M). This mixture was stirred for 1 h, incubated overnight at 4 °C, and centrifuged at 4200 × g for 45 min. The pellet was resuspended in 2 ml of sterile 0.15 M N2HPO4 and the final concentrated sample volume stored at −80 °C.
Samples were analyzed by qPCR on a LightCycler 480 (Roche Diagnostics, Mannheim, Germany) for six human viruses (adenovirus, enterovirus, norovirus genogroups I and II, hepatitis A virus, and polyomavirus) bovine polyomavirus, influenza A virus, and C. jejuni following the reverse transcription and PCR procedures described in Borchardt et al. 2012. Primers (IDT, Coralville, IA) and hydrolysis probes (TIB Molbio, Berlin, Germany) are reported in Table 3. PCR inhibition was measured for every sample following the methods of Gibson et al. (2012). Among 118 samples, six were PCR-inhibited; inhibition was mitigated by 1:5 dilution with nuclease-free water. No-template controls were included for every batch of analysis steps: nucleic acid extraction, reverse transcription, and PCR. All no-template controls were negative—i.e., no cycle quantification (Cq) value. Performance parameters of the standard curves are reported in Table 4. Standards consisted of g-blocks (targets > 125 base pairs) and ultramers (targets ≤ 125 base pairs) both supplied by IDT. To facilitate identifying amplicon contamination from the standards, the sequence between primer and probe was scrambled while maintaining the wildtype base composition. One standard of each target with a Cq value near 30 was used as the positive control for every PCR analysis batch.
Glass wool filter performance was evaluated twice with samples collected from sites MW1 and MW3. Poliovirus Sabin type 3 was seeded into the samples and recovery measured following the methods described in Lambertini et al. (2008). Percent recovery for sites MW1 and MW3 were 53 and 39%, respectively, both which fall within the recovery range for enteroviruses concentrated by glass wool filtration (Lambertini et al. 2008).
Collection of groundwater samples for E. coli and total coliforms closely followed the guidelines set forth by US EPA Method 1604 (2002). As groundwater concentrations of E. coli and total coliforms were expected to be low, sample volumes were increased from the suggested 100 to 1,000 ml. Groundwater samples for bacterial analyses were collected in triplicate into autoclaved 1-L HDPE bottles and transported on ice to the University of Guelph.
Samples for E. coli and total coliform analyses were analyzed within 24 h of their collection using vacuum filtration following EPA Method 1604 (2002). Each groundwater sample was filtered onto pre-sterilized 47-mm diameter, grid-marked, cellulose ester membranes and placed on Oxoid CM 1038 Differential Coliform (DC) agar. After 24 h of incubation at 35 °C, the plates were removed and checked for bacterial growth. Pink colonies were counted as total coliforms and blue colonies were counted as E. coli. The plates were incubated for an additional 24 h followed by another round of colony counting. Blank samples were run between each filtration for quality control.
Data analysis
The association between well characteristics and virus levels was initially evaluated by Fisher’s exact test on a 4 × 2 table where the rows were categories of overburden type (gravel, diamicton, sand, none) and the columns represented the dichotomous response (virus detected versus virus not detected). The same analytic procedure was then undertaken separately for each of three continuous-scaled characteristic variables (total depth, overburden thickness, open interval length) to examine their relationship with virus levels. First, the response was treated as dichotomous and a parametric log-binomial regression model was fit to estimate the probability of virus detection across the observed range of values for the well characteristic variable (McNutt et al. 2003). To determine the appropriate representation (linear, quadratic, etc.) of the well characteristic in the log-binomial model, a preliminary step was undertaken where a graphic derived from a nonparametric generalized additive model (GAM) was examined (Hastie and Tibshirani 1990). In the second analysis for each continuous-scaled well characteristic, the data were restricted to wells where virus was detected and robust regression techniques were used to evaluate the association between the characteristic and continuous-scaled virus concentration levels (Huber 1973).
Precipitation data were acquired from the Guelph Turfgrass Institute and the Elora Research Station, University of Guelph. For each date on which sampling took place, precipitation levels were tabulated at lags of 0–7, 8–14 and 15–21 days prior to the sampling date; analyses were carried out separately for the three lag categories. The unit of analysis was a sampling date, and the same analytic approach as described above (GAM, log-binomial regression, robust regression) was used to examine the relationship between precipitation level and virus detection/concentration, except that a different robust regression technique was employed to accommodate the specific characteristics of the data (Yohai 1987). All analyses were performed in SAS 9.4 (SAS Institute Inc., Cary, NC).
Results
Of the 22 wells sampled in southern Wellington County, 10 were positive for human enteric viruses, ranging in concentrations from 0.1 to 16 genomic copies per L (Table 5). Five of these wells were private wells, while the other five were municipal supply wells. Five out of 11 (45%) private wells that were sampled exhibited virus detections, while 5 out of 8 (62%) municipal supply wells were virus-positive. The three viruses that were detected included human polyomavirus, adenovirus A, and GII Norovirus (Fig. 4). None of the positive wells tested positive on more than one occasion nor did they exhibit more than one type of virus.
The association between type of overburden at the well locations and virus detection was marginally significant (p = 0.09), where more viruses were detected in gravel and diamicton. Overburden thickness and well depth were not significantly associated with the probability of detecting viruses. The estimated relationship in both cases indicated somewhat of a threshold effect (Fig. 5); in the log-binomial model for over-burden thickness one outlier was excluded, a well with overburden thickness of 78.9 m and no viruses detected. In the wells that were virus-positive, overburden thickness was positively associated with virus concentration (slope = 0.0965, p = 0.029) and a significant negative association was present for well depth (slope = −0.0199, p = 0.005; Fig. 5).Wells with longer open intervals were more likely to be virus-positive (p = 0.044), and virus concentration was negatively associated with open interval length in wells where virus was detected (slope = −0.0177, p = 0.028; Fig. 5).
Association of three well construction characteristics (overburden thickness, well depth, and open interval length) with the probability of virus detection (left-panel plots: a–c) and virus concentrations (right-panel plots: d–f). The evaluation of virus concentrations was restricted to the subset of wells that were virus-positive. Blue-shading represents the 95% confidence limits
Precipitation amount was not significantly associated with the probability of virus detection for any of the three lag periods evaluated (i.e., cumulative precipitation within 0–7, 8–14 and 15–21 days prior to the sampling date). The estimated relationships for the lag periods assumed different shapes but generally reflected positive associations (Fig. 6). On sampling dates with ≥1 virus-positive well (n = 7), virus concentration was positively associated with cumulative precipitation amounts in the periods 8–14 days (slope = 0.1690, p = 0.033) and 15–21 days before sampling (slope = 0.2127, p = 0.012; Fig. 6). The relationship between virus concentration and the amount of precipitation 0–7 days prior to sampling was positive but not significant (slope = 0.0916, p = 0.457; Fig. 6).
Association of cumulative precipitation during the periods 0–7 days; 8–14 days; and 15–21 days prior to the sample collection date with the probability of virus detection (left-panel plots: a–c) and virus concentrations (right-panel plots: d–f). The evaluation of virus concentrations was restricted to the subset of sampling dates with ≥1 virus-positive well. Blue-shading represents the 95% confidence limits
E. coli was detected in six out of the 22 wells (27%) sampled in this investigation including 1 municipal supply well, 2 monitoring wells and 3 private wells. All six wells with detectable E. coli would be above the Canadian Drinking Water Guideline of No Detections 100 m L−1 for non-disinfected groundwater. While two wells were positive for E. coli during both sampling events, the other four positive wells only had detectable E. coli in one of the sampling events. The presence of E. coli was not always an indicator of virus detection. Only one well that was positive for human polyomavirus also contained detectable levels E. coli; additionally, the two wells exhibiting the highest concentrations of E. coli did not exhibit any viruses.
Total coliforms were detected in 11 out of 22 wells (50%) including 3 municipal supply wells, 2 monitoring wells, and 6 private wells. Total coliform concentrations were significantly higher than E. coli with 2 private wells exhibiting too many colony-forming units to count. Of the 11 wells to exhibit positive detections of total coliforms, five were also positive for human viruses, including human polyomavirus (3) and adenovirus A (2).
Discussion
Vulnerability of the dolostone aquifer
Previous assessments of the Grand River watershed suggested that the dolostone aquifers of the Guelph area exhibit a medium–to–high vulnerability to contaminants introduced at the ground surface due to the combination of permeable overburden with spatially variable thicknesses and the highly fractured nature of the bedrock aquifers (Lake Erie Region Source Protection Committee 2012). The detection of viruses in these aquifers is, perhaps, therefore not surprising. This 2012 assessment, however, is not specific to pathogen contaminants and has inconsistencies where vulnerability is discussed for this fractured rock aquifer where fracture porosities have been estimated to be in the 10−4 range (Belan 2010; Munn 2012) and yet the report used effective porosities of 10−1 (1%) for estimating the 2-year well-head-protection areas, a reasonable estimate of virus longevity in Guelph area groundwater. In comparison to other studies, the detection of viruses in 10 out of the 22 wells sampled in this investigation is the highest detection rate of human enteric viruses in groundwater samples. On a per sample basis, however, only 10 of 118 virus samples exhibited detectable concentrations of viruses. While the ephemeral nature of virus contamination has been well established in the literature, the low per sample detection frequency was unexpected for the rapid flow regime and highly vulnerable setting of the fractured bedrock aquifers of Guelph, Ontario; furthermore, when viruses were detected, the concentrations were consistently very low (1–16 genomic copies L−1) in comparison to typical values found in wastewater (104–106 genomic copies L−1; Bradbury et al. 2013). This suggests strong dilution due potentially to large well capture zones creating large amounts of mixing and/or weak inputs from sources during the study period.
Vulnerability of private wells
In the current investigation, five out of the 11, or 45% of private wells sampled, exhibited human enteric viruses including human polyomavirus, adenovirus A, and GII norovirus. These findings suggest that private wells completed in the fractured Silurian dolostone aquifer of the greater Guelph area are several times more vulnerable to virus contamination than private wells in Wisconsin where the only other study that had investigated virus contamination of private wells completed in fractured bedrock aquifers has been conducted (Table 6; Borchardt et al. 2003). The difference in well contamination rates between the two studies is likely not attributed to well depth, casing depth, sample volume, groundwater pH, or groundwater temperature as these parameters were similar between the two studies. Well age was much younger in the study by Borchardt et al. (2003), but well age in the current study was not associated with detection rates. Two key differences between the studies are the types of fractured bedrock aquifers investigated and the sampling frequency. In the current study, dolostone aquifers were sampled five or six times over eight consecutive months. Borchardt et al. (2003) also examined igneous and sandstone aquifers, and sampled each private well once per quarter over a calendar year. Virus contamination in wells is highly transient (Bradbury et al. 2013); therefore, the increased frequency of sampling on a monthly interval in this study may have contributed to a higher detection rate.
Vulnerability of municipal supply wells
The municipal supply wells of the study area also had detectable levels of viruses. Relying on 15 studies of viruses in groundwater, the US EPA (2006) estimated that a randomly selected municipal supply well in the United States had a 23.6% probability of virus contamination during its operational lifetime. In the current study, five out of eight (62%) large capacity supply wells exhibited viruses during the 8-month sampling period. Note, the EPA’s estimate relied on cell culture techniques for virus detections, whereas the current study relied on qPCR, which tends to yield higher virus contamination rates compared to cell culture because it can detect inactivated viruses and a greater number of virus types.
Other studies have investigated the occurrence of viruses in municipal supply wells completed in fractured bedrock aquifers (summarized in Table 1). While the studies in the Ozark Plateaus (Davis and Witt 2000; Femmer 2000) and Pennsylvania (Lindsey et al. 2002) sampled a wide array of wells, the virus detection rates are suspected of being low due to insufficient sampling frequencies of each well. In contrast, studies by Lieberman et al. (2002), Borchardt et al. (2007), Locas et al. (2007), and Bradbury et al. (2013) each collected monthly samples in order to account for the temporal variability and short term occurrence of viruses in groundwater. The well contamination rate for viruses in the current study, 62%, is on the high end of the range reported for the previous nine North American studies.
Groundwater flow systems influences on virus detection
The fractured Silurian dolostone aquifer in this study area (southern Wellington County, Ontario) are more susceptible to virus contamination and mobilization than aquifers composed of porous media. The individual grains of porous media aquifers provide greater surface area and thus more sites available for virus adsorption and attenuation compared to fractured bedrock aquifers. Additionally, groundwater velocities within porous media are much slower and may result in virus inactivation before transport has been completed from the source to the vulnerable well.
Belan (2010) and Munn (2012) investigated the hydraulic properties of the Guelph and Gasport formations that are the main water-supply aquifer in southern Wellington County, Ontario using various combinations of hydraulic tests and core logging techniques. Using FLUTe transmissivity profiling (Keller et al. 2014) in conjunction with ALS temperature logging (Pehme et al. 2010), packer tests (Quinn et al. 2011), and a 13-day pumping test, Belan (2010) estimated that the transmissivity of the bulk aquifer (i.e., the Guelph and Gasport formations combined) ranged from 2.5 × 10−5 to 6 × 10−4 m2 s−1 and that fracture apertures ranged from 9 to over 430 μm with a mean of 69 μm. At the same study site, Munn (2012) completed a more detailed 3-D characterization of the fracture network using a suite of high-resolution, depth-discrete data collection methods including core logging, borehole geophysics (acoustic televiewer), and hydraulic testing (FLUTe transmissivity profiling) in 2 inclined coreholes and several nearby vertical coreholes. This yielded estimates of transmissivity between 2.0 × 10−4 to 4.2 × 10−4 m2 s−1 and fracture apertures between 15 and 407 μm with a slightly larger geometric mean between 104 and 159 μm in comparison to the estimates made by Belan. While Belan estimated an effective fracture porosity of the bulk aquifer on the order of 5 × 10−4, Munn suggested the mean effective fracture porosity of the Guelph formation is approximately 1 × 10−3 and between 3 × 10−4 and 5 × 10−4 for the Gasport Formation. Such low effective fracture porosities are what lead to rapid groundwater velocities and transport times associated with fractured bedrock aquifers. As estimated by Belan (2010), the hydraulic conductivity and groundwater velocities of the fractured Silurian dolostone aquifer of southern Wellington County range from 6 × 10−6 m/s to 9 × 10−6 m s–1 and from 0.002 to 28 m day–1 under natural gradients, respectively. With fracture apertures 103–104 times larger than virus particles, groundwater hydraulic conductivities and velocities promoting transport up to 10 km in a single year, limited virus adsorption due to the limited surface area of fracture apertures and cool groundwater temperatures that favor virus survival, the intrinsic vulnerability of wells completed in fractured bedrock aquifers is much higher than wells completed in porous media aquifers regardless of well construction and surrounding land use.
The type of overburden material will likely influence virus attenuation and transport to the underlying aquifer. In this investigation, virus detections were greatest in wells surrounded by sediments composed of gravel or diamict, consistent with the rapid groundwater velocities characteristic of these sediments. Although a well may be located in an area of tight diamict and therefore relatively low permeability, the high mud content makes it susceptible to having fractures, providing preferential pathways for rapid groundwater flow and transport of viruses and bacteria from sources (Cherry et al. 2006), therefore increasing the well’s vulnerability to contamination.
Overburden thickness was associated with virus concentration although, contrary to expectations, the association was positive. It was expected that the greater attenuating properties of thicker unsaturated overburden sediments would result in decreased virus concentrations in the underlying fractured bedrock aquifer. The positive correlation could be due to an unknown confounder or spurious from the small number of positive samples. In addition, overburden thickness at the well head (as was determined in this study) may not truly reflect well vulnerability as fractured bedrock aquifers are characterized by large capture zones that could have a contaminant source a substantial distance from the well. Assessing the importance of overburden thickness within entire capture zones and not just at the well head could lead to a better understanding of well vulnerability to virus contamination in fractured bedrock aquifers.
Open interval length of the wells was positively associated with the probability of detecting viruses. Wells with longer open intervals are more likely to capture more groundwater flow paths, increasing the likelihood that one flow path will include a virus source upgradient.
Open interval length and well depth were negatively associated with virus concentration. As most of the wells were only cased through the overburden and left open from the top of rock to the base of the well, deeper wells had longer open intervals. Well depth was indeed highly correlated with open interval length (Pearson and Spearman correlation coefficients = 0.93, p < 0.0001, n = 22). Deeper wells with longer open intervals have greater capacity for extracting groundwater (well yields) and dilution that could lower the concentration of any viruses present in the well. Furthermore, deeper wells will be extracting groundwater traveling along deeper and longer flow paths with longer residence times allowing attenuation of pathogens prior to reaching the well. The complexity of the three-dimensional (3-D) groundwater flow system with groundwater pumping from long open-well screens in fractured bedrock aquifers provides strong mixing of variable aged groundwater and, therefore, travel paths, leading to very low concentrations when detections occur. Hence, what is most striking for this study is the frequency of positive detects in a large number of wells but consistently at very low concentrations.
In this investigation, greater amounts of precipitation 8–14 days or 15–21 days previous to the sampling date were associated with higher virus concentrations in the virus positive wells. Precipitation can enhance virus transport by infiltrating water-carrying viruses downward to groundwater. Moreover, low ionic strength water, as is characteristic of precipitation, can desorb viruses that had been attached to sediment grains (Hunt and Johnson 2016). Bradbury et al. (2013) also demonstrated a strong association between precipitation events and increased virus detection rates, and heavy precipitation rates have been associated with increased incidence of acute gastrointestinal illness (Uejio et al. 2014).
While most of the statistical associations identified in the present study have reasonable mechanistic explanations or have been previously reported in the literature their interpretation still requires caution. The datasets used in the statistical analyses were small (10 virus positive samples in 22 wells, 7–36 sampling dates with ≥1 virus-positive well), which limited our ability to detect significant effects that may exist. The small sample size also limited our ability to account for the nonindependence of the data through methods such as mixed models or robust variance estimation. As a result, it is possible that some of the reported p-values are artificially low. The statistical modeling also did not include potentially important covariates, for example, proximity of the virus contamination source.
Virus transport to the study wells
Determining capture zones and time of travel were outside the scope of the current study. On the other hand, both Ogorzaly et al. (2010) and Charles et al. (2009) showed the adenovirus genome (three positive wells) degraded within less than 2 years in groundwater and Bofill-Mas et al. (2006) showed human polyomavirus (four positive samples) to have persistence characteristics similar to adenovirus. This suggests that the bedrock wells in the southern Wellington County area that were positive for viruses were influenced by water less than 2 years old and were susceptible to rapid sewage-derived contamination.
Risk associated with virus contamination of a fractured bedrock aquifer
While numerous wells within the Guelph area exhibited positive virus detections throughout this study, the population is not necessarily at risk for waterborne disease transmission. In Ontario municipal water supplies must abide by the Safe Drinking Water Act and meet the Ontario Drinking Water Quality Standards. As these standards call for the treatment and chlorination of water distributed to the public as drinking water, viruses present in the supply wells are likely inactivated prior to distribution. Private well owners, however, are not required to treat or disinfect their drinking water. Although the infectious nature of the viruses detected in the current investigation was not determined, there may still be some risk associated with their detection. Borchardt et al. (2012) showed qPCR-measured enteroviruses and genogroup I noroviruses were strongly associated with acute gastrointestinal illness in communities supplied with non-disinfected groundwater. Consumption of inadequately treated or raw groundwater in the study area may similarly result in elevated disease risk. As many private well owners drink untreated or minimally treated water, they may be at a risk of acute gastrointestinal illnesses derived from their drinking water.
Traditional fecal indicators and virus detection
In the United States, the Ground Water Rule (US EPA 2006) relies heavily on the requirements of the Total Coliform Rule (1989), which ultimately calls for the monitoring of the distribution systems of all public water systems for total coliforms at a frequency proportional to the number of people served by that water system. In Canada, the Safe Drinking Water Act requires similar monitoring of entry points to drinking water distribution systems (Government of Ontario, MOE, 2002); however, the regional Public Health authority, Public Health Wellington-Dufferin-Guelph (2016) recommends that private well owners sample their water 3 times per year. In the current study, virus samples were collected up to 6 times from some of the sampling wells while samples for bacterial indicators were only collected twice over 8 months; therefore, the results for the fecal indicators that are presented here are more representative of sampling frequencies of private wells than public water systems.
Of the 10 virus occurrences detected in the current study, 4 occurred during sampling events when bacterial samples were also being collected. Of these four occurrences, only one of the virus detections was accompanied by a bacterial indicator; the detection of Adenovirus A in MSW4 was accompanied by the detection of total coliforms at an average concentration of 0.043 CFU 100 m L−1. The other three virus detections went unaccompanied by traditional fecal indicators. Of the six virus detections that occurred without simultaneous sampling for fecal indicators, four of the wells that exhibited viruses had positive detections of total coliforms 1–4 months prior to the virus detection and only one of these detections was also positive for E. coli. Therefore, five of the 10 wells that exhibited virus detections also had detections of total coliforms at some point in time but the remainder of the virus detections went completely undetected by traditional fecal indicators. From these results, it is clear that analyzing water for total coliforms and E. coli at the frequency suggested by Public Health Ontario can miss up to 50% of fecal contamination incidents.
The fact, however, that total coliforms and E. coli are not 100% efficient indicators of virus contamination is not a new concept (Keswick and Gerba 1980; Lindsey et al. 2002; Borchardt et al. 2003). As these bacteria can be up to 100 times larger than viruses, their transport properties in fractured bedrock aquifers can be significantly different. What makes the statistics presented here interesting is the fact that the sample volumes collected during this investigation were 10 times greater than typical sampling methods would suggest; as the concentrations of the fecal indicators were expected to be low, sample volumes were increased to approximately 1,000 ml in comparison to the 100 ml suggested by Method 1604 (US EPA 2002). This proved to be a valuable modification as the maximum concentration of the triplicate samples collected at MSW4 was 0.147 CFU 100 m L−1 (1 colony 680 m L−1), which is likely to have been missed if only the suggested 100-ml sample had been collected. In addition, total coliforms were detected in a total of nine wells during both bacterial sampling events. Of the nine detections during the first event, seven detections had average concentrations less than 1 CFU 100 m L−1. The second sampling event exhibited 6 detections with average concentrations less than 1 CFU 100 m L−1. For E. coli, three out of the four detections for both sampling events had average concentrations less than 1 CFU 100 m L−1. This would suggest that even with high frequency sampling of large public water systems, fecal contamination could go undetected if 100-ml sample volumes are continued to be used.
Conclusions
During an 8-month sampling campaign with six sampling events per well, human enteric viruses were found in five of eight municipal supply wells and five of 11 private wells in the fractured dolostone aquifer of Southern Wellington County. Wells with longer open interval lengths were more likely to be virus positive. In the virus-positive wells, virus concentrations decreased with increasing well depths and longer open interval lengths. Virus concentrations increased with increasing amounts of precipitation in the periods 8–14 days or 15–21 days prior to the sample date. Fractured bedrock aquifers are known for their relatively high degree of vulnerability to contamination due to rapid groundwater flow rates in well-connected fracture networks, and as previous investigations reported in the literature would suggest, also vulnerable to virus contamination. As far as the authors are aware, this study reports the most comprehensive sampling for enteric viruses in a fractured dolostone aquifer. Such aquifers in regions like southern Wellington County where the overburden is thin in groundwater recharge areas, well casings are shallow and extend only into the upper few feet of bedrock, the regional water table is shallow, groundwater velocities in the fractures are 1–10 m day–1, and cool groundwater temperatures promote virus longevity are all characteristics in combination that create an ideal hydrogeologic setting for virus transport and widespread contamination, although at very low concentrations.
References
Abd-Elmaksoud S, Spencer SK, Gerba CP, Tamimi AH, Jokela WE, Borchardt MA (2014) Simultaneous concentration of bovine viruses and agricultural zoonotic bacteria from water using sodocalcic glass wool filters. Food Environ Virol 6:253–259
Allen SA (2013) Vulnerability of a fractured dolostone aquifer to emerging sewage-derived contaminants and their use as indicators of virus contamination. Master of Science Thesis, University of Guelph, Guelph, pp 158. http://hdl.handle.net/10214/7581. Accessed Feb 2017
Belan K P (2010) Characterizing a fractured rock aquifer with hydraulic testing at a contaminated municipal well using flexible liner methods and depth discrete monitoring. MSc Thesis, University of Guelph, Guelph, ON, Canada, 206 pp
Bofill-Mas S, Albinana-Gimenez N, Clemente-Casares P, Hundesa A, Rodriguez-Manzano J, Allard A, Calvo M, Girones R (2006) Quantification and stability of human adenoviruses and polyomavirus JCPyV in wastewater matrices. Appl Environ Microbiol 72(12):7894–7896
Borchardt MA, Bertz PD, Spencer SK, Battigelli DA (2003) Incidence of enteric viruses in groundwater from household wells in Wisconsin. Appl Environ Microbiol 69(2):1172–1180
Borchardt MA, Bradbury KR, Gotkowitz MB, Cherry JA, Parker BL (2007) Human enteric viruses in groundwater from a confined bedrock aquifer. Environ Sci Technol 41(18):6606–6612
Borchardt MA, Bradbury KR, Alexander EC, Kolberg RJ, Alexander SC, Archer JR, Braatz LA, Forest BM, Green JA, Spencer SK (2011) Norovirus outbreak caused by a new septic system in a dolomite aquifer. Ground Water 49(1):85–97
Borchardt MA, Spencer SK, Kieke BA, Lambertini E, Loge FJ (2012) Viruses in nondisinfected drinking water from municipal wells and community incidence of acute gastrointestinal illness. Environ Health Perspect 120(9):1272–1279
Bradbury KR, Borchardt MA, Gotkowitz M, Spencer SK, Zhu J, Hunt RJ (2013) Source and transport of human enteric viruses in deep municipal water supply wells. Environ Sci Technol 47:4096–4103
Brunton FR (2009) Update of revisions to the Early Silurian stratigraphy of the Niagara Escarpment: integration of sequence stratigraphy, sedimentology and hydrogeology to delineate hydrogeologic units. In: Summary of field work and other activities 2009. Ontario Geologic Survey, Open file report 6240, pp 25-1–25-20
Charles KJ, Shore J, Sellwood J, Laverick M, Hart A, Pedley S (2009) Assessment of the stability of human viruses and coliphage in groundwater by PCR and infectivity methods. J Appl Microbiol 106(6):1827–1837
Cherry JA, Parker BL, Bradbury KR, Eaton TT, Gotkowitz M, Hart DJ, Borchardt MA (2006) Contaminant transport through aquitards: a state-of-the-science review. American Water Works Association Research Foundation, Denver, CO, 126 pp
Davies CM, Logan MR, Rothwell VJ, Krogh M, Ferguson CM, Charles K, Deere DA, Ashbolt NJ (2006) Soil inactivation of DNA viruses in septic seepage. J Appl Microbiol 100:365–374
Davis JV, Witt EC, (2000) Microbiological and chemical quality of groundwater used as a source of public supply in southern Missouri-Phase 1, May 1997–March 1998. US Geol Surv Water Resour Invest Rep 00-4038
De Leon R, Shieh C, Baric RS, Sobsey MD (1990) Detection of enterovirus and hepatitis A virus in environmental samples by gene probes and polymerase chain reaction. Proceedings of the American Water Works Association Water Quality Technology Conference, Denver, CO, November 1990, pp 833–853
Femmer SR (2000) Microbiological and chemical quality of groundwater used as a source of public supply in southern Missouri: Phase II, April–July 1998. US Geol Surv Water Resour Invest Rep 2000-4260
Freeze RA, Cherry JA (1979) Groundwater. Prentice Hall, Upper Saddle River, NJ, 604 pp
Gibson KE, Schwab KJ, Spencer SK, Borchardt MA (2012) Measuring and mitigating inhibition during quantitative real time PCR analysis of viral nucleic acid extracts from large-volume environmental water samples. Water Res 46:4281–4291
Golder Associates (2006) Wellington county groundwater protection study, prepared for the Grand River Conservation Authority, Golder, Golden, CO
Hastie TJ, Tibshirani RJ (1990) Generalized additive models. Chapman and Hall, New York
Huber PJ (1973) Robust regression: asymptotics, conjectures and Monte Carlo. Ann Stat 1:799–821
Hunt RJ, Johnson WP (2016) Pathogen transport in groundwater systems: contrasts with traditional solute transport. Hydrogeol J. doi:10.1007/s10040-016-1502-z
Hurst CJ (1988) Influence of aerobic microorganisms upon virus survival in soil. Can J Microbiol 34:696–699
Johnson TB, Mckay LD, Layton AC, Jones SW, Johnson GC, Cashdollar JL, Dahling DR, Villegas LF, Fout GS, Saylor G (2011) Viruses and bacteria in karst and fractured rock aquifers in East Tennessee, USA. Ground Water 49(1):98–110
Jothikumar N, Lowther JA, Henshilwood K, Lees DN, Hill VR, Vinjé J (2005) Rapid and sensitive detection of noroviruses by using TaqMan-based one-step reverse transcription-PCR assays and application to naturally contaminated shellfish samples. Appl Environ Microbiol 71:1870–1875
Kageyama T, Kojima S, Shinohara M, Uchida K, Fukushi S, Hoshino FB, Takeda N, Katayama K (2003) Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. J Clin Microbiol 41:1548–1557
Keller CE, Cherry JA, Parker BL (2014) New method for continuous transmissivity profiling in fractured rock. Ground Water 52(3):352–367
Keswick BH, Gerba CP (1980) Viruses in groundwater. Environ Sci Technol 14(11):1290–1297
Kuo DH, Simmons F, Xagoraraki I (2009) A new set of PCR assays for the identification of multiple human adenovirus species in environmental samples. J Appl Microbiol 107:1219–29
Lake Erie Region Source Protection Committee (2012) Grand River source protection area: approved assessment report. Lake Erie Region Source Protection Committee, Cambridge, ON
Lambertini E, Spencer SK, Bertz PD, Loge FJ, Borchardt MA (2008) Concentration of enteroviruses, adenoviruses, and noroviruses from drinking water by use of glass wool filters. Appl Environ Microbiol 74:2990–2996
Lieberman RJ, Shadix LC, Newport BS, Frebis CP, Moyer MWN, Safferman RS, Stetler RE, Lye D, Fout GS, Dahling DR (2002) Microbial monitoring of vulnerable public groundwater supplies. American Water Works Association Research Foundation report 90914, AWW, Denver, CO
Lindsey BD, Rasberry JS, Zimmerman TM (2002) Microbiological quality of water from noncommunity supply wells in carbonate and crystalline aquifers of Pennsylvania. US Geol Surv Water Resour Invest Rep 2001-4268
Locas A, Barthe C, Barbeau B, Carriere A, Payment P (2007) Virus occurrence in municipal groundwater sources in Quebec, Canada. Can J Microbiol 53:688–694
McNutt LA, Wu C, Xue X, Hafner JP (2003) Estimating the relative risk in cohort studies and clinical trials of common outcomes. Am J Epidemiol 157(10):940–943
McQuaig SM, Scott TM, Lukasik JO, Paul JH, Harwood VJ (2009) Quantification of human polyomaviruses JC virus and BK virus by TaqMan quantitative PCR and comparison to other water quality indicators in water and fecal sample. Appl Environ Microbiol 75:3379–3388
Millen HT, Gonnering JC, Berg RK, Spencer SK, Jokela WE, Pearce JM, Borchardt JS, Borchardt MA (2012) Glass wool filters for concentrating waterborne viruses and agricultural zoonotic pathogens. J Vis Exp 61:LSW2930. doi:10.3791/3930
Monpoeho S, Dehée A, Mignotte B, Schwartzbrod L, Marechal V, Nicolas JC, Billaudel S, Férré V (2000) Quantification of enterovirus RNA in sludge samples using single tube real-time RT-PCR. Biotechniques 29:88–93
Munn JD (2012) High-resolution discrete fracture network characterization using inclined coreholes in a Silurian aquifer in Guelph, Ontario. MSc Thesis, University of Guelph, Guelph, ON, Canada, 60 pp. http://hdl.handle.net/10214/3242. Accessed February 2017
O’Connor DR (2002) Part One: a summary report of the Walkerton inquiry: the events of May 2000 and related issues. Ontario Ministry of the Attorney General, Toronto
Ogorzaly L, Bertrand I, Paris M, Maul A, Gantzer C (2010) Occurrence, survival, and persistence of human adenoviruses and F-specific RNA phages in raw groundwater. Appl Environ Microbiol 76(24):8019–8025
Ontario Government (2002). Drinking water system. In: Safe Drinking Water Act, 2002, Ontario regulation 170/03. https://www.ontario.ca/laws/regulation/030170#BK0
Ontario Ministry of Northern Development and Mines (2013) OGS Earth, Queen’s printer for Ontario. Ontario Ministry of Northern Development and Mines, Toronto
Pehme PE, Parker BL, Cherry JA, Greenhouse JP (2010) Improved resolution of ambient flow through fractured rock with temperature logs. Ground Water 48(2):191–205
Public Health Wellington-Dufferin-Guelph (2016) Private well water. https://www.wdgpublichealth.ca/?q=adultwellwater. Accessed July 2016
Quinn PM, Parker BL, Cherry JA (2011) Using constant head step tests to determine apertures in fractured rock. J Contam Hydrol 126(1–2):85–99
Schwab KJ, DeLeon R, Sobsey MD (1995) Concentration and purification of beef extract mock eluates from water samples for the detection of enteroviruses, hepatitis A virus and Norwalk virus by reverse transcription PCR. Appl Environ Microbiol 61:531–537
Spackman E, Senne DA, Myers TJ (2002) Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. J Clin Microbiol 40:3256–3260
US Environmental Protection Agency (US EPA) (1989) Drinking water; National Primary Drinking Water Regulations; Total Coliform Rule (TCR) 54 FR 27544–27568, vol 54, no. 124, US EPA, Washington, DC
US Environmental Protection Agency (2002) Method 1604: total coliforms and Escherichia coli in water by membrane filtration using a simultaneous detection technique (MI medium). EPA 821-R-02-024, US EPA, Washington, DC, 14 pp. Available at https://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=P1002D57.txt. Accessed February 2017
US Environmental Protection Agency (2006) Economic analysis for the final groundwater rule. Office of Water 815R06014, US EPA, Washington, DC
Uejio CK, Yale SH, Malecki K, Borchardt MA, Anderson HA, Patz JA (2014) Drinking water systems, hydrology, and childhood gastrointestinal illness. Am J Public Health 104:639–646
Wallender EK, Ailes EC, Yoder JS, Roberts VA, Brunkard JM (2014) Contributing factors to disease outbreaks associated with untreated groundwater. Ground Water 52:886–897
Weisbrod N, Meron H, Walker S, Gitis V (2013) Virus transport in a discrete fracture. Water Res 47:1888–1898
Wong K, Xagoraraki I (2011) Evaluating the prevalence and genetic diversity of adenovirus and polyomavirus in bovine waste for microbial source tracking. Appl Microbiol Biotechnol 90:1521–1526
Yates MV, Gerba CP, Kelley LM (1985) Virus persistence in groundwater. Appl Environ Microbiol 49(4):778–781
Yeager JG, O’Brien RT (1979) Enterovirus inactivation in soil. Appl Environ Microbiol 38(4):694–701
Yohai VJ (1987) High breakdown point and high efficiency robust estimates for regression. Ann Stat 15:642–656
Zhang W, Tang X, Weisbrod N, Guan Z (2012) A review of colloid transport in fractured rocks. J Mater Sci 9:770–787
Acknowledgements
We thank Jordan Gonnering, Hana Millen, and Susan Spencer for laboratory technical assistance. Funding for this work from an Ontario Research Fund Research Excellence Round 3 grant No. RE03-061 to Dr. Beth Parker as lead principal investigator.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the special issue “Hydrogeology and Human Health”
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Allen, A.S., Borchardt, M.A., Kieke, B.A. et al. Virus occurrence in private and public wells in a fractured dolostone aquifer in Canada. Hydrogeol J 25, 1117–1136 (2017). https://doi.org/10.1007/s10040-017-1557-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10040-017-1557-5