Abstract
Purpose
The use of mesh is a common practice in ventral hernia repair (VHR). Lack of consensus on which prosthetic material works better in different settings remains. This meta-analysis aims to summarize the available evidence on hernia recurrence and complications after repair with synthetic, biologic, or biosynthetic/bioabsorbable meshes in hernias grade 2–3 of the Ventral Hernia Working Group modified classification.
Methods
A literature search was conducted in January 2021 using Web of Science (WoS), Scopus, and MEDLINE (via PubMed) databases. Randomized Controlled Trials (RCTs) and observational studies with adult patients undergoing VHR with either synthetic, biologic, or biosynthetic/bioabsorbable mesh were included. Outcomes were hernia recurrence, Surgical Site Occurrence (SSO), Surgical Site Infection (SSI), 30 days re-intervention, and infected mesh removal. Random-effects meta-analyses of pooled proportions were performed. Quality of the studies was assessed, and heterogeneity was explored through sensitivity analyses.
Results
25 articles were eligible for inclusion. Mean age ranged from 47 to 64 years and participants’ follow-up ranged from 1 to 36 months. Biosynthetic/bioabsorbable mesh reported a 9% (95% CI 2–19%) rate of hernia recurrence, lower than synthetic and biologic meshes. Biosynthetic/bioabsorbable mesh repair also showed a lower incidence of SSI, with a 14% (95% CI 6–24%) rate, and there was no evidence of infected mesh removal. Rates of seroma were similar for the different materials.
Conclusions
This meta-analysis did not show meaningful differences among materials. However, the best proportions towards lower recurrence and complication rates after grade 2–3 VHR were after using biosynthetic/slowly absorbable mesh reinforcement. These results should be taken with caution, as head-to-head comparative studies between biosynthetic and synthetic/biologic meshes are lacking. Although, biosynthetic/bioabsorbable materials could be considered an alternative to synthetic and biologic mesh reinforcement in these settings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Incisional ventral hernia repair is a common surgical practice. The use of mesh reinforcement has become a reference to prevent hernia recurrence [1, 2]. However, the variety of devices—with over 150 mesh products on the market—and surgical techniques make it difficult to evaluate prostheses performance [3, 4]. Lack of consensus remains regarding the best practices for ventral hernia repair (VHR) in the setting of a contaminated surgical field [2]. Permanent synthetic meshes are made of non-absorbable materials such as polyester, polypropylene, polyvinylidene fluoride, or polytetrafluoroethylene (PTFE). Although longer used, these prostheses are associated with a risk of postoperative infection and subsequent complications in contaminated or clean-contaminated settings [1, 5,6,7]. This previous context led to the development of biologic implants, as an alternative to synthetic meshes [7, 8]. However, concerns about their possible advantages remain [8,9,10].
Biosynthetic/slowly absorbable meshes have recently been introduced as a potential alternative to biological implants in contaminated abdominal wall reconstruction [6]. The absorption rates of these meshes may vary according to their composition: 6 months for polyglycolic acid (PGA): trimethylene carbonate materials (TMC), 12–18 months for poly-4-hydroxybutyrate (P4HB) component, and 12–36 months for a mixture of copolymer fibers of polyglycolide, polylactide, and polytrimethylene carbonate and fibers of polylactide and polytrimethylene carbonate, respectively, in comparison to classical glycolide and lactide mesh, which resorbs in less than two months [1, 11].
The Ventral Hernia Working Group (VHWG) 2010 classification system [5] stratifies patients with ventral hernia according to their risk for postoperative Surgical Site Occurrence (SSO), depending on the patient and wound characteristics, such as smoker, obese, diabetic, immunosuppressed, chronic obstructive pulmonary disease (COPD), or history of wound infection and hernia defects. Thus, it suggests the use of synthetic mesh, based on surgeon preference, for low-risk (grade 1) and the use of biologic mesh for increased risk and co-morbid patients (grade 2) as well as for contaminated or infected wounds (grades 3 and 4). However, the higher costs of biologic implants could have limited their use, even for high-risk patients [12]. In 2012 the VHWG classification was modified [13] stratifying patients as grade 1 (clean cases; low risk of complications), grade 2 (clean cases; co-morbidity, history of infection), or grade 3 (clean-contaminated, contaminated, and dirty wounds). In the setting of complex VHR, newly developed biosynthetic/slowly absorbable meshes have shown promising results for hernia recurrence and other postoperative complications [14].
In the previous context and given the lack of consensus on the most appropriate material for hernia repair in clean-contaminated and contaminated wounds, the present work aims to review the existing literature and gather evidence of postoperative outcomes of synthetic, biologic, and biosynthetic/slowly absorbable meshes in patients undergoing modified VHWG (2012) grade 2–3 complex ventral hernias.
Methods
A systematic review and meta-analysis were conducted following the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 statement [15] and the Meta-analysis of Observational Studies in Epidemiology guidelines [16]. The review was registered in the Prospective Register of Systematic Reviews (CRD42021265235).
Search strategy
A literature search was conducted in January 2021. The search only considered those studies published from 2000 to 2021, written in English or Spanish, with the last search update on January 27. Search strategy was performed by an expert documentalist using the following databases: Web of Science (WoS), Scopus and MEDLINE (via PubMed). The search strategy included a combination of keywords and MeSH terms about hernia and mesh-related terms (see Supplementary file 1 for searching strategies in each database). Two independent reviewers (SMC and MLC) assessed the title and abstract of each record for inclusion and exclusion criteria. After preliminary screening, the selected publications were scanned in full text by the authors for eligibility. Discrepancies were solved after a discussion between reviewers.
Study selection, data extraction and quality assessment
We established the inclusion criteria for study selection according to observational studies, as well as randomized control trials (RCTs) of any design, with adult patients (≥ 18 years of age) undergoing grade 2–3 complex VHR with prosthetic reinforcement, either with synthetic, biologic, or biosynthetic/slowly absorbable mesh. Hernia grade was defined according to the modified VHWG grading system [13]. Publications reporting on at least one of the following primary outcomes were included: hernia recurrence, SSO, as described by the VHWG including infection, wound dehiscence, seroma, or development of an enterocutaneous fistula [13] and Surgical Site Infection (SSI), according to the Center for Disease Control (CDC) [17] definition. Secondary outcomes of interest were 30 days re-intervention and infected mesh removal.
Exclusion criteria included no reported clinical outcomes of interest, preclinical studies and/or review articles, editorials, data from registries, articles with non-full text available or comments to a study, single case reports, studies related to parastomal, inguinal, or hiatal hernia repair, repair using either no mesh or two different meshes, as well as prophylactic mesh insertion. For duplicate data reported by the same author(s), the article with the longest follow-up period was selected [18, 19].
Data were extracted by two independent reviewers using a Microsoft®Excel (Microsoft® Corporation 2016) data extraction form. Predefined variables such as year of publication, study design, follow-up period and number of patients were retrieved. Results reported in the studies were recorded according to mesh type. In addition, patient characteristics and comorbidities associated with increased risk for postoperative infection were extracted, including age, sex, Diabetes Mellitus (DM), obesity (Body Mass Index > 30 kg/m2), Chronic Obstructive Pulmonary Disease (COPD), active smoker, and history of wound infection. Hernia characteristics and operative details were extracted as well: CDC wound classification [17], VHWG hernia grade [13], mesh type, setting of intervention (emergency or elective), repair technique and mesh placement location. In terms of postoperative complications, data on hernia recurrence, SSO, seroma, and SSI, as well as 30 days re-intervention, mesh removal and length of hospital stay were detailed. Those articles in which VHWG grade was not explicitly reported but had a cohort of patients with one or more of the comorbid conditions mentioned above, which allowed for a definition of VHWG grade, were selected and included in the review (classified as grade ≥ 2). Articles with grade 1–3 hernias for which outcomes were disaggregated by hernia grade were included in the analysis only collecting data for grade 2–3 outcomes. Discrepancies in data extraction were solved by the team of authors involved.
Bias of the included studies has been assessed using the Cochrane RoB 2 Tool for RCT [20] and robvis tool to create risk-of-bias plots [21]. The appropriate adaptations of the Newcastle–Ottawa scale were used for cohort or cross-sectional studies [22, 23]. This scale is based on a cumulative score in three categories: selection of study groups, comparability of their cases and controls and ascertainment of the outcome/exposure. There are two versions of the scale, one for cohort studies and one for case–control studies, and other version adapted from cross-sectional studies. If the assessed study includes criteria for an item, a score of one point is allocated to the selection and ascertainment of outcome/exposure category; two points can be allocated to the comparability category, resulting in a maximum of 9 points. We considered studies that received a score of 9–7 to have a low risk of bias, studies with 4–6 points as medium risk of bias, and studies with 0–3 points as a high risk of bias.
Two authors evaluated the articles’ methodological quality, and any discrepancies were solved through discussion with a third investigator.
Data synthesis and statistical analysis
The meta-analyses were carried out considering one effect size per each study included, regardless of the type of mesh. A random-effects model was fitted to estimate pooled weighted proportions with corresponding 95% confidence intervals (CIs). CIs 95% excluding 0 were considered significant. Statistical heterogeneity was assessed using the χ2 and I2 statistics to analyse the extent of inconsistency and variation across the studies. Heterogeneity was considered high if I2 was > 50% [24]. Heterogeneity was explored with sensitivity analyses to reflect the influence of several subgroups on the pooled proportions adjusted by mesh type. The variables explored in the subgroup analysis were based on a time horizon of outcomes (studies with only long-term complications), population characteristics (studies with a patient with mean BMI ≥ 30 and mean BMI < 30, studies with a proportion of smokers ≥ 25% and smokers < 25%), study design (prospective vs retrospective) and risk of study bias (low vs moderate).
The possibility of publication bias was examined using the Doi plot and Luis Furuya-Kanamori asymmetry index (LFK index) [25] by a subgroup of mesh type. This approach has been suggested to be more robust for meta-analyses that include less than 10 studies. In the presence of symmetry, one can conclude as no publication bias but in the absence of symmetry, one can expect publication bias. This publication bias was measured by the asymmetry index (LFK index). An LFK index within ± 1, out of ± 1 but within ± 2, and >± 2 is to mean no asymmetry, minor asymmetry, and major asymmetry, respectively. The influence of potential publication bias on results was explored by using the trim-and-fill procedure [25]. P < 0.05 was considered statistically significant in all analyses. These were conducted using STATA (16.1, StataCorp LLC, College Station, TX).
Results
Study selection
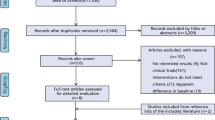
A total of 2793 articles were identified in the primary literature search (Fig. 1). After the removal of duplicates, the literature search identified 1331 publications. Following the screening of the title and abstract, 62 articles were assessed for full-text review. 25 articles met the inclusion criteria and qualified for the study. The main reasons for exclusion after full-text assessment were existing systematic reviews, lack of information on hernia characteristics (VHWG grade 2–3), lack of information on patient comorbidities and contamination to define hernia grade if missing, and lack of results for outcomes of interest. A subsequent article with the same data from the same authors but with a longer follow-up replaced the publication with a shorter follow-up.
Study characteristics
Of the 25 publications that met the inclusion criteria, only two were RCTs [8, 26]; 17 were observational retrospective studies [12, 14, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41] and six were observational prospective studies [1, 3, 6, 9, 19, 42]. Thirteen studies compared permanent synthetic to biologic mesh [8, 12, 26,27,28,29,30,31,32, 34,35,36, 38]; one publication compared synthetic to biosynthetic/slowly absorbable mesh [40]; two compared biosynthetic/slowly absorbable to biologic implant [14, 41] and, 9 articles presented outcomes of biosynthetic/slowly absorbable meshes [1, 3, 6, 9, 14, 19, 33, 39, 42] (six for P4HB mesh, two publications referred to PGA: TMC mesh, and one referred to both P4HB and PGA: TMC meshes. Among the studies using synthetic mesh, polypropylene was the most common material [8, 12, 26, 27, 29, 30, 32, 34, 35, 38, 40]. For biologic prosthetics, nine articles described porcine dermis meshes [8, 14, 26, 28, 32, 34, 35, 38, 41], whereas one article mentioned both porcine and human dermis prosthetics [29].
The 25 selected studies included a total of 3771 patients. Mean age ranged from 47 to 64 years and participants’ follow-up ranged from 1 to 36 months. A clear description of the VHWG hernia grade was reported in 13 studies [1, 3, 9, 26, 29,30,31, 33, 34, 37,38,39,40], although the remaining articles presented data on comorbid conditions which allowed for a definition of VHWG grade if patients had one or more of the listed comorbidities (hernias classified as grade ≥ 2) [6, 8, 12, 14, 27, 32, 35, 36, 41, 42]. Two articles reported on “high risk” patients, as those immunosuppressed and comorbid [19, 28]. In three studies with grade 1 hernias, operative outcomes were disaggregated by hernia grade, so only data on operative outcomes fulfilling the hernia grade criteria (grades 2 and 3) were derived separately [1, 33, 37]. Regarding wound contamination, most of the studies reported on CDC wound class [1, 6, 8, 9, 12, 19, 26, 27, 29, 30, 33,34,35,36,37, 39,40,41,42]. Surgical characteristics and outcomes are shown in Supplementary file 2. Only one article did not report any information on mesh location [27]. For the outcomes of interest, two studies lacked information on hernia recurrence [29, 40]; three articles did not present results on SSI [1, 28, 30]; 12 articles did not report on SSO [3, 14, 19, 26,27,28,29,30,31,32, 38, 41] while only five articles did not report on seroma [1, 14, 27, 30, 32]. Regarding operative technique, 12 studies reported on component separation techniques (CST) [1, 6, 9, 12, 29, 30, 33,34,35,36,37,38] (Table 1).
Quality assessment
The 2 RCT studies included in the review have an overall low risk of bias in the evaluation of methodological quality (Fig. 2). For the cohort studies, eleven obtained a high score and twelve a moderate score. The most frequent risks of bias were related to the risk of comparability based on the design or analysis (Table 2).
Meta-analysis findings
Seroma
Rates of seroma are reported in 20 studies; however, only 13 articles reported in SSO rates. For this reason, only the former was included in the pooled analysis. 10 studies reported rates of seroma for biosynthetic/slowly absorbable meshes of 8% (95% CI 4–13%). A similar rate of 9% (95% CI 5–13%) was obtained from 11 studies on biologic mesh, as well as from the 11 articles on synthetic meshes, with a 9% rate of seroma (95% CI 5–13%). Heterogeneity ranged from I2 = 56.72% to I2 = 71.37% (Table 3).
Surgical site infection
Ten studies reported on SSI rates for biosynthetic/slowly absorbable meshes. The pooled estimation showed lower SSI rates than biologic and synthetic mesh: 14% (95% CI 6–24%), compared to 18% (14–24%) for synthetic meshes (12 studies) and a higher rate of 27% (95% CI 17–38%) obtained from 13 articles reporting on biologic prothesis. There was high heterogeneity among studies (I2 statistics values between 68.93 and 89.91%; Fig. 3).
Infected mesh removal
Infected meshes were reported in 9 studies with a total of 79 events. The pooled estimation in the subgroup of biosynthetic/slowly absorbable mesh was 0% (95% CI 0–2%) with a very low heterogeneity (I2 = 0%) among 4 studies, compared with the subgroup of biologic and synthetic mesh, with pooled estimations of 2% (95% CI 0–7%) and 9% (95% CI 0–25%), respectively, and high heterogeneity among 5 studies (I2 = 64.60% and 92.33%).
Hernia recurrence
Meta-analysis showed that the rate of hernia recurrence was lower for biosynthetic/slowly absorbable mesh when compared to synthetic and biologic meshes. Ten studies on biosynthetic/slowly absorbable meshes reported an incidence of 9% (95% CI 2–19%), versus 12 studies on synthetic mesh, with a recurrence rate of 13% (95% CI 8–19%) and a hernia recurrence of 20% (95% CI 14–25%) obtained for the 14 studies on a biologic mesh. The I2 statistic reported substantial heterogeneity among the studies (93.32%, 77.59% and 69.53% for biosynthetic/bioabsorbable, synthetic, and biologic mesh, respectively).
Re-intervention
30 days re-invention was reported in 5 studies for synthetic mesh and 7 studies for biologic and biosynthetic/slowly absorbable each. Meta-analysis resulted in a pooled re-intervention estimation of 8% (5–12%) for biosynthetic/slowly absorbable and 11% (95% CI 8–12%) for biologic. Heterogeneity was low-moderate I2 = 37.40% and I2 = 3.38%.
Sensitivity analysis
For the sensitivity analysis, we first analysed the subgroup of studies reporting results on long-term complications. In this sense, only 3 studies [9, 29, 40] reported short-term complication outcomes of seroma, SSI, and reintervention derived from the use of biosynthetic and synthetic meshes. The two studies with short-term complications of synthetic mesh [29, 40] had a seroma rate of 0% (95% CI 0–14%) [29] and 4% (95% CI 2–8%) [40], SSI rate of 21% (95% CI 9–40%) [29] and 11% (95% CI 7–16%) [40], and reoperation rate of 0% (95% CI 0–14%) [29] and 4% (95% CI 2–18%) [40]. Similarly, studies reporting short-term complications of biosynthetic mesh reported a seroma rate of 2% (95% CI 0–9%) [40] and 8% (95% CI 4–16%) [9], SSI of 22% (95% CI 14–35%) [40] and 13% (95% CI 7–22%) [9] and reoperation rate of 14% (95% CI 7–25%) [40]. After removing these 3 articles, we pooled subgroup analysis only for studies with long-term complications of meshes. Results are shown in Supplementary file 4. We can observe a few differences with respect to the results of the total pooled sample by mesh type. Seroma and SSI rates increase slightly in synthetic and biosynthetic mesh [from 9 to 11% (95% CI 6–17%] and from [18 to 20% (95% CI 15–25%)], for the respective long-term complications. In contrast, the long-term reintervention rates for synthetic mesh increase from 6 to 10% (95% CI 7–13%) and remain unchanged for biosynthetic mesh.
To explore risk factors, two subgroups were analysed, considering the main patient characteristics influencing the outcomes of interest. Results are shown in Supplementary file 4. In the subgroup of studies whose patients had an average BMI ≥ 30, there is a higher rate of hernia recurrence, infected mesh, and SSI for biologic mesh. However, complication rates are lower for synthetic mesh, apart from seroma. For biosynthetic/slowly absorbable mesh, there is a trend towards lower rates of SSI, infected mesh removal and hernia recurrence, although this percentage remains higher for the re-intervention variable. The subgroup of studies with, at least, 25% of smokers, presented higher rates of seroma, infected mesh, and re-intervention, independently of mesh type, although lower rates are observed for hernia recurrence if compared to the subgroup smoker < 25%. For the subgroups analysed (BMI ≥ 30, smoker ≥ 25%) mesh type does not seem to influence the outcomes. These analyses present significant heterogeneity and wide confidence intervals, which shows that differences among complication rates in patient subgroups are not significant. Similar results are obtained in sensitivity analyses for study design and risk of bias. There were only eight studies with a prospective design: six out of them were single-arm studies assessing results on biosynthetic/slowly absorbable mesh use and, the remaining were RCTs that compared biologic to synthetic mesh. Outcomes of biosynthetic/slowly absorbable mesh in the prospective studies, which represented a larger sample of studies, although not significant, show a lower rate for seroma, SSI, and recurrence. Finally, the subgroup analysis that classified studies according to low risk and medium risk of bias showed that studies with lower risk had a higher rate of SSI, hernia recurrence and re-intervention for biosynthetic/slowly absorbable and biologic mesh. Again, these results are not significant.
Publication bias
There was evidence of major publication bias regarding the proportion of seroma for each type of mesh, SSI for biologic, infected mesh for synthetic and biologic as indicated by the LFK index. The obtained LFK index and Doi plot based on the outcome by a subgroup of mesh type is presented in Supplementary file 3. The ‘‘trim and fill’’ procedure did not find any possible “missing” study.
Discussion
Based on meta-analysis findings, the rate of hernia recurrence seems lower after biosynthetic/slowly absorbable mesh, with a pooled proportion of 9% recurrence as opposed to synthetic and biologic repair, with a recurrence of 13% and 20%, respectively. Mellia et al. [43] reviewed clinical outcomes of P4HB biosynthetic mesh and reported a similar recurrence of 9.1%. By contrast, the study by Renard et al. [41] compared the efficacy of rapidly absorbable polyglactin mesh versus biologic implant and reported a 68% recurrence for the former. This variability among the results might come from grouping different prosthetic materials within the same mesh category, as their behaviour and health outcomes may differ. Hodgkinson et al. [44] also stratified their analysis by repair method, although they included suture repair alone without mesh placement. Reported hernia recurrence was higher in this study as grade 2 hernias were ruled out: recurrence rate was 21% for non-absorbable synthetic mesh and 28.5% for biologic materials. Pooled estimation for SSI also showed a trend towards lower rates for biosynthetic/slowly absorbable mesh repair, with a 14% rate, compared to 18% for synthetic meshes and a higher rate of 27% for biologic repair. In the same systematic review, Mellia et al. [43] reported a 6.8% rate for SSI. This difference might come from the percentage of grade 1 hernias included in their analysis, associated with a lower risk of postoperative complications and recurrence, with no comorbidities or previously infected wounds. They suggest that anti-microbial properties of P4HB for the repair of contaminated wounds might explain the lower incidence of SSI in their study. In addition, available data for seroma revealed a rate of 8% for biosynthetic/slowly absorbable mesh, similar to the 9% reported for synthetic and biologic materials. In the present analysis, no infected meshes were removed after repair with biosynthetic/slowly absorbable mesh, with very low heterogeneity (I2 = 0%) among the studies, whereas rates of 2% and 9% were estimated for biologic and synthetic meshes, respectively, with high heterogeneity. However, this result should be interpreted with caution, given the small number of studies reporting on the infected mesh. Re-intervention resulted in an 8% rate for biosynthetic/slowly absorbable mesh and 11% for biologics.
This analysis, however, presents several limitations. Data have been reported as a percentage of complications (in absolute terms) derived from each mesh, rather than relative odds ratios from studies comparing one mesh versus another. The reason is the lack of comparative studies of biosynthetic/slowly absorbable mesh with other materials (most of them are single-arm studies). Only two RCTs [8, 26] were included in the review, although their quality assessment reported a low risk of bias. Most of the retrieved publications were retrospective observational studies with a moderate risk of bias because of a lack of comparability of the cases, their design, or the type of analysis. In addition, the I2 statistic reported substantial heterogeneity among the studies for the outcomes of interest. Sensitivity analyses performed to assess heterogeneity have shown that patient characteristics are not a source of this heterogeneity. Similar results are reported in the review by Samson et al. [45]. This review included studies using biological implants in open VHR and analyzed reporting data on early complications, late complications, and recurrences. Thus, reasons for the heterogeneity might come from other demographic characteristics of the patients, different methodologies, repair strategies, and prosthetic materials used in the studies, which limits the comparison of variables between the articles and makes it difficult to derive sound conclusions. However, as other authors stated, we must bear in mind that “…in proportional meta-analysis, I2 is usually high. This can be due to the nature of proportional data, where little variance is observed even in studies with small sample size. Moreover, true heterogeneity is expected in prevalence and incidence estimates due to differences in the time and place where included studies were conducted. Therefore, high I2 in the context of proportional meta-analysis does not necessarily mean that data is inconsistent. As such, the results of this test should be interpreted conservatively” [46].
Moreover, a clear description of the VHWG hernia grade was only reported in 13 studies [1, 3, 9, 26, 29,30,31, 33, 34, 37,38,39,40]. Thus, the grade had to be derived by the reviewers from the patient and wound characteristics. In our search of literature, we sought to select comparative studies for synthetic meshes and biologic implants. On the other hand, for biosynthetic/slowly absorbable meshes, given the lack of comparative publications, we allowed for the inclusion of single-arm studies. The inclusion of these articles could lead to a higher heterogeneity in the results. In addition, there is no universal acceptance of the VHWG grading scale [13].
Also, a recent review includes 274 studies with 28,868 patients to describe till 63 individual predictors for hernia recurrence [47], but we have only considered in our methodology 5 selected items (seroma formation, wound infection, mesh removal, hernia recurrence and re-intervention), in our opinion most important and appropriate to reach our goal.
Finally, our study does not include an analysis of data on preclinical testing and animal studies, which could supplement the scientific evidence for safety and effectiveness.
In conclusion, meta-analysis did not show meaningful differences among materials; although, it seems to be a trend towards lower recurrence and complication rates after grade 2–3 VHR using biosynthetic/slowly absorbable mesh reinforcement. Therefore, biosynthetic/slowly absorbable mesh could be an interesting option as an alternative to synthetic meshes and biologic implants in 2–3 grades. Also, these results should be interpreted with caution because of the lack of direct comparisons between biosynthetic/slowly absorbable versus synthetic meshes and/or biological implants. Differences in patient characteristics and the selection bias in single-arm observational studies make it difficult to have conclusive results.
Future research should focus on randomized controlled trials with defined control groups to allow for head-to-head comparisons among the different mesh materials like COMpACT-BIO study (RCT NCT04597840, clinicaltrials.gov) whose purpose is to investigate the clinical and economic benefit of the use of biosynthetic mesh in contaminated incisional hernia repair in comparison to the standard of repair. This is a multicenter, prospective longitudinal and randomized study, which also offers a standardized technique of repair, that still is open and in a recruitment phase.
Finally, due to the cost differences between the different types of materials, this information should be supplemented with registers, cost-effectiveness studies and/ or budget impact analyses informing about the health care value of the three alternatives.
Change history
01 November 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10029-022-02705-8
References
Vauclair E, Bert M, Facy O, Cheynel N, Rat P, Ortega-Deballon P (2021) What results can be expected one year after complex incisional hernia repair with biosynthetic mesh? J Visc Surg 158(2):111–117. https://doi.org/10.1016/j.jviscsurg.2020.07.008
Morris MP, Mellia JA, Christopher AN, Basta MN, Patel V, Qiu K et al (2021) Ventral hernia repair with synthetic mesh in a contaminated field: a systematic review and meta-analysis. Hernia 25(4):1035–1050. https://doi.org/10.1007/s10029-020-02358-5
Rognoni C, Cuccurullo D, Borsoi L, Bonavina L, Asti E, Crovella F et al (2020) Clinical outcomes and quality of life associated with the use of a biosynthetic mesh for complex ventral hernia repair: analysis of the “Italian Hernia Club” registry. Sci Rep 10(1):10706. https://doi.org/10.1038/s41598-020-67821-w
Deeken CR, Lake SP (2017) Mechanical properties of the abdominal wall and biomaterials utilized for hernia repair. J Mech Behav Biomed Mater 1(74):411–427. https://doi.org/10.1016/j.jmbbm.2017.05.008
Breuing K, Butler CE, Ferzoco S, Franz M, Hultman CS, Kilbridge JF et al (2010) Incisional ventral hernias: review of the literature and recommendations regarding the grading and technique of repair. Surgery 148(3):544–558. https://doi.org/10.1016/j.surg.2010.01.008
Rosen MJ, Bauer JJ, Harmaty M, Carbonell AM, Cobb WS, Matthews B et al (2017) Multicenter, prospective, longitudinal study of the recurrence, surgical site infection, and quality of life after contaminated ventral hernia repair using biosynthetic absorbable mesh: the COBRA study. Ann Surg 265(1):205–211. https://doi.org/10.1097/SLA.0000000000001601
Atema JJ, de Vries FEE, Boermeester MA (2016) Systematic review and meta-analysis of the repair of potentially contaminated and contaminated abdominal wall defects. Am J Surg 212(5):982–995. https://doi.org/10.1016/j.amjsurg.2016.05.003
Harris HW, Primus F, Young C, Carter JT, Lin M, Mukhtar RA et al (2021) Preventing recurrence in clean and contaminated hernias using biologic versus synthetic mesh in ventral hernia repair: the PRICE randomized clinical trial. Ann Surg 273(4):648–655. https://doi.org/10.1097/SLA.0000000000004336
van Rooijen MMJ, Jairam AP, Tollens T, Jørgensen LN, de Vries Reilingh TS, Piessen G et al (2020) Outcomes of a new slowly resorbable biosynthetic mesh (Phasix™) in potentially contaminated incisional hernias: a prospective, multi-center, single-arm trial. Int J Surg 83:31–36. https://doi.org/10.1016/j.ijsu.2020.08.053
Shah BC, Tiwari MM, Goede MR, Eichler MJ, Hollins RR, McBride CL et al (2011) Not all biologics are equal! Hernia 15(2):165–171. https://doi.org/10.1007/s10029-010-0768-7
Deeken CR, Matthews BD (2013) Characterization of the mechanical strength, resorption properties, and histologic characteristics of a fully absorbable material (poly-4-hydroxybutyrate-PHASIX mesh) in a porcine model of hernia repair. ISRN Surg. https://doi.org/10.1155/2013/238067
Majumder A, Winder JS, Wen Y, Pauli EM, Belyansky I, Novitsky YW (2016) Comparative analysis of biologic versus synthetic mesh outcomes in contaminated hernia repairs. Surgery 160(4):828–838. https://doi.org/10.1016/j.surg.2016.04.041
Kanters AE, Krpata DM, Blatnik JA, Novitsky YM, Rosen MJ (2012) Modified hernia grading scale to stratify surgical site occurrence after open ventral hernia repairs. J Am Coll Surg 215(6):787–793. https://doi.org/10.1016/j.jamcollsurg.2012.08.012
Buell JF, Sigmon D, Ducoin C, Shapiro M, Teja N, Wynter E et al (2017) Initial experience with biologic polymer scaffold (poly-4-hydroxybuturate) in complex abdominal wall reconstruction. Ann Surg 266(1):185–188. https://doi.org/10.1097/SLA.0000000000001916
Moher D, Liberati A, Tetzlaff J, Altman DG, for the PRISMA Group (2019) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D et al (2000) Meta-analysis of observational studies in epidemiology. A proposal for reporting. JAMA 283(15):2008–2012. https://doi.org/10.1001/jama.283.15.2008
Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR (1999) Guideline for prevention of surgical site infection, 1999. Hospital infection control practices advisory committee. Infect Control Hosp Epidemiol 20(4):247–280. https://doi.org/10.1086/501620
Roth JS, Anthone GJ, Selzer DJ, Poulose BK, Bittner JG, Hope WW et al (2018) Prospective evaluation of poly-4-hydroxybutyrate mesh in CDC class I/high-risk ventral and incisional hernia repair: 18-month follow-up. Surg Endosc 32(4):1929–1936. https://doi.org/10.1007/s00464-017-5886-1
Roth JS, Anthone GJ, Selzer DJ, Poulose BK, Pierce RA, Bittner JG et al (2021) Prospective, multicenter study of P4HB (Phasix™) mesh for hernia repair in cohort at risk for complications: 3-Year follow-up. Ann Med Surg 1(61):1–7. https://doi.org/10.1016/j.amsu.2020.12.002
Higgins JPT, Altman DG, Sterne J (2017) Chapter 8: assessing risk of bias in included studies. In: Cochrane Handbook for Systematic Reviews of Interventions version 520 [Internet]. John Wiley and Sons [cited 2021 Sep 22]. https://ora.ox.ac.uk/objects/uuid:58a5b92e-c51d-44f0-b265-edf92e7688c2
McGuinness LA, Higgins JPT (2021) Risk-of-bias VISualization (robvis): an R package and shiny web app for visualizing risk-of-bias assessments. Res Synth Methods 12(1):55–61. https://doi.org/10.1002/jrsm.1411
Stang A (2010) Critical evaluation of the Newcastle–Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25(9):603–605. https://doi.org/10.1007/s10654-010-9491-z
Modesti PA, Reboldi G, Cappuccio FP, Agyemang C, Remuzzi G, Rapi S et al (2016) Panethnic differences in blood pressure in Europe: a systematic review and meta-analysis. PLoS ONE 11(1):e0147601. https://doi.org/10.1371/journal.pone.0147601
Higgins JPT, Green S (2011) Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. [Internet]. The Cochrane Collaboration [cited 2021 Nov 25]. www.handbook.cochrane.org.
Furuya-Kanamori L, Barendregt JJ, Doi SAR (2018) A new improved graphical and quantitative method for detecting bias in meta-analysis. Int J Evid Based Healthc 16(4):195–203. https://doi.org/10.1097/XEB.0000000000000141
Olavarria OA, Bernardi K, Dhanani NH, Lyons NB, Harvin JA, Millas SG et al (2021) Synthetic versus biologic mesh for complex open ventral hernia repair: a pilot randomized controlled trial. Surg Infect 22(5):496–503. https://doi.org/10.1089/sur.2020.166
Bondre IL, Holihan JL, Askenasy EP, Greenberg JA, Keith JN, Martindale RG et al (2016) Suture, synthetic, or biologic in contaminated ventral hernia repair. J Surg Res 200(2):488–494. https://doi.org/10.1016/j.jss.2015.09.007
Brescia A, Tomassini F, Berardi G, Pezzatini M, Dall’Oglio A, Pindozzi F et al (2016) Post-incisional ventral hernia repair in patients undergoing chemotherapy: improving outcomes with biological mesh. World J Surg Oncol 14(1):257. https://doi.org/10.1186/s12957-016-1011-5
Chamieh J, Tan WH, Ramirez R, Nohra E, Apakama C, Symons W (2017) Synthetic versus biologic mesh in single-stage repair of complex abdominal wall defects in a contaminated field. Surg Infect 18(2):112–118. https://doi.org/10.1089/sur.2016.106
de Vries FEE, Hodgkinson JD, Claessen JJM, van Ruler O, Leo CA, Maeda Y et al (2020) Long-term outcomes after contaminated complex abdominal wall reconstruction. Hernia 24(3):459–468. https://doi.org/10.1007/s10029-020-02124-7
DeNoto G, Reaven N, Funk S (2013) Ventral hernia: retrospective cost analysis of primary repair, repair with synthetic mesh, and repair with acellular xenograft implant. Open Access Surg 6:23–32. https://doi.org/10.2147/OAS.S44647
El-Gazzaz G, Erem HH, Aytac E, Salcedo L, Stocchi L, Kiran RP (2013) Risk of infection and hernia recurrence for patients undergoing ventral hernia repair with non-absorbable or biological mesh during open bowel procedures. Tech Coloproctol 17(3):315–320. https://doi.org/10.1007/s10151-012-0928-0
Finch D, Mehmood S, Varghese J (2021) Abdominal wall reconstruction using biosynthetic absorbable mesh in high-risk complex ventral hernia. Swiss Med Wkly. https://doi.org/10.4414/smw.2021.20449
Fischer JP, Basta MN, Mirzabeigi MN, Kovach SJ III (2014) A comparison of outcomes and cost in VHWG grade II hernias between Rives-Stoppa synthetic mesh hernia repair versus underlay biologic mesh repair. Hernia 18(6):781–789. https://doi.org/10.1007/s10029-014-1309-6
Koscielny A, Widenmayer S, May T, Kalff J, Lingohr P (2017) Comparison of biological and alloplastic meshes in ventral incisional hernia repair. Langenbecks Arch Surg 403(2):255–263. https://doi.org/10.1007/s00423-017-1639-9
López-Cano M, Teresa Quiles M, Antonio Pereira J, Armengol-Carrasco M, Arbós Vía MA (2017) Complex abdominal wall hernia repair in contaminated surgical fields: factors affecting the choice of prosthesis. Am Surg 83(6):583–590. https://doi.org/10.1177/000313481708300622
Messa CA, Kozak G, Broach RB, Fischer JP (2019) When the mesh goes away: an analysis of poly-4-hydroxybutyrate mesh for complex hernia repair. Plast Reconstr Surg Glob Open 7(11):e2576. https://doi.org/10.1097/GOX.0000000000002576
Nockolds CL, Hodde JP, Rooney PS (2014) Abdominal wall reconstruction with components separation and mesh reinforcement in complex hernia repair. BMC Surg 14(1):1–7. https://doi.org/10.1186/1471-2482-14-25
Pakula A, Skinner R (2020) Outcomes of open complex ventral hernia repairs with retromuscular placement of poly-4-hydroxybutyrate bioabsorbable mesh. Surg Innov 27(1):32–37. https://doi.org/10.1177/1553350619881066
Sahoo S, Haskins IN, Huang L-C, Krpata DM, Derwin KA, Poulose BK et al (2017) Early wound morbidity after open ventral hernia repair with biosynthetic or polypropylene mesh. J Am Coll Surg 225(4):472–480. https://doi.org/10.1016/j.jamcollsurg.2017.07.1067
Renard Y, de Mestier L, Henriques J, de Boissieu P, de Mestier P, Fingerhut A et al (2020) Absorbable polyglactin vs. non-cross-linked porcine biological mesh for the surgical treatment of infected incisional hernia. J Gastrointest Surg 24(2):435–443. https://doi.org/10.1007/s11605-018-04095-8
Plymale MA, Davenport DL, Dugan A, Zachem A, Roth JS (2018) Ventral hernia repair with poly-4-hydroxybutyrate mesh. Surg Endosc 32(4):1689–1694. https://doi.org/10.1007/s00464-017-5848-7
Mellia JA, Othman S, Naga HI, Messa CA, Elfanagely O, Byrnes YM et al (2020) Outcomes of poly-4-hydroxybutyrate mesh in ventral hernia repair: a systematic review and pooled analysis. Plast Reconstr Surg Glob Open 8(12):e3158. https://doi.org/10.1097/GOX.0000000000003158
Hodgkinson JD, Maeda Y, Leo CA, Warusavitarne J, Vaizey CJ (2017) Complex abdominal wall reconstruction in the setting of active infection and contamination: a systematic review of hernia and fistula recurrence rates. Colorectal Dis 19(4):319–330. https://doi.org/10.1111/codi.13609
Samson DJ, Gachabayov M, Latifi R (2021) Biologic mesh in surgery: a comprehensive review and meta-analysis of selected outcomes in 51 studies and 6079 patients. World J Surg 45(12):3524–3540. https://doi.org/10.1007/s00268-020-05887-3
Barker TH, Migliavaca CB, Stein C, Colpani V, Falavigna M, Aromataris E et al (2021) Conducting proportional meta-analysis in different types of systematic reviews: a guide for synthesisers of evidence. BMC Med Res Methodol 21(1):189. https://doi.org/10.1186/s12874-021-01381-z
Parker SG, Mallett S, Quinn L, Wood CPJ, Boulton RW, Jamshaid S et al (2021) Identifying predictors of ventral hernia recurrence: systematic review and meta-analysis. BJS Open 5(2):zraa071. https://doi.org/10.1093/bjsopen/zraa071
Acknowledgements
Funding was provided by Asociación Española de Cirujanos.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This work received financial support from the Spanish Association of Surgeons (AEC). The authors declare that they have the following conflicts of interest: S. Morales-Conde has received honoraria for consultancy, lectures, support for travels and participation in review activities from BD-Bard, Medtronic, Johnson & Johnson, Gore, BBraun, Dipro, Baxter, Olympus, Stryker and Storz. M. López-Cano has received honoraria for consultancy, lectures, support for travels and participation in review activities from BD-Bard, Medtronic, Gore and Abex. P. Hernández-Granados has received honoraria for consultancy and lectures from BD and BBraun. M. Verdaguer does not report conflicts of interest. L. Tallón-Aguilar has received honoraria for lectures and support for travels from Medtronic, Johnson & Johnson, Gore, Dynamesh, Vifor Pharma, Rovi and Abex.
Human and animal rights, and Informed consent
Since it is a proportional meta-analysis, we consider that it does not require an ethical approval, human and animal rights or informed consent. We do not use confidential information, only public information collected in the articles included in the review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original article has been updated: Due to retrospective open choice request.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Morales-Conde, S., Hernández-Granados, P., Tallón-Aguilar, L. et al. Ventral hernia repair in high-risk patients and contaminated fields using a single mesh: proportional meta-analysis. Hernia 26, 1459–1471 (2022). https://doi.org/10.1007/s10029-022-02668-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10029-022-02668-w