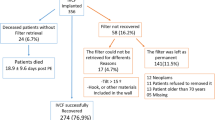
The TrapEase filter is a permanent, symmetric nitinol filter that can be deployed through a low-profile sheath. Although the TrapEase is enjoying an increasing market share of inferior vena cava (IVC) filters, there are still limited clinical follow-up data on its use. This study is a retrospective review of 189 consecutive infrarenal TrapEase filters placed at our institution. The study included 80 men and 109 women, with an average age of 73 years (24-102). The most common indication for filter placement was a contraindication to warfarin. In total, 13% of the patients were treated with warfarin. The filter was successfully deployed in all cases, via the right femoral vein in 57% of the patients, via the left femoral vein in 32%, and via the right internal jugular vein in 11%. Follow-up studies were performed as clinically indicated. During the investigation, there were three cases of IVC thrombosis (1.5%) and one case of retroperitoneal hemorrhage potentially caused by filter placement. Two of the thrombosis cases resulted in serious sequelae. This study also represents the first report of a symptomatic pulmonary embolism (PE) after TrapEase filter placement. While this investigation does demonstrate a low overall complication rate of this new device, it raises the concern of an increase in IVC thrombosis rate.
Similar content being viewed by others
Author information
Authors and Affiliations
About this article
Cite this article
Schutzer, R., Ascher, E., Hingorani, A. et al. Preliminary Results of the New 6F TrapEase Inferior Vena Cava Filter . Ann Vasc Surg 17, 103–106 (2003). https://doi.org/10.1007/s10016-001-0328-9
Issue Date:
DOI: https://doi.org/10.1007/s10016-001-0328-9