Abstract
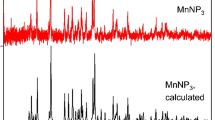
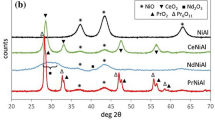
Electrochemical dissolution of immobilised microparticulate Mn(III,IV) oxides in slightly acidic solution (pH 4.4) was found to be a very general reaction, which is responsible for well-defined voltammetric peaks. Dissolution of six Mn(III,IV) oxides is initiated by the reduction of Mn(IV) to Mn(III) in the solid phase, which is followed by a massive dissolution via further reduction of Mn(III) to Mn(II), which finally yields soluble Mn2+. The reactivity of manganese oxides depends on their structure: the most reactive are amorphous (δ-MnO2) and layered structures (birnessite); more resistant toward reductive dissolution are α- and λ-MnO2 and electrochemical manganese dioxide; and least reactive is β-MnO2. Reductive dissolution of LiMn2O4 resembles that of λ-MnO2, whereas CaMnO3 dissolves via a different reaction mechanism.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 11 October 1999 / Accepted: 2 December 1999
Rights and permissions
About this article
Cite this article
Bakardjieva, S., Bezdička, P., Grygar, T. et al. Reductive dissolution of microparticulate manganese oxides. J Solid State Electrochem 4, 306–313 (2000). https://doi.org/10.1007/s100089900104
Issue Date:
DOI: https://doi.org/10.1007/s100089900104