Abstract
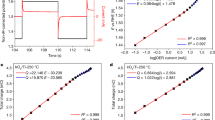
Charge-transfer resistance [R ct = (dη/di)η = 0] and Tafel plots of current density (i) versus overpotential (η) data are generally known to yield values of the energy-transfer coefficient (α) and exchange current density (i o) of an electrochemical reaction. In the present investigation, the resistance (dη/di)η≠0 that could be calculated by differentiating a wide range of i−η curves was also shown to provide the values of α and i o, by plotting ln(dη/di)η≠0 against η. Since α and i o could also be evaluated directly from the experimental DC polarization data, the procedure was not of significant importance. Nevertheless, it was considered important in evaluating α and i o from AC impedance data, because the procedure was based on data analysis, which was much simpler than that reported in the literature. A cobalt electrode prepared from fine metal powder was used in 1 M KOH electrolyte and the hydrogen evolution reaction was studied by AC impedance at several potentials. The resistance values measured from the complex plane impedance diagram were plotted against the potential, and the values of α and i o were evaluated.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 8 October 1998 / Accepted: 11 January 1999
Rights and permissions
About this article
Cite this article
Elumalai, P., Vasan, H. & Munichandraiah, N. A note on overpotential dependence of AC impedance data. J Solid State Electrochem 3, 470–473 (1999). https://doi.org/10.1007/s100080050183
Issue Date:
DOI: https://doi.org/10.1007/s100080050183