Abstract
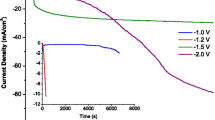
Conductive polypyrrole (PPy) films and PPy films containing Ge microparticles were synthesized by anodic oxidation of pyrrole in acidic nitrate solutions using a bare passivated titanium electrode. Well-adhering black PPy films were obtained both under galvanostatic and potentiodynamic polarization. After the formation of the PPy film, during the first anodic cycle, an increase of the anodic deposition current with the number of cycles was observed, revealing the increase of conductivity of the growing film. The variations of the electrode surface area were estimated by impedance spectroscopy measurements. The kinetics of the PPy film formation is controlled by diffusion of the Py monomer in the solution. The diffusion coefficient, estimated by two different methods, was ca. 2×10–6 cm2 s–1. The reduction rate of oxygen and protons at the Ti/PPy/Ge electrodes depends on how the Ge microparticles are incorporated in the PPy film. Optimum conditions for this incorporation are realized with thin PPy films and high Ge loading. Thermogravimetric analysis shows that the PPy film containing Ge microparticles is more thermally stable than the blank PPy film.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Mokrane, S., Makhloufi, L., Hammache, H. et al. Electropolymerization of polypyrrole, modified with germanium, on a passivated titanium electrode in aqueous nitrate solution: new results on catalytic reduction of protons and dissolved oxygen. J Solid State Electrochem 5, 339–347 (2001). https://doi.org/10.1007/s100080000161
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s100080000161