Abstract
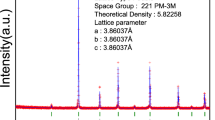
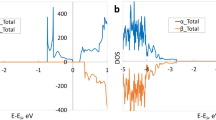
Oxygen nonstoichiometry of perovskite-like SrFe1–x Si x O3–δ (x = 0.05–0.20), studied by thermogravimetric analysis and coulometric titration in the oxygen partial pressure range 10−20–0.5 atm at 700–950 °C, decreases with Si4+ additions. The equilibrium \( {p}_{{\mathrm{O}}_2} \)–T−δ diagrams can be adequately described by a model accounting for anion site-exclusion effects near highly stable SiO4 tetrahedra and energetic favorability of the defect clusters formed by two tetrahedra sharing one oxygen vacancy. This model was validated by atomistic computer simulations. The standard thermodynamic functions for oxygen incorporation and iron disproportionation reactions are essentially independent of silicon concentration, as for the migration activation energies of the p- and n-type electronic charge carriers. On the contrary, at low temperatures, Si-doping leads to a higher oxygen deficiency, simultaneously suppressing long-range vacancy ordering and increasing oxygen coordination of iron cations as estimated from the Mössbauer spectra. These phenomena are associated, again, with vacancy trapping near randomly distributed Si4+. The Mössbauer spectroscopy, transmission electron microscopy, and electron diffraction studies showed that Si4+ substitution progressively reduces the content of brownmillerite-like nanodomains typical for SrFeO3-based materials.














Similar content being viewed by others
References
Lee JH, Mori T, Li JG, Ikegami T, Komatsu M, Haneda H (2000) J Electrochem Soc 147:2822–2829
Kharton VV, Marques FMB, Tsipis EV, Viskup AP, Patrakeev MV, Harkavy AV, Samigullina R, Frade JR (2003) Ionics 9:122–126
Viitanen MM (2002) Welzenis RGv, Brongersma HH, Van Berkel FPF. Solid State Ionics 150:223–228
Liu Y, Zhu X, Li M, Li W, Yang W (2015) J Membrane Sci 492:173–180
Perz M, Bucher E, Gspan C, Waldhausl J, Hofer F, Sitte W (2016) Solid State Ionics 288:22–27
Tsipis EV, Kharton VV, Frade JR (2007) Electrochim Acta 52:4428–4435
Schuler JA, Wuillemin Z, Hessler-Wyser A (2011) Van herle J. Electrochem Solid-State Lett 14:B20–B22
Porras-Vazquez JM, Smith RI, Slater PR (2014) J Solid State Chem 213:132–137
Hancock CA, Slater PR (2011) Dalton Trans 40:5599–5603
Porras-Vazquez JM, Pike T, Hancock CA, Marco JF, Berrya FJ, Slater PR (2013) J Mater Chem A1:11834–11841
Porras-Vazquez JM, Losilla ER, Keenan PJ, Hancock CA, Kemp TF, Hanna JV, Slater PR (2013) Dalton Trans 42:5421–5429
Merkulov OV, Markov AA, Patrakeev MV, Chukin AV, Leonidov IA, Kozhevnikov VL (2016) Solid State Ionics 292:83–87
Mizusaki J, Sasamoto T, Cannon WR, Bowen HK (1983) J Am Ceram Soc 66:247–252
Kobayashi K, Yamaguchi S, Mukaida M, Tsunoda T (2001) Solid State Ionics 144:315–320
Samsonov G (ed) (1982) The oxide handbook. Springer, NY
Draper NR, Smith H (1981) Applied regression analysis. John Wiley & Sons, New York
CaRIne Crystallography 4.0, Cyrille Boudias & Daniel Monceau. http://carine.crystallography.pagespro-orange.fr/. Accessed 22 June 2017
Waerenborgh JC, Rojas DP, Shaula AL, Mather GC, Patrakeev MV, Kharton VV, Frade JR (2005) Mater Lett 59:1644–1648
Waerenborgh JC, Tsipis EV, Yaremchenko AA, Kharton VV (2011) J Solid State Chem 184:2610–2614
Patrakeev MV, Leonidov IA, Kozhevnikov VL (2011) J Solid State Electrochem 15:931–954
Merkulov OV, Naumovich EN, Patrakeev MV, Markov AA, Bouwmeester HJM, Leonidov IA, Kozhevnikov VL (2016) Solid State Ionics 292:116–121
Markov AA, Chesnokov KY, Patrakeev MV, Leonidov IA, Chukin AV, Leonidova ON, Kozhevnikov VL (2016) J Solid State Electrochem 20:225–234
Gale JD (1997) J Chem Soc Faraday Trans 93:629–637
Gale JD, Rohl AL (2003) Mol Simul 29:291–234
Patrakeev MV, Kharton VV, Bakhteeva YA, Shaula AL, Leonidov IA, Kozhevnikov VL, Naumovich EN, Yaremchenko AA, Marques FMB (2006) Solid State Sci 8:476–487
Nakayama N, Takano M, Inamura S, Nakanishi N, Kosuge K (1987) J Solid State Chem 71:403–417
Alario-Franco MA, Gonzalez-Calbet JM, Vallet-Regi M (1983) J Solid State Chem 49:219–231
Gallagher PK, McChesney JB, Buchanan DNE (1964) J Chem Phys 41:2429–2433
Waerenborgh JC, Tsipis EV, Auckett JE, Ling CD, Kharton VV (2013) J Solid State Chem 205:5–9
Yaremchenko AA, Tsipis EV, Kovalevsky AV, Waerenborgh JC, Kharton VV (2011) Solid State Ionics 192:259–268
Patrakeev MV, Markov AA, Shalaeva EV, Tsipis EV, Waerenborgh JC, Kharton VV, Leonidov IA, Kozhevnikov VL (2013) Solid State Ionics 244:17–22
Binomial Distribution (2003) e–Handbook of Statistical Methods, NIST/SEMATECH. http://www.itl.nist.gov/div898/handbook/. Accessed 8 Feb 2017
Markov AA, Shalaeva EV, Tyutyunnik AP, Kuchin VV, Patrakeev MV, Leonidov IA, Kozhevnikov VL (2013) J Solid State Chem 197:191–197
Patrakeev MV, Leonidov IA, Kozhevnikov VL, Kharton VV (2004) Solid State Sci 6:907–913
Markov AA, Savinskaya OA, Patrakeev MV, Nemudry AP, Leonidov IA, Pavlyukhin YT, Ishchenko AV, Kozhevnikov VL (2009) J Solid State Chem 182:799–806
Shannon RD (1976) Acta Crystallogr A 32:751–767
Lewis V, Catlow CRA (1985) J Phys C Solid State Phys 18:1149–1161
Cherry M, Islam MS, Catlow CRA (1995) J Solid State Chem 118:125–132
Woodley SM, Battle PD, Gale JD, Richard C, Catlow A (1999) Phys Chem Chem Phys 1:2535–2542
Acknowledgments
Financial support from the Russian Foundation for Basic Research (grant 17-08-01029), Ministry of Education and Science of the Russian Federation (project 14.B25.31.0018), Russian Science Foundation (project 17-79-30071), FCT, Portugal (projects UID/EMS/00481/2013 and UID/Multi/04349/2013), and FEDER, Portugal (project CENTRO-01-0145-FEDER-022083) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Merkulov, O.V., Naumovich, E.N., Patrakeev, M.V. et al. Defect formation, ordering, and transport in SrFe1–x Si x O3–δ (x = 0.05–0.20). J Solid State Electrochem 22, 727–737 (2018). https://doi.org/10.1007/s10008-017-3797-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-017-3797-7