Abstract
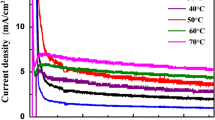
Through introducing appropriate additives into the electrolytes, the morphology and growth efficiency of TiO2 nanotube arrays (TNTAs) have been greatly influenced. The anodizing current transients and the corresponding morphology of TNTAs were investigated and compared in detail by SEM. To further understand the mechanism, the measured current-time curves obtained during the anodization of titanium in the electrolytes with different additives are simulated. Notably, in the total anodizing current, the ionic current is separated from the electronic current according to the present model, and that the electronic current and ionic current make different contributions to the growth of TNTAs. It is found that the initiation of nanopores may be caused by the rupture of the oxygen bubbles occluded in the growing oxide, and the opening of nanotubes is thought to be close related to the disturbance effect of the rising bubbles (caused by electronic current). The present results would be helpful for understanding the formation mechanism of TNTAs from the perspective of ionic and electronic current. And practically, the nanotube length can be predicted and deduced quantitatively via simulating and comparing electronic and ionic current.






Similar content being viewed by others
References
Zwilling V, Aucouturier M, Darque-Ceretti E (1999) Electrochim Acta 45:921–929
Li D, Chang PC, Chien CJ, Lu JG (2010) Chem Mater 22:5707–5711
Hsu HL, Tien CF, Leu J (2014) J Solid State Electr: 1–7
Chen B, Hou J, Lu K (2013) Langmuir 29:5911–5919
Chen K, Feng X, Hu R, Li Y, Xie K, Li Y, Gu H (2013) J Alloys Compd 554:72–79
Chen Q, Liu H, Xin Y, Cheng X, Zhang J, Li J, Wang P, Li H (2013) Electrochim Acta 99:152–160
Mazzarolo A, Lee K, Vicenzo A, Schmuki P (2012) Electrochem Commun 22:162–165
Yu L, Wang Z, Zhang L, Wu HB, Lou XW (2013) J Mater Chem A 1:122–127
Roy P, Berger S, Schmuki P (2011) Angew Chem Int Ed 50:2904–2939
Guan D, Wang Y (2012) Nanoscale 4:2968–2977
Gui Q, Yu D, Zhang S, Xiao H, Yang C, Song Y, Zhu X (2014) J Solid State Electrochem 18:141–148
Chen B, Lu K (2012) Langmuir 28:2937–2943
Jo Y, Jung I, Lee I, Choi J, Tak Y (2010) Electrochem Commun 12:616–619
So S, Lee K, Schmuki P (2012) J Am Chem Soc 134:11316–11318
Liu G, Hoivik N, Wang K, Jakobsen H (2011) J Mater Sci 46:7931–7935
Liu G, Wang K, Hoivik N, Jakobsen H (2012) Sol Energy Mater Sol Cells 98:24–38
Lai CW, Sreekantan S, Lockman Z (2012) J Nanosci Nanotechnol 12:4057–4066
Wang Y, Wu Y, Qin Y, Xu G, Hu X, Cui J, Zheng H, Hong Y, Zhang X (2011) J Alloys Compd 509:L157–L160
Krengvirat W, Sreekantan S, Noor AFM, Kawamura G, Muto H, Matsuda A (2013) Electrochim Acta 89:585–593
Sreekantan S, Wei LC, Lockman Z (2011) J Electrochem Soc 158:C397–C402
Allam NK, Shankar K, Grimes CA (2008) J Mater Chem 18:2341–2348
Hebert KR, Albu SP, Paramasivam I, Schmuki P (2012) Nat Mater 11:162–166
LeClere DJ, Velota A, Skeldon P, Thompson GE, Berger S, Kunze J, Schmuki P, Habazaki H, Nagata S (2008) J Electrochem Soc 155:C487–C494
Regonini D, Satka A, Jaroenworaluck A, Allsopp DWE, Bowen CR, Stevens R (2012) Electrochim Acta 74:244–253
Mazzarolo A, Curioni M, Vicenzo A, Skeldon P, Thompson GE (2012) Electrochim Acta 75:288–295
Diggle JW, Downie TC, Goulding CW (1969) Chem Rev 69:365–405
Zhu XF, Han H, Song Y, Ma HT, Qi WX, Lu C, Xu C (2012) Acta Phys Sin 61:228202
Houser JE, Hebert KR (2009) Nat Mater 8:415–420
Patermarakis G, Moussoutzanis K (2009) Electrochim Acta 54:2434–2443
Albella JM, Montero I, Martínez-Duart JM (1987) Electrochim Acta 32:255–258
Al-Abdullah ZTY, Shin Y, Kler R, Perry CC, Zhou W, Chen Q (2010) Nanotechnology 21:505601
Pauric AD, Baig SA, Pantaleo AN, Wang Y, Kruse P (2013) J Electrochem Soc 160:C12–C18
Song Y, Zhu X, Wang X, Che J, Du Y (2001) J Appl Electrochem 31:1273–1279
Yasuda K, Macak JM, Berger S, Ghicov A, Schmuki P (2007) J Electrochem Soc 154:C472–C478
Sreekantan S, Saharudin KA, Lockman Z, Tzu TW (2010) Nanotechnology 21:365603
Li H, Wang J, Huang K, Sun G, Zhou M (2011) Mater Lett 65:1188–1190
Yu DL, Song Y, Zhu XF, Yang RQ, Han AJ (2013) Appl Surf Sci 276:711–716
Valota A, Curioni M, Leclere DJ, Skeldon P, Falaras P, Thompson GE (2010) J Electrochem Soc 157:K243–K247
Yang RQ, Jiang LF, Zhu XF, Song Y, Yu DL, Han AJ (2012) RSC Adv 2:12474–12481
Zhu XF, Song Y, Liu L, Wang CY, Zheng J, Jia HB, Wang XL (2009) Nanotechnology 20:475303
Zhu XF, Song Y, Yu DL, Zhang CS, Yao W (2013) Electrochem Commun 29:71–74
Macak JM, Tsuchiya H, Schmuki P (2005) Angew Chem Int Ed 44:2100–2102
Arabatzis IM, Falaras P (2003) Nano Lett 3:249–251
Acknowledgments
This work was supported financially by the National Natural Science Foundation of China (grant nos. 61171043, 51377085), the National Science and Technology Major Project of the Ministry of Science and Technology of China (2009ZX01021-002), and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yu, D., Zhang, S., Zhu, X. et al. Simulation of anodizing current-time curves and morphology evolution of TiO2 nanotube arrays. J Solid State Electrochem 18, 2609–2617 (2014). https://doi.org/10.1007/s10008-014-2513-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-014-2513-0