Abstract
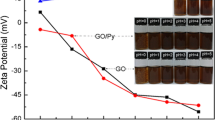
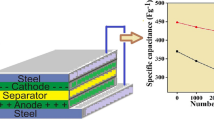
The layered polypyrrole-graphene oxide-sodium dodecylbenzene sulfonate (PPyGO-SDBS) nanocomposites were facilely fabricated via an in situ emulsion polymerization method with the assistance of SDBS as dopant and stabilizer. Scanning electron microscopy (SEM), transmission electron microscopy (TEM), Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), and electrochemical performance were employed to analyze the structure and the characteristics of the composites. The results showed that SDBS played an important role in improving the electrochemical performance of the PPyGO-SDBS, by dispersing the PPy between the layers of the GO. The obtained PPyGO-SDBS exhibited remarkable performance as an electrode material for supercapacitors, with a specific capacitance as high as 483 F g−1 at a current density of 0.2 A g−1 when the mass ratio of pyrrole to GO was 80:20. The attenuation of the specific capacitance was less than 20 % after 1,000 charge–discharge processes, supporting the idea that PPy inserted successfully into the GO interlayers. The excellent electrochemical performance seemed to arise from the synergistic effect between the PPy and the GO and the dispersion of the PPy induced by SDBS.







Similar content being viewed by others
References
Miller JR, Simon P (2008) Electrochemical capacitors for energy management. Science 321:651–652
Winter M, Brodd RJ (2004) What are batteries, fuel cells, and supercapacitors? Chem Rev 104:4245–4269
Fuertes AB, Lota G, Centeno TA, Frackowiak E (2005) Templated mesoporous carbons for supercapacitor application. Electrochim Acta 50:2799–2805
Simon P, Gogotsi Y (2008) Materials for electrochemical capacitors. Nat Mater 7:845–854
Burke A (2000) Ultracapacitors: why, how, and where is the technology. J Power Sources 91:37–50
Zhang LL, Zhao XS (2009) Carbon-based materials as supercapacitor electrodes. Chem Soc Rev 38:2520–2531
Gogotsi Y, Simon P (2011) True performance metrics in electrochemical energy storage. Science 334:917–918
Kuilla T, Bhadra S, Yao D, Kim NH, Bose S, Lee JH (2010) Recent advances in graphene based polymer composites. Prog Polym Sci 35:1350–1375
Dékány I, Krüger-Grasser R, Weiss A (1998) Selective liquid sorption properties of hydrophobized graphite oxide nanostructures. Colloid Polym Sci 276:570–576
Bourlinos AB, Gournis D, Petridis D, Szabó T, Szeri A, Dékány I (2003) Graphite oxide: chemical reduction to graphite and surface modification with primary aliphatic amines and amino acids. Langmuir 19:6050–6055
Liu N, Luo F, Wu H, Liu Y, Zhang C, Chen J (2008) One-step ionic-liquid-assisted electrochemical synthesis of ionic-liquid-functionalized graphene sheets directly from graphite. Adv Funct Mater 18:1518–1525
Zhuang XD, Chen Y, Liu G, Li PP, Zhu CX, Kang ET, Noeh KG, Zhang B, Zhu JH, Li YX (2010) Conjugated-polymer-functionalized graphene oxide: synthesis and nonvolatile rewritable memory effect. Adv Mater 22:1731–1735
Liu P, Gong K, Xiao P, Xiao M (2000) Preparation and characterization of poly(vinyl acetate)-intercalated graphite oxide nanocomposite. J Mater Chem 10:933–935
Zhang X, Yang W, Ma Y (2009) Synthesis of polypyrrole-intercalated layered manganese oxide nanocomposite by a delamination/reassembling method and its electrochemical capacitance performance. Electrochem Solid-State Lett 12:A95–A98
Zhang LL, Zhao S, Tian XN, Zhao XS (2010) Layered graphene oxide nanostructures with sandwiched conducting polymers as supercapacitor electrodes. Langmuir 26:17624–17628
Zhang K, Zhang LL, Zhao XS, Wu J (2010) Graphene/polyaniline nanofiber composites as supercapacitor electrodes. Chem Mater 22:1392–1401
Feng H, Wang B, Tan L, Chen N, Wang N, Chen B (2014) Polypyrrole/hxadecylpyridinium chloride-modified graphite oxide composites: fabrication, characterization, and application in supercapacitors. J Power Sources 246:621–628
Konwer S, Boruah R, Dolui SK (2011) Studies on conducting polypyrrole/graphene oxide composites as supercapacitor electrode. J Electron Mater 40:2248–2255
Gu Z, Zhang L, Li C (2009) Preparation of highly conductive polypyrrole/graphite oxide composites via in situ polymerization. J Macromol Sci B 48:1093–1102
Gu Z, Li C, Wang G, Zhang L, Li X, Wang W, Jin S (2010) Synthesis and characterization of polypyrrole/graphite oxide composite by in situ emulsion polymerization. J Polym Sci B Polym Phys 48:1329–1335
Li Y, Wu Y (2009) Coassembly of graphene oxide and nanowires for large-area nanowire alignment. J Am Chem Soc 131:5851–5857
Kim J, Cote LJ, Kim F, Yuan W, Shull KR, Huang J (2010) Graphene oxide sheets at interfaces. J Am Chem Soc 132:8180–8186
Chang H, Wang G, Yang A, Tao X, Liu X, Shen Y, Zheng Z (2010) A transparent, flexible, low-temperature, and solution-processible graphene composite electrode. Adv Funct Mater 20:2893–2902
Zhu C, Guo S, Fang Y, Dong S (2010) Reducing sugar: new functional molecules for the green synthesis of graphene nanosheets. ACS Nano 4:2429–2437
Zhu C, Guo S, Fang Y, Han L, Wang E, Dong S (2011) One-step electrochemical approach to the synthesis of graphene/MnO2 nanowall hybrids. Nano Res 4:648–657
Zhu C, Guo S, Wang P, Xing L, Fang Y, Zhai Y, Dong S (2010) One-pot, water-phase approach to high-quality graphene/TiO2 composite nanosheets. Chem Commun 46:7148–7150
Bose S, Kim NH, Kuila T, Lau KT, Lee JH (2011) Electrochemical performance of a graphene-polypyrrole nanocomposite as a supercapacitor electrode. Nanotechnology 22:295202–295210
Bose S, Kuila T, Uddin ME, Kim NH, Lau AKT, Lee JH (2010) In-situ synthesis and characterization of electrically conductive polypyrrole/graphene nanocomposites. Polymer 51:5921–5928
Bora C, Dolui SK (2012) Fabrication of polypyrrole/graphene oxide nanocomposites by liquid/liquid interfacial polymerization and evaluation of their optical, electrical and electrochemical properties. Polymer 53:923–932
Li L, Xia K, Li L, Shang S, Guo Q, Yan G (2012) Fabrication and characterization of free-standing polypyrrole/graphene oxide nanocomposite paper. J Nanoparticle Res 14:1–8
Zhang D, Zhang X, Chen Y, Yu P, Wang C, Ma Y (2011) Enhanced capacitance and rate capability of graphene/polypyrrole composite as electrode material for supercapacitors. J Power Sources 196:5990–5996
Zhu C, Zhai J, Wen D, Dong S (2012) Graphene oxide/polypyrrole nanocomposites: one-step electrochemical doping, coating and synergistic effect for energy storage. J Mater Chem 22:6300–6306
Pandey GP, Hashmi SA, Kumar Y (2010) Multiwalled carbon nanotube electrodes for electrical double layer capacitors with ionic liquid based gel polymer electrolytes. J Electrochem Soc 157:A105–A114
Pandey GP, Hashmi SA, Kumar Y (2010) Performance studies of activated charcoal based electrical double layer capacitors with ionic liquid gel polymer electrolytes. Energy Fuels 24:6644–6652
Zhang K, Mao L, Zhang LL, Chan HSO, Zhao XS, Wu J (2011) Surfactant-intercalated, chemically reduced graphene oxide for high performance supercapacitor electrodes. J Mater Chem 21:7302–7307
Li L, Qiu J, Wang S (2013) Three-dimensional ordered nanostructures for supercapacitor electrode. Electrochim Acta 99:278–284
Wang YG, Li HQ, Xia YY (2006) Ordered whiskerlike polyaniline grown on the surface of mesoporous carbon and its electrochemical capacitance performance. Adv Mater 18:2619–2623
Wang G, Huang J, Chen S, Gao Y, Cao D (2011) Preparation and supercapacitance of CuO nanosheet arrays grown on nickel foam. J Power Sources 196:5756–5760
Jaidev RS (2012) Poly(p-phenylenediamine)/graphene nanocomposites for supercapacitor applications. J Mater Chem 22:18775–18738
Acknowledgments
This study was supported by the College Scientific Plan Fund of Shandong Education Department (J10LD23) and the Doctoral Startup Foundation of Qilu University of Technology (12042826).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Li, M., Yang, L. et al. Facilely prepared polypyrrole-graphene oxide-sodium dodecylbenzene sulfonate nanocomposites by in situ emulsion polymerization for high-performance supercapacitor electrodes. J Solid State Electrochem 18, 2139–2147 (2014). https://doi.org/10.1007/s10008-014-2469-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-014-2469-0