Abstract
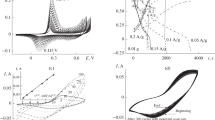
Nickel oxide (NiOx) and polyaniline (PAni) were electrocodeposited from NiSO4 and aniline through cyclic voltammetric scans to afford PAni–NiOx composite film at controlled pH environment. The electrochemical activities of the film were investigated by cyclic voltammetry in 0.1 M NaOH and 0.1 M H2SO4, respectively. Typical redox couples of PAni in 0.1 M H2SO4 appeared at approximately 0.2 and 0.4 V vs. saturated calomel electrode (SCE); Ni(II)/Ni(III) redox couple was observed at approximately 0.4 V vs. SCE in 0.1 M NaOH. The morphologies and elemental components of the films were inspected by scanning electron microscopy and energy dispersive X-ray diffraction. The stability of nickel oxide in the films was found to be enhanced against acidic environments. Electrochemical catalytic behavior of NiOx within the composite film was conserved and demonstrated by catalytic oxidation of methanol and ethanol.












Similar content being viewed by others
References
Pedro G-R (2001) Adv Mater 13:163. doi:10.1002/1521-4095(200102)13:3<163::AID-ADMA163>3.0.CO;2-U
Subramania A, Devi S-L (2008) Polym Adv Technol 19:725. doi:10.1002/pat.1016
Shen P-K, Huang H-T, Tseung A-C-C (1992) J Electrochem Soc 139:1840. doi:10.1149/1.2069508
Karlsson J, Roos A (2000) Sol Energy 68:493. doi:10.1016/S0038-092X(00)00021-9
Berchmans S, Gomathi H, Rao G-P (1995) J Electroanal Chem 394:267. doi:10.1016/0022-0728(95)04099-A
Cataldi T-R-I, Centonze D (1995) Anal Chim Acta 307:43. doi:10.1016/0003-2670(95)00039-3
Nam K-W, Yoon W-S, Kim K-B (2002) Electrochim Acta 47:3201. doi:10.1016/S0013-4686(02)00240-2
Song G-P, Bo J, Guo R (2005) Colloid Polym Sci 283:677. doi:10.1007/s00396-004-1205-1
Song G-P, Han J, Guo R (2007) Synth Met 157:170. doi:10.1016/j.synthmet.2006.12.007
Han J, Song G-P, Guo R (2006) J Polym Sci Part Polym Chem 44:4229. doi:10.1002/pola.21512
Sun L-J, Liu X-X (2008) Eur Polym J 44:219. doi:10.1016/j.eurpolymj.2007.10.017
Liu X-X, Bian L-J, Zhang L, Zhang L-J (2007) J Solid State Electrochem 11:1279. doi:10.1007/s10008-007-0287-3
Hu C-C, Chu C-H (2000) Mater Chem Phys 65:329. doi:10.1016/S0254-0584(00)00254-6
Liu X-X, Zhang L, Li Y-B, Bian L-J, Su Z, Zhang L-J (2005) J Mater Sci 40:4511. doi:10.1007/s10853-005-0854-x
Liu X-X, Zhang L, Li Y-B, Bian L-J, Huo Y-Q, Su Z (2006) Polym Bull 57:825. doi:10.1007/s00289-006-0653-9
Sun L-J, Liu X-X, Lau K-K-T, Chen L, Gu W-M (2007) Electrochim Acta 53:3036. doi:10.1016/j.electacta.2007.11.034
Casella I-G, Spera R (2005) J Electroanal Chem 578:55. doi:10.1016/j.jelechem.2004.11.043
Tench D, Warren L-F (1983) J Electrochem Soc 130:869. doi:10.1149/1.2119838
Prasad K-R, Miura N (2004) Appl Phys Lett 85:4199. doi:10.1063/1.1814816
Chigane M, Ishikawa M (1992) J Chem Soc Faraday Trans 88:2203. doi:10.1039/ft9928802203
Yang M-C, Lin C-K, Su C-L (1995) J Electrochem Soc 142:1189. doi:10.1149/1.2044150
Peng X-Y, Li W, Liu X-X, Hua P-J (2007) J Appl Polym Sci 105:2260. doi:10.1002/app.26227
Gospodinova N, Terlemezyan L (1998) Prog Polym Sci 23:1443. doi:10.1016/S0079-6700(98)00008-2
Duic L, Mandic Z, Kovac S (1995) Electrochim Acta 40:1681. doi:10.1016/0013-4686(95)00086-T
Pramanik P, Bhattacharya S (1990) J Electrochem Soc 137:3869. doi:10.1149/1.2086316
Huang W-S, Humphrey B-D, MacDiarmid A-G (1986) J Chem Soc Faraday Trans 82:2385. doi:10.1039/f19868202385
George M-V, Balachandran K-S (1975) Chem Rev 75:491. doi:10.1021/cr60296a004
Casells I-G, Cataldi T-R-I, Salvi A-M, Desimoni E (1993) Anal Chem 55:3143. doi:10.1021/ac00069a032
Acknowledgments
We gratefully acknowledge the financial support from National Natural Science Foundation of China (project number 50372011), Natural Science Foundation of Liaoning Province, China (project number 20052016), and the China–Ireland Collaboration Funding (cofunded by SFI, RCS, and CMOST). The author (K. Lau) also wishes to thank DCU fellowship award for the financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Peng, XY., Liu, XX., Hua, PJ. et al. Electrochemical codeposition of nickel oxide and polyaniline. J Solid State Electrochem 14, 1–7 (2010). https://doi.org/10.1007/s10008-008-0778-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-008-0778-x