Abstract
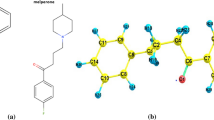
The present work explores the structural parameters and vibrational frequencies as well as molecular interactions of benzodiazepine derivatives, such as clothiapine (CT), clozapine (CZ), and loxapine (LX). Employing fitting experimental data to theoretical results is used to assess the structural parameters of heading composites. The main assignment is passed out according to the overall distribution of energy of the vibrational modes. From the hyper-conjugative interaction, the permanency of the structure had been predicted through natural bond orbital analysis; it is also used to identify the bonding and antibonding regions of the molecules. Moreover, electrostatic potential (ESP), density of states (DOS), and charge transfer occurring of the molecule among HOMO as well as LUMO energy were calculated and presented; utilizing electron localized field (ELF), localized orbital locator (LOL), and reduced density gradient (RDG), the chemical interactive regions are found. Additionally, mean polarizability (αtot), the first-order hyperpolarizability (βtot), and softness and hardness of the entitled compounds were also performed. The interaction between protein–ligand was also predicted by docking studies.





















Similar content being viewed by others
Availability of data and material
NA.
Code availability
NA.
References
Dourlat J, Liu WQ, Gresh N, Garbay C, Novel (2007) Med Chem Lett 17:2527–2530
Brodin T, Fick J, Jonsson M, Klaminder (2013) Science 339:814–815
Dong Z, Senn DB, Moran RE (2013) Shine Regul Toxicol Pharmacol 65:60–67
De Almeida CA, Brenner CGB, Minetto L, Mallmann CA, Martins AF (2013) Chemosphere 93:2349–2355
Fent K, Weston AA (2006) Caminada. Aquat Toxicol 76:122–159
Calisto V, Esteves VI (2009) Chemosphere 77:1257–1274
Kosjek T, Perko S, Zupanc M, Zanoški Hren M, Dragicevic TL, Žigon D, Kompare B (2012) Water Res 46:355–368
Huerta-Fontela M, Galceran MT (2010) J Chromatogr A 1217:4212–4222
Baker DR, Kasprzyk-Hordern B (2013) Sci Total Environ 454–455:442–456
Mendoza A (2014) López de Alda M, González-Alonso S, Mastroianni N, Barceló D, Valcárcel Y. Chemosphere 95:247–255
Chi Q, Dong S (1994) Anal Chim Acta 285(1–2):125–133
Karyakin AA, Karyakina EE, Schuhmann W (1999) Electroanalysis 11(8):553–557
Sokic-Lazic D, Minteer SD (2008) Biosens Bioelectron 24(4):939–944
McDowell JJH (1976) Acta Crystall Sect B 32:5
Levy LB (1992) J Polym Sci A Polym Chem 30(4):569–576
Ohlow MJ (2011) B. Drug Discov Today 16(3–4):119–131
Jaszczyszyn A et al (2012) Pharmacol Rep 64(1):16–23
Dordio AV, Candeias AJE, Pinto AP, da Costa CT (2009) Ecol Eng 35:290–302
Sevvanthi S, Muthu S, Aayisha S, Ramesh P, Raja MJ (2020) Chem Data Collect 30:10
S. Muthu, M. Prasath, R. Arun Balaji, J. Uma Maheswari International Journal of engineering TomeX 3 (2012) 1584–2673.
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA et al (2010) Gaussian 09, Revision B.01. Gaussian Inc, Wallingford
Dennington R, Keith T, Millam J (2009) GaussView, Version 5. Semichem Inc., Shawnee Mission
Jomroz MH. Vibrational Energy Distribution Analysis, VEDA4, 2004 -2010.
Merrick JP, Moran JD, Radom L (2007) The J Phy Chem A 111:11683–11700
O’Boyle NM, Tenderholt AL, Langner KM (2008) J Comput Chem 29:839–845
Tian Lu, Chen F (2012) J Comput Chem 33:580–592
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJJ (2009) Comput Chem 16:2785–2791
Ravikumar K (2005) Balasubramanian. Sridhar, Quetiapine hemifumarate, Acta Crystallographica Section E 61:o3245–o3248
Verma V, Bannigan P, Lusi M, Crowley CM, Hudson S, Hodnett BK, Davern P (2018) CrystEngComm 20(31):4370–4382
Bhardwaj RM, Johnston BF, Oswald IDH, Florence AJ (2013) Acta Crystallogr Sect C Cryst Struct Commun 69(11):1273–1278
John Wiley & Sons, Inc. SpectraBase; https://spectrabase.com/ (accessed 1/5/2021)
Sylaja B, Gunasekaran S, Srinivasan S (2017) Mater Res Innov 22(4):187–199
Muthu S, Prasath M, Balaji RA (2013) Spectrochim Acta 106:129–145
Alsalme A, Pooventhiran T, Al-Zaqri N, Rao DJ, Rao SS, Thomas R (2020) J Mol Model 26:341
Muthu S, Uma Maheswari J (2012) Acta Part A 92:154–163
Alsalme A, Pooventhiran T, Al-Zaqri N, Rao DJ, Thomas RJ (2020) Mol Liq 114555
Ullah Z, Thomas R (2021) Appl Organomet Chem e6077
Mary YS, Miniyar PB, Mary YS, Resmi KS, Panicker CY, Armakovic S, Armakovic SJ, Thomas R, Surehskumar B (2018) J Mol Struct 1173:469–480
Sureshkumar B, Mary YS, Resmi KS, Panicker CY, Armakovic S, Armakovic SJ, Van Alsenoy C, Narayana B, Suma S (2018) J Mol Struct 1156:336–347
Reed AE, Curtiss LA, Weinhold F (1988) J Chem 88:899–926
Mulliken RS (1995) J Chem Phys 23:1833–1840
Sun YX, Hao QL, Wei WX, Yu ZX, Lu LD, Wang X, Wang YS (2009) J Mol Struct: THEOCHEM 904:74–82
Zhang R, Du B, Sun G, Sun YX (2010) Spectrochim Acta A 75:1115–1124
Kleinman DA (1962) Nonlinear dielectric polarization in optical media. J Phys 126:1977–1979
Matondo A, Thomas R, Tsalu PV, Mukeba CT, Mudogo V (2019) J Mol Graph Model 88:237–246
Fleming I (1976) Frontier orbitals and organic chemical reactions. Wiley, New York
Parr R, Szentpaly L, Liu S (1999) Electrophilicity index. J Am Chem Soc 121:1922–1924
Pooventhiran T, Bhattacharyya U, Rao DJ, Chandramohan V, Karunakar P, Irfan A, Mary YS, Thomas R (2020) Struct Chem
Koopmans TA (1934) Physica 1:104–113
Prabakaran A, Muthu S (2014) Spectrochim Acta A Mol Biomol Spectrosc 118:578–588
Fathima Rizwana B (2019) Johanan Christian Prasana, S. Muthu, Christina Susan Abraham Materials Today: Proceedings 18:1770–1782
Schmider HL, Becke AD (2000) J Mol Struct (THEOCHEM) 51:527
Poater J, Duran M, Sola M, Silvi B (2005) Chem Rev 105:3911–3947
Schmider HL, Becke AD (2002) J Chem Phys 116:3184
Jacobsen H (2008) Can J Chem 86:695–702
Jacobsen H (2009) Can J Chem 87:695–973
Wu P, Chaudret R, Hu X, Yang W (2013) J Chem Theory Comput 9:2226–2234
Contreras-García J, Boto RA, Izquierdo-Ruiz F, Reva I, Woller T, Alonso M (2016) Theor Chem Accounts 135–242
Humphrey W, Dalke A, Schulten K (1996) J Mol Graph 14:33–38
Contreras-García J, Johnson ER, Keinan S, Chaudret R, Piquemal J-P, Beratan DN, YangW, (2011) J Chem Theory Comput 7:625–632
Lu T, Chen F (2012) J Mol Graph Model 38:314–323
Hu Y, Xu X, Jiang Y, Zhang G, Li W, Sun X, Tian WQ, Feng Y (2018) Chem Phys 20:12618–12623
Lipinski CA (2004) Drug Discov Today Technol 1(4):337–341
Daina A, Zoete V (2016) A BOILED-Egg Chem Med Chem 11:1117–1121
Potts RO, Guy RH (1992) Pharm Res 9:663–669
Ghose AK, Viswanadhan VN, Wendoloski JJ (1999) J Comb Chem 1(1):55–68
Ertl P, Rohde B, Selzer P (2000) Fast J Med Chem 43:3714–3717
Filimonov DA, Lagunin AA, Gloriozova TA, Rudik AV, Druzhilovskii DS, Pogodin PV, Poroikov VV (2014) Chem Heterocycl Compd 50(3):444–457
Wang S, Che T, Levit A, Shoichet BK, Wacker D, Roth BL (2018) Nature 555:269–273
Acknowledgements
We extend our appreciation to the Deanship of Scientific Research at King Khalid University (KKU), Saudi Arabia, for funding through research groups program under grant number R.G.P.1/110/42
Author information
Authors and Affiliations
Contributions
Conceptualization: S. Sarala; methodology: S. Sarala, S.K.Geetha; formal analysis and investigation: S. Muthu, Ahmad Irfan; writing—original draft, preparation: S. Sarala; writing—review and editing: S. Muthu; funding acquisition: – NA; resources: S.K. Geetha, Ahmad Irfan; supervision: S. Muthu
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarala, S., Geetha, S.K., Muthu, S. et al. Computational investigation, comparative approaches, molecular structural, vibrational spectral, non-covalent interaction (NCI), and electron excitations analysis of benzodiazepine derivatives. J Mol Model 27, 266 (2021). https://doi.org/10.1007/s00894-021-04877-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-021-04877-z