Abstract
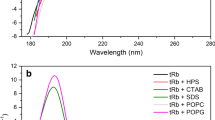
In this research, we present a preliminary computational study of four Dermaseptin-related peptides from the skin exudate of the gliding tree frog Agalychnis spurrelli. Experimentally, the amino acid sequence of these peptides was elucidated through molecular cloning and tandem mass spectrometry and synthetic peptides were assayed against E. coli, S. aureus, and C. albicans to determine their antimicrobial properties. With the sequences on hand, a computational study of the structures was carried out, obtaining their physicochemical properties, secondary structure, and their similarity to other known peptides. A molecular docking study of these peptides was also performed against cell membrane and several enzymes are known to be vital for the organisms. Results showed that Dermaseptin-related peptides are α-helical cationic peptides with an isoelectric point above 9.70 and a positive charge of physiological pH. Introducing theses peptides in a database, it was determined that their identity compared with known peptides range from 36 to 82% meaning these four Dermaseptins are novel peptides. This preliminary study of molecular docking suggests the mechanism of action of this peptide is not given by the inhibition of essential enzymatic pathways, but by cell lysis.

Graphical abstract








Similar content being viewed by others
References
Conlon JM (2012) The potential of frog skin antimicrobial peptides for development into therapeutically valuable anti-infective agents, Chapter 3 In Small Wonders. Peptides for Disease Control; ACS Symposium Series; American Chemical Society, Washington
Lacombe C, Piesse C, Sagan S, Combadière C, Rosenstein Y, Auvynet C (2015) Pachymodulin, a new functional formyl peptide receptor 2 peptidic ligand isolated from frog skin has Janus-like immunomodulatory capacities. J Med Chem 58:1089–1099. https://doi.org/10.1021/jm501018q
Holthausen D, Lee S, Kumar V, Bouvier N, Krammer F, Ellebedy A, Wrammert J, Lowen A, George S, Pillai M, Jacob J (2017) An amphibian host defense peptide is virucidal for human H1 hemagglutinin-bearing influenza viruses. Immunity 46:587–595. https://doi.org/10.1016/j.immuni.2017.03.018
Scorciapino M, Manzo G, Rinaldi A, Sanna R, Casu M, Pantic J, Lukic M, Conlon J (2013) Conformational analysis of the frog skin peptide, plasticin-L1 and its effects on the production of proinflammatory cytokines by macrophages. Biochemistry 52(41):7231–7241. https://doi.org/10.1021/bi4008287
Manzo G, Casu M, Rinaldi M, Montaldo N, Luganini A, Gribaudo G, Scorciapino M (2014) Folded structure and insertion depth of the frog-skin antimicrobial peptide esculentin-1b (1–18) in the presence of differently charged membrane-mimicking micelles. J Nat Prod 77(11):2410–2417. https://doi.org/10.1021/np5004406
Samgina T, Vorontsov E, Gorshkov V, Hakalehto E, Hanninen O, Zubarev R, Lebedevl A (2012) Composition and antimicrobial activity of the skin peptidome of Russian Brown Frog Rana temporaria. J Proteome Res 11(12):6213–6222. https://doi.org/10.1021/pr300890m
Marani M, Dourado F, Quelemes P, Rodrigues A, Gomes M, Alves E, Costa L, Rodrigues A, Barroso E, Eaton P, Figueiró J, Bentes R, Delerue-Matos C, Leite J (2015) Characterization and biological activities of ocellatin peptides from the skin secretion of the frog Leptodactylus pustulatus. J Nat Prod 78(7):1495–1504. https://doi.org/10.1021/np500907t
Stutz K, Muller A, Hiss J, Schneider P, Blatter M, Pfeiffer B, Posselt G, Kanfer G, Kornmann B, Wrede P, Altmann K, Wessler S, Schneider G (2017) Peptide-membrane interaction between targeting and lysis. ACS Chem Biol 12(9):2254–2259. https://doi.org/10.1021/acschembio.7b00504
Ortega-Andrade HM (2008) Agalychnis spurrelli Boulenger (Anura: Hylidae): variación, distribución y sinonimia. Papéis Avulsos de Zoologia 48:103–1117
Duellman WE (2001) Hylid Frogs of Middle America. Society for the Study of Amphibians and Reptiles, Ithaca
Gray AR (1997) Observations on the biology of Agalychnis spurrelli from the Caribbean lowlands of Costa Rica. Observaciones sobre la biología de Agalychnis spurrelli de las tierras bajas del caribe de Costa Rica. Journal of the International Herpetological Society 22:61–70
MECN (2010) Serie Herpetofauna del Ecuador: El Choco Esmeraldeño. Monografía. Museo Ecuatoriano de Ciencias Naturales. Quito-Ecuador 5:1–232
Gasteiger E, Hoogland C, Gattiker A, Duvaud S, Wilkins MR, Appel RD, Bairoch A (2005) Protein Identification and Analysis Tools on the ExPASy Server. In: En Walker J (ed) The Proteomics Protocols Handbook. Springer, London
Innovagen (2015) Peptide property calculator, Innovagen AB, https://pepcalc.com/
Bio-Synthesis. (2018). Peptide Property Calculator, Bio-Synthesis Inc. https://www.biosyn.com/peptidepropertycalculatorlanding.aspx
Drozdetskiy A, Cole C, Procter J, Barton GJ (2015) JPred4: a protein secondary structure prediction server. Nucleic Acids Res 43:389–394
Jones DT (1999) Protein secondary structure prediction based on position-specific scoring matrices. J Mol Biol 292:195–202
The PyMOL Molecular Graphics System (2017) Version 2.0 Schrödinger, LLC. https://pymol.org/2/support.html?#citing
ChembioDraw (2009) PerkinElmer Informatics. http://informatics.perkinelmer.com/Support/KnowledgeBase/details/Default?TechNote=3411
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Fox DJ (2009) Gaussian 09. Revision A.01. Gaussian, Inc, Wallingford https://doi.org/111\
Berman H, Westbrook J, Feng Z, Gilliland G, Bhat T, Weissig H, Shindyalov I, Bourne P (2000) The Protein Data Bank. Nucleic Acids Res 28(1):235–242
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J Comput Chem 31(2):455–461
Gill SC, von Hippel PH (1989) Calculation of protein extinction coefficients from amino acid sequence data. Anal Biochem 182(2):319–326
Cuesta S, Arias J, Gallegos F, Proaño C, Blasco-Zúñiga A, Rivera M, Meneses L (2019) Molecular modeling of Dermaseptine SP2 extracted from Agalychnis spurrelli. InfoANALÍTICA 7(1):41–56. https://doi.org/10.26807/ia.v7i1.95
Guruprasad K, Reddy BVB, Pandit MW (1990) Correlation between stability of a protein and its dipeptide composition: a novel approach for predicting in vivo stability of a protein from its primary sequence. Protein Eng 4:155–161
Li RF, Lu ZF, Sun YN, Chen SH, Yi YJ, Zhang HR, Yang SY, Yu GH, Huang L, Li CN (2016) Molecular design, structural analysis and antifungal activity of derivatives of peptide CGA-N46. Interdisciplinary sciences, computational life sciences 8(3):319–326
Fieser TM, Tainer JA, Geysen HM, Houghten RA, Lerner RA (1987) Influence of protein flexibility and peptide conformation on reactivity of monoclonal anti-peptide antibodies with a protein alpha-helix. Proc Natl Acad Sci U S A 84(23):8568–8572
Karplus PA, Schulz GE (1985) Prediction of chain flexibility in proteins: a tool for the selection of peptide antigens. Naturwissenschaften 72:212–213
Huang F, Nau WM (2003) A conformational flexibility scale for amino acids in peptides. Angew Chem Int Ed Eng 42(20):2269–2272
Melchiorri P, Negri L (2009) Amphibian peptides, Encyclopedia of Neuroscience. Elsevier Academic Press, San Diego
Park MS, Gao C, Stern HA (2011) Estimating binding affinities by docking/scoring methods using variable protonation states. Proteins 79(1):304–314. https://doi.org/10.1002/prot.22883
Pantsar T, Poso A (2018) Binding affinity via docking: fact and fiction. Molecules 23(8):1899
Wishart DS, Feunang YD, Guo AC, Lo EJ, Marcu A, Grant JR, Sajed T, Johnson D, Li C, Sayeeda Z, Assempour N, Iynkkaran I, Liu Y, Maciejewski A, Gale N, Wilson A, Chin L, Cummings R, Le D, Pon A, Knox C, Wilson M (2017) DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res 46:1074–1082
Wang G (2014) Human antimicrobial peptides and proteins. Pharmaceuticals 7(5):545–594
Bechinger B, Gorr SU (2017) Antimicrobial peptides: mechanisms of action and resistance. J Dent Res 96(3):254–260
Acknowledgments
CPB obtained the peptides sequences in Queen’s University Belfast funded by the Natural drug discovery group and the Ecuadorian Secretariat of Science and Technology (SENESCYT) through a scholarship. Collection and rearing of frogs in Ecuador were done under permits of the Ecuadorian Ministerio de Ambiente (MAE): 001-13 IC-FAU-DNB/MA, 003-11 IC-FAU-DNB/MA, 005-15 IC-FAU-DNB/MA (Issued to the Centro Jambatu). This research is part of the project “Conservation of Ecuadorian amphibian diversity and sustainable use of its genetic resources”, which involves MAE, Ikiam-Universidad Regional Amazónica, Queen’s University Belfast, and Centro Jambatu, and help of the Global Environmental Facility (GEF) and “Programa de las Naciones Unidas para el Desarrollo” (PNUD).
Funding
This research was funded by two research grants to L.M and M.R. of the Dirección General Académica of the Pontificia Universidad Católica del Ecuador, projects: QINV0035-IINV529010100 and QIV0046-IINV529010100.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This paper belongs to Topical Collection QUITEL 2018 (44th Congress of Theoretical Chemists of Latin Expression)
Electronic supplementary material
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Cuesta, S., Gallegos, F., Arias, J. et al. Molecular modeling of four Dermaseptin-related peptides of the gliding tree frog Agalychnis spurrelli. J Mol Model 25, 260 (2019). https://doi.org/10.1007/s00894-019-4141-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-019-4141-1