Abstract
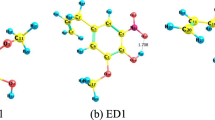
Luteolin is an excellent antioxidant found in a wide variety of natural foods, such as honey and pollen. In this work, the effect of the surrounding environments on the structure and antioxidative activity of luteolin was carried out using density functional theory (DFT) calculation. The studied environments are gas, benzene, chloroform, pyridine, acetonitrile, ethanol, DMSO, and water. The structure of the luteolin monomer in different environments was optimized. The hydrogen-bond was especially focused, and the antioxidative capacity of luteolin was analyzed from the thermodynamic aspect. It is found that: (1) hydrogen atom transfer (HAT) is the most thermodynamically favorable mechanism in the gas, benzene, and chloroform phases, while sequential proton loss electron transfer (SPLET) is more favorable than HAT and single electron transfer followed by proton transfer (SET-PT) in pyridine, acetonitrile, ethanol, DMSO, and water phases. (2) The 4’−OH group could more strongly participate in the free radical scavenging process of luteolin than other OH groups, while the 5−OH group is the least favored one in the studied environments. (3) The antioxidative capacity of luteolin is strongest in pyridine.


Similar content being viewed by others
References
Fang YZ, Yang S, Wu G (2002) Nutrition 18:872
Dizdaroglu M, Jaruga P, Birincioglu M, Rodriguez H (2002) Free Radic Biol Med 32:1102
Andersen ØM, Markham KR (eds) (2006) Flavonoids: chemistry, biochemistry and applications. Taylor and Francis, Boca Raton
Ross JA, Kasum CM (2002) Annu Rev Nutr 22:19
Arts MJ, Haenen GR, Voss HP, Bast A (2001) Food Chem Toxicol 39:787
Villano D, Fernández-Pachón MS, Moyá ML, Troncoso AM, García-Parrilla MC (2007) Talanta 71:230
Knekt P, Kumpulainen J, Järvinen R, Rissanen H, Heliövaara M, Reunanen A et al (2002) Am J Clin Nutr 76:560
Nenadis N, Sigalas MP (2011) Food Res Int 44:114
Benayahoum A, Amira-Guebailia H, Houache O (2013) J Mol Model 19:2285
Nenadis N, Tsimidou MZ (2012) Food Res Int 48:538
Rocca MVL, Rutkowski M, Ringeissen S et al (2016) J Mol Model 22:250
Vargas-Sánchez RD, Mendoza-Wilson AM, Torrescano-Urrutia GR, Sánchez-Escalante A (2015) Comput Theor Chem 1066:7
Lengyel J, Rimarčík J, Vagánek A, Klein E (2013) Phys Chem Chem Phys 15:10895
Zheng YZ, Zhou Y, Liang Q, Chen DF, Guo R, Xiong CL et al (2017) Dyes Pigments 141:179
Vagánek A, Rimarčík J, Dropková K, Lengyel J, Klein E (2014) Comput Theor Chem 1050:31
Zheng YZ, Deng G, Chen DF, Liang Q, Guo R, Fu ZM (2018) Food Chem 240:323
Xue Y, Zheng Y, An L, Dou Y, Liu Y (2014) Food Chem 151:198
Wang G, Xue Y, An L, Zheng Y, Dou Y, Zhang L et al (2015) Food Chem 171:89
Laskar RA, Sk I, Roy N, Begum NA (2010) Food Chem 122:233
Sarkar A, Middya TR, Jana AD (2012) J Mol Model 18:2621
Wright JS, Johnson ER, DiLabio GA (2001) J Am Chem Soc 123:1173
Leopoldini M, Russo N, Toscano M (2011) Food Chem 125:288
Stepanić V, Trošelj KG, Lučić B, Marković Z, Amić D (2013) Food Chem 141:1562
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, VothGA SP, Dannenberg JJ, Dapprich S, DanielsAD FÖ, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian09, revision B.01. Gaussian Inc, Wallingford
Zhao Y, Truhlar DG (2008) Acc Chem Res 41:157–167
Espinosa E, Molins E, Lecomte C (1998) Chem Phys Lett 285:170
Bader RFW (1994) Atoms in molecules: a quantum theory. Clarendon, Oxford
Lu T, Chen F (2012) J Comput Chem 33:580
Bartmess JE (1994) J Phys Chem 98:6420
Rimarčík J, Lukeš V, Klein E, Ilčin M (2010) THEOCHEM J Mol Struct 952:25
Parker VD (1992) J Am Chem Soc 114:7458
Pauling L (1960) The nature of the chemical bond. Cornell University Press, New York
Roohi H, Nowroozi AR, Anjomshoa E (2011) Comput Theor Chem 965:211
Rozas I, Alkorta I, Elguero J (2000) J Am Chem Soc 122:11154
Pacios LF (2004) J Phys Chem A 108:1177
Acknowledgments
This work was supported by the Natural Science Foundation of China (21703035), Earmarked Fund for China Agriculture Research System (CARS-44-KXJ7), and the Fujian Agriculture and Forestry University Foundation for excellent youth teachers (xjq201715).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, YZ., Chen, DF., Deng, G. et al. The surrounding environments on the structure and antioxidative activity of luteolin. J Mol Model 24, 149 (2018). https://doi.org/10.1007/s00894-018-3680-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-018-3680-1