Abstract
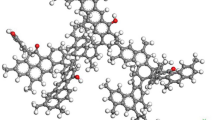
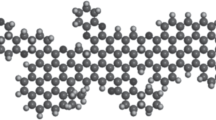
The mechanism for the competitive adsorption of CH4 and CO2 on coal vitrinite (DV-8, maximum vitrinite reflectance R o,max = 0.58%) was revealed through simulation and experimental methods. A saturated state was reached after absorbing 17 CH4 or 22 CO2 molecules per DV-8 molecule. The functional groups (FGs) on the surface of the vitrinite can be ranked in order of decreasing CH4 and CO2 adsorption ability as follows: [−CH3] > [−C=O] > [−C–O–C–] > [−COOH] and [−C–O–C–] > [−C=O] > [−CH3] > [−COOH]. CH4 and CO2 distributed as aggregations and they were both adsorbed at the same sites on vitrinite, indicating that CO2 can replace CH4 by occupying the main adsorption sites for CH4–vitrinite. High temperatures are not conducive to the adsorption of CH4 and CO2 on vitrinite. According to the results of density functional theory (DFT) and grand canonical Monte Carlo (GCMC) calculations, vitrinite has a higher adsorption capacity for CO2 than for CH4, regardless of whether a single-component or binary adsorbate is considered. The equivalent adsorption heat (EAH) of CO2–vitrinite (23.02–23.17) is higher than that of CH4–vitrinite (9.04–9.40 kJ/mol). The EAH of CO2–vitrinite decreases more rapidly with increasing temperature than the EAH of CH4–vitrinite does, indicating in turn that the CO2–vitrinite bond weakens more quickly with increasing temperature than the CH4–vitrinite bond does. Simulation data were found to be in good accord with the corresponding experimental results.













Similar content being viewed by others
Change history
03 June 2019
The authors have retracted this article [1] because it significantly overlaps with a previously published article [2]. All authors agree with this retraction.
References
Tian H, Li T, Zhang T, Xiao X (2016) Int J Coal Geol 156:36–49
Kang Y, Huang F, You L, Li X, Gao B (2016) Int J Coal Geol 154:123–135
An H, Wei XR, Wang GX, Massarotto P, Wang FY, Rudolph V, Golding SD (2015) Int J Coal Geol 152:15–24
Busch A, Gensterblum Y (2011) Int J Coal Geol 87(2):49–71
Cui XJ, Bustin RM, Dipple G (2004) Fuel 83:293–303
Qiu NX, Xue Y, Guo Y, Sun WJ, Chu W (2012) Comput Theor Chem 992:37–47
Liu XQ, Xue Y, Tian ZY, Mo JJ, Qiu NX, Chu W, Xie HP (2013) Appl Surf Sci 285:190–197
Domazetis G, Raoarun M, James BD (2007) Energy Fuel 21:2531–2542
Liu J, Qu WQ, Zheng CG (2010) Energy Fuel 24:4425–4429
Xua H, Chu W, Huang X, Sun WJ, Jiang CF, Liu ZQ (2016) Appl Surf Sci 375:196–206
Liu YY, Wilcox J (2011) Environ Sci Technol 45:809–814
Zhao Y, Jiang C, Chu W (2012) Int J Min Sci Technol 22(6):757–761
Thierfelder C, Witte M, Blankenburg S, Rauls E, Schmidt WG (2011) Surf Sci 605(7):746–749
Ricca A, Bauschlicher CW (2006) Chem Phys 324(2):455–458
Rubes M, Kysilka J, Nachtigall P, Bludský O (2010) Phys Chem Chem Phys 12(24):6438–6444
Liu XQ, Tian ZY, Chu W, Xue Y (2014) Acta Phys Chim Sin 30(2):251–256
Liu F, Chu W, Sun W (2012) J Nat Gas Chem 21:708–712
Goodman AL, Campus LM, Schroeder KT (2005) Energy Fuel 19(2):471–476
Goodman AL, Favors RN, Larsen JW (2006) Energy Fuel 20(6):2537–2543
Goodman AL (2009) Energy Fuel 23(2):1101–1106
Hu H, Li X, Fang Z, Wei N, Li Q (2010) J Coal Sci Eng 7:58–63
Sun PD (2001) J Coal Sci Eng 7:58-63
White CM, Smith DH, Jones KL, Goodman AL, Jikich SA, LaCount RB, DuBose SB, Ozdemir E, Morsi BI, Schroeder KT (2005) Energy Fuel 19(3):659–724
Nishino J (2001) Fuel 80(5):757–764
Larsen JW, Flowers RA, Hall PJ, Carlson G (1997) Energy Fuel 11(5):998-1002
Larsen JW (2004) Int J Coal Geol 57(1):63-70
Xiang JH, Zeng FG, Liang HZ, Li B, Song XX (2014) Sci Chi Earth Sci 57(8):1749–1759
Yu CF, Chen KL, Cheng HC, Chen WH (2016) Comput Mater Sci 117:127–138
Mcnamara JP, Sharma R, Vincent MA, Hillier IH, Morgado CA (2008) Phys Chem Chem Phys 10(1):128–135
Lithoxoos GP, Labropoulos A, Peristeras LD, Kanellopoulos N, Samios J, Economou IG (2010) J Supercrit Fluid 55(2):510–523
Kowalczyk P, Tanaka H, Kaneko K, Do DD (2005) Langmuir 21(12):5639–5646
Cao DP, Gao GT, Wang WC (2000) J Chem Ind Eng 1:23–30
Li X, Lin B, Xu H (2014) Int J Min Sci Technol 24(1):17–22
Yu S, Yan-ming Z, Wu L (2017) Appl Surf Sci 396:291–302
Zhao Y, Feng Y, Zhang X (2016) Fuel 165:19–27
Valentini P, Schwartzentruber TE, Cozmuta I (2011) Surf Sci 605(23):1941–1950
Carlson GA (1992) Energy Fuel 6(6):771–778
Nakamura K (1993) Energy Fuel 7(3):347–350
Rogel E, Carbognani L (2003) Energy Fuel 17(2):378–386
Delley B (2000) J Chem Phys 113(18):7756–7764
Grimme S, Ehrlich S, Goerigk L (2011) J Comput Chem 32(7):1456–1465
Xu H, Chu W, Huang X, Xia H, Sun W, Jiang C, Liu Z (2016) Appl Surf Sci 375:196–206
Alonso JA (2000) Chem Rec 100(2):637–678
Mark SS, Andrew ED (1992) J Chem Phys 97(5):3386–3398
Reuse FA, Khanna SN (1995) Chem Phys Lett 234(1–3):77–81
Castro M, Jamorski C, Salahub DR (1997) Chem Phys Lett 271(1–3):133–142
Perdew JP, Burke K, Wang Y (1996) Phys Rev B 54(23):16533
Grimme S (2006) J Comput Chem 27(15):1787–1799
Inada Y, Orita H (2008) J Comput Chem 29(2):225–232
Tambach TJ, Mathews JP, van Bergen F (2009) Energy Fuel 23(10):4845–4847
Saghafi A, Faiz M, Roberts D (2007) Int J Coal Geol 70(1):240–254
Einstein A (1905) Ann Phys Berl 17:549–560
Li W, Zhu Y (2014) Energy Fuel 28(6):3645–3654
Li W, Zhu Y (2014) Energ Source Part A 36(15):1650–1658
Cosoli P, Ferrone M, Pricl S, Fermeglia M (2008) Chem Eng J 145(1):86–92
Li W, Zhu YM, Wang G, Wang Y, Liu Y (2015) J Mol Model 21(8):188
Kaplan IG (1986) Theory of molecular interactions. New York: Elsevier 1986:178-251
Liu SS, Meng ZP (2015) J China Coal Soc 40(6):1422–1427
Hao S, Wen J, Yu X, Chu W (2013) Appl Surf Sci 264:433–442
Zhang S, Sang S, Yang Z (2009) J China Univ Min Technol 5:022
Crosdale PJ, Beamish BB, Valix M (1998) Int J Coal Geol 35(1):147–158
Jiang WP, Cui YJ, Zhang Q, Zhong L, Hui LY (2007) J China Coal Soc 3:015
Chen CG, Wei XW, Xian XF (2000) J Chongqing Uni (Nat Sci Ed) 23:77–79
Hu H, Du L, Xing Y, Li X (2017) Fuel 187:220–228
Haus JW, Kehr KW (1987) Phys Rep 150(5):263–406
Baumgärtner A, Moon M (1989) Eur Phys Lett 9(3):203
Jing W (2010) Molecular simulation of adsorption and diffusion of methane in deformed coal. Taiyuan University of Technology Press: 61-62
Zeng YY, Zhang BJ (2008) Acta Phys Chim Sin 24(8):1493–1497
Acknowledgements
The authors greatly appreciate Prof. F.G. Zeng (Key Laboratory of Coal Science and Technology of Ministry of Education, Taiyuan University of Technology, Shanxi Province), who helped us to access the Materials Studio software package. This work was supported by the National Natural Science Foundation of China (nos. 41430317, 41072117).
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have retracted this article because it significantly overlaps with a previously published article. All authors agree with this retraction.
About this article
Cite this article
Yu, S., Bo, J. & Jiahong, L. RETRACTED ARTICLE: Simulations and experimental investigations of the competitive adsorption of CH4 and CO2 on low-rank coal vitrinite. J Mol Model 23, 280 (2017). https://doi.org/10.1007/s00894-017-3442-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-017-3442-5