Abstract
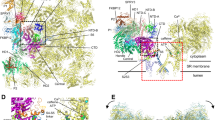
Ryanodine receptors (RyRs) are the largest known ion channels, and are of central importance for the release of Ca2+ from the sarco/endoplasmic reticulum (SR/ER) in a variety of cells. In cardiac and skeletal muscle cells, contraction is triggered by the release of Ca2+ into the cytoplasm and thus depends crucially on correct RyR function. In this work, in silico mutants of the RyR pore were generated and MD simulations were conducted to examine the impact of the mutations on the Ca2+ distribution. The Ca2+ distribution pattern on the luminal side of the RyR was most affected by G4898R, D4899Q, E4900Q, R4913E, and D4917A mutations. MD simulations with our wild-type model and various ion species showed a preference for Ca2+ over other cations at the luminal pore entrance. This Ca2+-accumulating characteristic of the luminal RyR side may be essential to the conductance properties of the channel.



Similar content being viewed by others
References
Zalk R, Lehnart SE, Marks AR (2007) Modulation of the ryanodine receptor and intracellular calcium. Annu Rev Biochem 76:367–385
Lanner JT, Georgiou DK, Joshi AD, Hamilton SL (2010) Ryanodine receptors: structure, expression, molecular details, and function in calcium release. Cold Spring Harb Perspect Biol 2:a003996
Van Petegem F (2012) Ryanodine receptors: structure and function. J Biol Chem 287:31624–31632
Fink RH, Veigel C (1996) Calcium uptake and release modulated by counter-ion conductances in the sarcoplasmic reticulum of skeletal muscle. Acta Physiol Scand 156:387–396
Wu S, Ibarra MCA, Malicdan MCV, Murayama K, Ichihara Y, Kikuchi H, Nonaka I, Noguchi S, Hayashi YKH, Nishino I (2006) Central core disease is due to RYR1 mutations in more than 90% of patients. Brain 129:1470–1480
Zhou H, Jungbluth H, Sewry CA, Feng L, Bertini E, Bushby K, Straub V, Roper H, Rose MR, Brockington M, Kinali M, Manzur A, Robb S, Appleton R, Messina S, D’Amico A, Quinlivan R, Swash M, Müller CR, Brown S, Treves S, Muntoni F (2007) Molecular mechanisms and phenotypic variation in RYR1-related congenital myopathies. Brain 130:2024–2036
Kimlicka L, Van Petegem F (2011) The structural biology of ryanodine receptors. Sci China Life Sci 54:712–724
Ludtke SJ, Serysheva II, Hamilton SL, Chiu W (2005) The pore structure of the closed RyR1 channel. Structure 13:1203–1211
Samsó M, Wagenknecht T, Allen PD (2005) Internal structure and visualization of transmembrane domains of the RyR1 calcium release channel by cryo-EM. Nat Struct Mol Biol 12:539–544
Samsó M, Feng W, Pessah IN, Allen PD (2009) Coordinated movement of cytoplasmic and transmembrane domains of RyR1 upon gating. PLoS Biol 7:e85
Efremov RG, Leitner A, Aebersold R, Raunser S (2015) Architecture and conformational switch mechanism of the ryanodine receptor. Nature 517:39–43
Zalk R, Clarke OB, des Georges A, Grassucci RA, Reiken S, Mancia F, Hendrickson WA, Frank J, Marks AR (2015) Structure of a mammalian ryanodine receptor. Nature 517:44–49
Yan Z, Bai X-C, Yan C, Wu J, Li Z, Xie T, Peng W, Yin C-C, Li X, Scheres SHW, Shi Y, Yan N (2015) Structure of the rabbit ryanodine receptor RyR1 at near-atomic resolution. Nature 517:50–55
Gillespie D, Xu L, Wang Y, Meissner G (2005) (De)constructing the ryanodine receptor: modeling ion permeation and selectivity of the calcium release channel. J Phys Chem B 109:15598–15610
Gillespie D (2008) Energetics of divalent selectivity in a calcium channel: the ryanodine receptor case study. Biophys J 94:1169–1184
Welch W, Rheault S, West DJ, Williams AJ (2004) A model of the putative pore region of the cardiac ryanodine receptor channel. Biophys J 87:2335–2351
Ramachandran S, Serohijos AWR, Xu L, Meissner G, Dokholyan NV (2009) A structural model of the pore-forming region of the skeletal muscle ryanodine receptor (RyR1). PLoS Comput Biol 5:e1000367
Ramachandran S, Chakraborty A, Xu L, Mei Y, Samsó M, Dokholyan NV, Meissner G (2013) Structural determinants of skeletal muscle ryanodine receptor gating. J Biol Chem 288:6154–6165
Schilling R, Fink RHA, Fischer WB (2014) MD simulations of the central pore of ryanodine receptors and sequence comparison with 2B protein from coxsackie virus. Biochim Biophys Acta 1838:1122–1131
Shirvanyants D, Ramachandran S, Mei Y, Xu L, Meissner G, Dokholyan NV (2014) Pore dynamics and conductance of RyR1 transmembrane domain. Biophys J 106:2375–2384
Jiang Y, Lee A, Chen J, Cadene M, Chait BT, MacKinnon R (2002) The open pore conformation of potassium channels. Nature 417:523–526
Zhao M, Li P, Li X, Zhang L, Winkfein RJ, Chen SR (1999) Molecular identification of the ryanodine receptor pore-forming segment. J Biol Chem 274:25971–25974
Gao L, Balshaw D, Xu L, Tripathy A, Xin C, Meissner G (2000) Evidence for a role of the lumenal M3-M4 loop in skeletal muscle Ca(2+) release channel (ryanodine receptor) activity and conductance. Biophys J 79:828–840
Wang Y, Xu L, Pasek DA, Gillespie D, Meissner G (2005) Probing the role of negatively charged amino acid residues in ion permeation of skeletal muscle ryanodine receptor. Biophys J 89:256–265
Xu L, Wang Y, Yamaguchi N, Pasek DA, Meissner G (2008) Single channel properties of heterotetrameric mutant RyR1 ion channels linked to core myopathies. J Biol Chem 283:6321–6329
Mead-Savery FC, Wang R, Tanna-Topan B, Chen SR, Welch W, Williams AJ (2009) Changes in negative charge at the luminal mouth of the pore alter ion handling and gating in the cardiac ryanodine-receptor. Biophys J 96:1374–1387
Lee SY, Lee A, Chen J, MacKinnon R (2005) Structure of the KvAP voltage-dependent K+ channel and its dependence on the lipid membrane. Proc Natl Acad Sci USA 102:15441–15446
Pronk S, Pall S, Schulz R, Larsson P, Bjelkmar P, Apostolov R, Shirts MR, Smith JC, Kasson PM, van der Spoel D, Hess B, Lindahl E (2013) GROMACS 4.5: a high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 29:845–854
Schuler LD, Daura X, van Gunsteren WF (2001) An improved GROMOS96 force field for aliphatic hydrocarbons in the condensed phase. J Comput Chem 22:1205–1218
Krüger J, Fischer WB (2008) Exploring the conformational space of Vpu from HIV-1: a versatile adaptable protein. J Comput Chem 29:2416–2424
Chandrasekhar I, Kastenholz M, Lins RD, Oostenbrink C, Schuler LD, Tieleman DP, van Gunsteren WF (2003) A consistent potential energy parameter set for lipids: dipalmitoylphosphatidylcholine as a benchmark of the GROMOS96 45A3 force field. Eur Biophys J 32:67–77
Chen R, Chung SH (2013) Complex structures between the N-type calcium channel (Ca2.2) and ω-conotoxin GVIA predicted via molecular dynamics. Biochemistry 52:3765–3772
Tang L, Gamal El-Din TM, Payandeh J, Martinez GQ, Heard TM, Scheuer T, Zheng N, Catterall WA (2014) Structural basis for Ca2+ selectivity of a voltage-gated calcium channel. Nature 505:56–61
Goonasekera SA, Beard NA, Groom L, Kimura T, Lyfenko AD, Rosenfeld A, Marty I, Dulhunty AF, Dirksen RT (2007) Triadin binding to the C-terminal luminal loop of the ryanodine receptor is important for skeletal muscle excitation contraction coupling. J Gen Physiol 130:365–378
Smith JS, Imagawa T, Ma J, Fill M, Campbell KP, Coronado R (1988) Purified ryanodine receptor from rabbit skeletal muscle is the calcium-release channel of sarcoplasmic reticulum. J Gen Physiol 92:1–26
Lindsay AR, Manning SD, Williams AJ (1991) Monovalent cation conductance in the ryanodine receptor-channel of sheep cardiac muscle sarcoplasmic reticulum. J Physiol 439:463–480
Tinker A, Williams AJ (1992) Divalent cation conduction in the ryanodine receptor channel of sheep cardiac muscle sarcoplasmic reticulum. J Gen Physiol 100:479–493
Acknowledgments
W.B.F. thanks the National Science Council for financial support (NSC101-2112-M-010-002-MY3). R.S. was supported by a Ph.D. scholarship from the Heidelberg Medical School and the Baden-Württemberg-Stipendium. R.H.A.F is grateful for the financial support of the German Excellence Initiative II–Global Networks.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 2845 kb)
Rights and permissions
About this article
Cite this article
Schilling, R., Fink, R.H.A. & Fischer, W.B. Interaction of ions with the luminal sides of wild-type and mutated skeletal muscle ryanodine receptors. J Mol Model 22, 37 (2016). https://doi.org/10.1007/s00894-015-2906-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2906-8