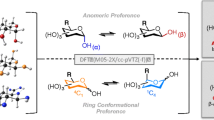
Abstract
We examined the conformational preferences of the 2-O-sulfated-3,6-α-D-anhydrogalactose (compound I) and two 1,3 linked disaccharides constituting-κ or ι-carrageenans using density functional and ab initio methods in gas phase and aqueous solution. Systematic modifications of two torsion angles leading to 324 and 144 starting geometries for the compound I and each disaccharide were used to generate adiabatic maps using B3LYP/6-31G(d). The lower energy conformers were then fully optimized using B3LYP, B3PW91 and MP2 with several basis sets. Overall, we discuss the impact of full relaxation on the energy and structure of the dominant conformations, present the performance comparison with previous molecular mechanics calculations if available, and determine whether our results are impacted, when polarization and diffuse functions are added to the 6-31G(d) basis set, or when the MP2 level of theory is used.




Similar content being viewed by others
References
Genicot-Joncour S, Poinas A, Richard O, Potin P, Rudolph B, Kloareg B, Helbert W (2009) Am Soc Plant Biol 151:1609–1616
Stortz CA (2006) Carbohydr Res 341:2531–2542
Stortz CA (2005) In: Yarema KJ (ed) In: Handbook of carbohydrate engineering. Taylor and Francis, Boca Raton, pp 211–245
Campo VL, Kawano DF, da Silva DB Jr, Carvalho I (2009) Carbohydr Polymers 77:167–180
Strati GL, Willett JL, Momany FA (2002) Carbohydr Res 337:1851–1859
Navarro DA, Stortz CA (2003) Carbohydr Res 338:2111–2118
Le Questel JY, Cros S, Mackie W, Perez S (1995) Int J Biol Macromol 17:161–175
Parra E, Caro HN, Jimènez-Barbero J, Martin-Lomas M, Bernabé M (1990) Carbohydr Res 208:83–92
Urbani R, Di Blas A, Cesàro A (1993) Int J Biol Macromol 15:24–29
Ueda K, Ochiai H, Imamura A, Nakagawa S (1995) Bull Chem Soc Jpn 68:95–106
Ueda K, Brady JW (1996) Biopolymers 38:461–469
Stortz CA, Cerezo AS (2000) J Carbohydr Chem 19:1115–1130
Stortz CA, Cerezo AS (1994) J Carbohydr Chem 13:235
Stortz CA, Cerezo AS (1995) An Asoc Quim Argent 83:171–181
Stortz CA, Cerezo AS (1998) J Carbohydr Chem 17:1405–1419
Stortz CA (1999) Carbohydr Res 322:77–86
Ragazzi M, Ferro D, Provasoli A (1986) J Comput Chem 7:105–112
Ferro DR, Pumilia P, Cassinari A, Ragazzi M (1995) Int J Biol Macromol 17:131–136
Huige CJM, Altona C (1995) J Comput Chem 16:56–79
Ferro DR, Pumilia P, Ragazzi M (1997) J Comput Chem 18:351–367
Lamba D, Glover S, Mackie W, Rashid A, Sheldrick B, Pérez S (1994) Glycobiology 4:151–163
Ueda K, Saiki M, Brady JW (2001) J Phys Chem B 105:8629–8638
Ueda K, Iwama K, Nakayama H (2001) Bull Chem Soc Jpn 74:2269–2277
Schnupf U, Willett JL, Bosma WB, Momany FA (2007) Carbohydr Res 342:2270–2285
Ha SN, Madson LJ, Brady JW (1988) Biopolymers 27:1927–1952
Tran V, Buleon A, Imberty A, Pérez S (1989) Biopolymers 28:679–690
Dowd MK, Zeng J, French AD, Reilly PJ (1992) Carbohydr Res 230:223–244
Kouwijzer MLC, Grootenhuis PDJ (1995) J Phys Chem 99:13426–13436
Kuttel MM, Naidoo K (2005) J Phys Chem B 109:7468–7474
Gould IR, Bettley HA, Bryce RA (2007) J Comput Chem 28:1965–1973
Landström J, Widmalm G (2010) Carbohydr Res 345:330–333
Hatcher E, Säwén E, Widmalm G, MacKerell AD Jr (2011) J Phys Chem B 115:597–608
Perič-Hassler L, Hansen HS, Baron R, Hünenberger PH (2010) Carbohydr Res 345:1781–1801
Stortz CA, Johnson GP, French AD, Csonka GI (2009) Carbohydr Res 344:2217–2228
Momany FA, Appell M, Willett JL, Schnupf U, Bosma WB (2006) Carbohydr Res 341:525–537
Da Silva CO, Nascimento MAC (2004) Carbohydr Res 339:113–122
French AD, Johnson GP, Kelterer AM, Csonka GI (2005) Tetrahedron-Asymmetry 16:577–586
Momany FA, Willett JL (2000) J Comput Chem 21:1204–1219
Strati GL, Willett JL, Momany FA (2002) Carbohydr Res 337:1833–1849
Bosma WB, Appell M, Willett JL, Momany FA (2006) J Mol Struct (THEOCHEM) 776:1–19
Bosma WB, Appell M, Willett JL, Momany FA (2006) J Mol Struct (THEOCHEM) 776:13–24
Appell M, Strati GL, Willett JL, Momany FA (2004) Carbohydr Res 339:537–551
Appell M, Willett JL, Momany FA (2005) Carbohydr Res 340:459–468
Schnupf U, Willett JL, Bosma WB, Momany FA (2007) Carbohydr Res 342:196–216
Momany FA, Appell MA, Strati GL, Willett JL (2004) Carbohydr Res 339:553–567
Hricovini M, Scholtzovà E, Bizik F (2007) Carbohydr Res 342:1350–1356
Lii J-H, Ma B, Allinger NL (1999) J Comput Chem 20:1593–1603
Tvaroska I, Taravel FR, Utille JP, Carver JP (2002) Carbohydr Res 337:353–367
Tissot B, Salpin JY, Martinez M, Gaigeot MP, Daniel R (2006) Carbohydr Res 341:598–609
Momany FA, Appell M, Willett JL, Bosma WB (2005) Carbohydr Res 340:1638–1655
Pogány P, Kovács A (2009) Carbohydr Res 344:1745–1752
Dauchez M, Lagant P, Derreumaux P, Vergoten G, Sekkal M (1994) Spectrochim Acta A Mol Spectrosc 50:105–118
Dauchez M, Derreumaux P, Lagant P, Vergoten G, Sekkal M, Legrand P (1994) Spectrochim Acta A Mol Biomol Spectrosc 50:87–104
Kuttel JM, Brady W, Naidoo K (2002) J Comput Chem 23:1236–1243
Schnupf U, Willett JL, Momany FA (2010) J Comput Chem 31:2087–2097
Yousfi N, Sekkal-Rahal M, Sayede A, Springborg M (2010) J Comput Chem 31:1312–1320
Gonçalves PFB, Stassen H (2002) J Comput Chem 23:706–714
Langella E, Rega N, Improta R, Crescenzi O, Barone B (2002) J Comput Chem 23:650–661
Curutchet C, Cramer CJ, Truhlar DG, Ruiz-Lopez MF, Rinaldi D, Orozco M, Luque FJ (2003) J Comput Chem 24:284–297
Fattebert JL, Gygi F (2002) J Comput Chem 23:662–666
Csonka GI, French AD, Johnson GP, Stortz CA (2009) J Chem Theor Comput 5:679–692
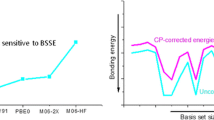
Kristyan S, Pulay P (1994) Chem Phys Lett 229:175–180
IUPAC-IUB Commission on Biochemical Nomenclature (1971) Arch Biochem Biophys 145:405
Arnott S, Scott WE Rees DA, McNab CGA (1974) J Mol Biol 90:253–267
Janaswamy S, Chandrasekaran R (2001) Carbohydr Res 335:181–194
Janaswamy S, Chandrasekaran R (2002) Carbohydr Res 337:523–535
French AD, Dowd MK (1993) J Mol Struct (THEOCHEM) 286:183–201
Onsager L (1936) J Am Chem Soc 58:1486–1493
Foresman JB, Frisch A (1996) Gaussian94 user’s guide. Exploring chemistry with electronic structure methods, 2nd edn. Gaussian, Inc, Pittsburgh
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, Revision D.02. Gaussian, Inc, Pittsburgh, PA
Golden Software, Inc (2002) Surface mapping system. Golden Software, Inc. Golden, CO
Marchessault RH, Pérez S (1979) Biopolymers 18:2369–2374
Navarro DA, Stortz CA (2008) Carbohydr Res 343:2292–2298
Engelsen SB, Kocà J, Braccini I, du Hervé PC, Pérez S (1995) Carbohydr Res 276:1–30
Dowd MK, French AD, Reilly PJ (1994) Carbohydr Res 264:1–19
Wong CHS, Siu FM, Ma NL, Tsang CW (2001) J Mol Struct (THEOCHEM) 536:227–234
Lii J-H, Chen K-H, Durkin KA, Allinger NL (2003) J Comput Chem 24:1473–1489
Lii J-H, Chen K-H, Allinger NL (2003) J Comput Chem 24:1504–1513
Momany FA, Willett JL (2000) Carbohydr Res 326:194–209
Momany FA, Willett JL (2000) Carbohydr Res 326:210–226
Navarro DA, Stortz CA (2005) Carbohydr Res 340:2030–2038
Jeffrey GA (1990) Acta Crystallogr B46:89–103
Longchambon F, Gillier-Pandraud H (1977) Acta Crystallogr B32:1822–1826
Van Eijck BP, Mooij WTM, Kroon J (2001) J Phys Chem B 105:10573–10578
McDonnell C, López O, Murphy P, Fernández Bolanõs JG, Hazell R, Bols M (2004) J Am Chem Soc 126:12374–12385
Barone V, Cossi M, Tomasi J (1998) J Comput Chem 19:404–417
Rashid A, Mackie W (1992) Carbohydr Res 223:147–155
Millane RP, Chandrasekaran R, Arnott S, Dea ICM (1988) Carbohydr Res 182:1–17
Acknowledgments
N. B-B. B thanks D. J. Fox from his helpful suggestions to perform calculations and M. S-R thanks the Alexander von Humboldt foundation (Bonn) for grants
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure SI.1
Structures including hydrogen bonds distances (in Å) of the four conformers of compound I in the gas phase, (a) obtained from the map 2a, and (b) after full optimization (DOC 151 kb)
Figure SI.2
Structures including hydrogen bonds distances (in Å) of the four conformers of compound I in water, (a) obtained from the map 2b, and (b) after full optimization (DOC 153 kb)
Figure SII.1
Structures including hydrogen bonds distances (in Å) of the two conformers of compound II in the gas phase, (a) obtained from the map 3a, and (b) after full optimization (DOC 126 kb)
Figure SII.2
Structures including hydrogen bonds distances (in Å) of the two conformers of compound II in water, (a) obtained from the map 3b, and (b) after full optimization (DOC 132 kb)
Figure SIII.1
Structures including hydrogen bonds distances (in Å) of the two conformers of compound III in the gas phase, (a) obtained from the map 4a, and (b) after full optimization (DOC 135 kb)
Figure SIII.2
Structures including hydrogen bonds distances (in Å) of the three conformers of compound III in water, (a) obtained from the map 4b, and (b) after full optimization (DOC 172 kb)
Rights and permissions
About this article
Cite this article
Bestaoui-Berrekhchi-Berrahma, N., Derreumaux, P., Sekkal-Rahal, M. et al. Density functional conformational study of 2-O-sulfated 3,6 anhydro-α-D-galactose and of neo-κ- and ι-carrabiose molecules in gas phase and water. J Mol Model 19, 893–904 (2013). https://doi.org/10.1007/s00894-012-1621-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1621-y